Abstract
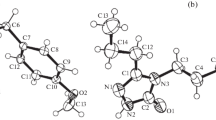
Terfenadine, C32H41NO2, 1, contains an α,α-diphenyl-4-piperidinomethanol moiety, which is related to the H1-receptor blocking activity, facilitating its prior use as an antihistamine drug. In addition to its bioactivity, terfenadine is useful as a model, small-molecule crystalline solid for studying several material properties. Despite a history of therapeutic use, the absence of a crystal structure has limited current studies of the physicochemical behavior of this material. In the present manuscript, the elusive X-ray crystal structure of 1 was solved and refined at 296 K using single crystals grown from a co-solvent mixture of acetonitrile:methanol:ethanol (0.50:0.25:0.25). Terfenadine crystallizes in the monoclinic space group P21/n, and exhibits a chair conformation of the piperidine ring and a gauche conformation of the n-butyl chain. Hydrogen bonds between O–H⋯O and O–H⋯N, along with weak van der Waals interactions between C16–H16B⋯H16B’–C16’ and C2–H2⋯H16A–C16A were confirmed using Hirshfeld-Surface analysis. Differential scanning calorimetry and X-ray powder diffraction confirmed that the crystal structure reported herein was that of the most thermodynamically stable monotropic polymorph of terfenadine (form I).
Graphic Abstract
The elusive single crystal structure of terfenadine is solved and refined. Characterization using differential scanning calorimetry and powder X-ray diffraction confirms the structure to be the thermodynamically most stable polymorph (form I).







Similar content being viewed by others
Data Availability
CCDC 2060883 contains the crystallographic data for compound 1 reported in this article. These data can be obtained free of charge at www.ccdc.cam.ac.uk/conts/retrieving.html or from the Cambridge Crystallographic Data Center (CCDC), 12 Union Road, Cambridge CB2 1EZ, UK: fax: +44(0)1223-336,033; email: deposit@ccdc.cam.ac.uk.
References
Zhang M, Ter Laak A, Timmerman H (1993) Structure-activity relationships within a series of analogues of the histamine H1-antagonist terfenadine. Eur J Med Chem 28(2):165–173
Carr A, Meyer D (1982) Synthesis of terfenadine. Arzneimittelforschung 32(9a):1157–1159
Pratt CM, Hertz RP, Ellis BE, Crowell SP, Louv W, Moyé L (1994) Risk of developing life-threatening ventricular arrhythmia associated with terfenadine in comparison with over-the-counter antihistamines, ibuprofen and clemastine. Am J Card 73(5):346–352
Bookwala M, DeBoyace K, Buckner IS, Wildfong PLD (2020) Predicting density of amorphous solid materials using molecular dynamics simulation. AAPS PharmSciTech 21(3):1–11
Amidon GL, Lennernäs H, Shah VP, Crison JR (1995) A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res 12(3):413–420
DeBoyace K, Buckner IS, Gong Y, Ju TR, Wildfong PLD (2018) Modeling and prediction of drug dispersability in polyvinylpyrrolidone-vinyl acetate copolymer using a molecular descriptor. J Pharm Sci 107(1):334–343
DeBoyace K, Zdaniewski C, Wildfong PLD (2018) Differential scanning calorimetry isothermal hold times can impact interpretations of drug-polymer dispersability in amorphous solid dispersions. J Pharm Bio Ana 150:43–50
Dudognon E, Bama J-A, Affouard F (2019) Molecular mobility of terfenadine: investigation by dielectric relaxation spectroscopy and molecular dynamics simulation. Mol Pharm 16(11):4711–4724
Moore MD, Wildfong PLD (2011) Informatics calibration of a molecular descriptors database to predict solid dispersion potential of small molecule organic solids. Int J Pharm 418(2):217–226
Vasa DM, Dalal N, Katz JM, Roopwani R, Nevrekar A, Patel H, Buckner IS, Wildfong PLD (2014) Physical characterization of drug:polymer dispersion behavior in polyethylene glycol 4000 solid dispersions using a suite of complementary analytical techniques. J Pharm Sci 103(9):2911–2923
Lin Y, Cogdill RP, Wildfong PLD (2009) Informatic calibration of a materials properties database for predictive assessment of mechanically activated disordering potential for small molecule organic solids. J Pharm Sci 98(8):2696–2708
Pajula K, Lehto V-P, Ketolainen J, Korhonen O (2012) Computational approach for fast screening of small molecular candidates to inhibit crystallization in amorphous drugs. Mol Pharm 9(10):2844–2855
Bruker (1998) SMART and SAINT (Version 6.02). Bruker AXS Inc, Madison, Wisconsin
Sheldrick GM (1996) Sadabs. University of Göttingen, Germany
Sheldrick GM (2015) Crystal structure refinement with SHELXL. Acta Crystallogr C 71(1):3–8
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr A 64(1):112–122
Hübschle CB, Sheldrick GM, Dittrich B (2011) ShelXle: a Qt graphical user interface for SHELXL. J Appl Crystallogr 44(6):1281–1284
Leităo M, Canotilho J, Cruz M, Pereira J, Sousa A, Redinha J (2002) Study of polymorphism from DSC melting curves; polymorphs of terfenadine. J Therm Anal Calorim 68(2):397–412
Yonemochi E, Hoshino T, Yoshihashi Y, Terada K (2005) Evaluation of the physical stability and local crystallization of amorphous terfenadine using XRD–DSC and micro-TA. Thermochim Acta 432(1):70–75
Tessler L, Goldberg I (2005) The methanol disolvate and the dihydrate of fexofenadine, an antihistamine drug. Acta Crystallogr C 61(12):707–710
Ellena J, Punte G, Rivero B, Remedi M, De Vargas E, De Rossi R (1995) X-ray structural and spectroscopic investigation of 1-piperidine-2, 4-dinitrobenzene. J Chem Crystallogr 25(12):801–805
Parvez M, Bakhtiar M, Baqir M, Zia-ur-Rehman M (2014) Syntheses and crystal structures of four novel thiophene/phenyl-piperidine hybrid chalcones. J Chem Crystallogr 44(11):580–585
Allen F, Kennard O, Watson D, Brammer L, Orpen A, Taylor R (1987) Molecular structures and dimensions. J Chem Soc Perkin Trans 2:S1–S19
Danovich D, Shaik S, Neese F, Echeverria J, Aullon G, Alvarez S (2013) Understanding the nature of the CH··· HC interactions in alkanes. J Chem Theory Comput 9(4):1977–1991
Li J, Wang Y, An L, Chen J, Yao L (2018) Direct observation of CH/CH van der Waals interactions in proteins by NMR. J Am Chem Soc 140(9):3194–3197
Mirzaei M, Nikpour M, Bauzá A, Frontera A (2015) On the Importance of C–H/π and C–H⋅⋅⋅ H–C interactions in the solid state structure of 15-lipoxygenase inhibitors based on eugenol derivatives. ChemPhysChem 16(10):2260–2266
Spackman MA, Jayatilaka D (2009) Hirshfeld surface analysis. CrystEngComm 11(1):19–32
Salpage SR, Smith MD, Shimizu LS (2016) Crystal structures and Hirshfeld surface analyses of 6-substituted chromones. J Chem Crystallogr 46(4):170–180
Janiak C (2000) A critical account on π–π stacking in metal complexes with aromatic nitrogen-containing ligands. J Chem Soc Dalton Trans 21:3885–3896
Montgomery MJ, O’Connor TJ, Tanski JM (2015) Crystal structures of 4-chloropyridine-2-carbonitrile and 6-chloropyridine-2-carbonitrile exhibit different intermolecular π-stacking, C–H⋯ Nnitrile and C–H⋯ Npyridine interactions. Acta Crystallogr E 71(7):852–856
Quinn TR, Tanski JM (2014) Crystal structure of 4-chloro-2-iodoaniline. Acta Crystallogr E 70(9):944–945
Badwan AA, Al Kaysi HN, Owais LB, Salem MS, Arafat TA (1990) Terfenadine. In: Anal Profiles Drug Subst, vol 19. Elsevier, pp 627–662
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of MB, AG, JAA, PLDW. MB, AG, JAA, PLDW have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bookwala, M., Gumireddy, A., Aitken, J.A. et al. Single Crystal Structure of Terfenadine Form I. J Chem Crystallogr 52, 81–88 (2022). https://doi.org/10.1007/s10870-021-00892-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-021-00892-3