Abstract
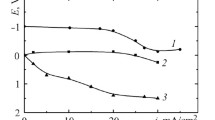
We have studied the cathodic processes on a platinum electrode in ozonized sulfuric acid solutions of different concentrations. The kinetic parameters of corrosion processes have been determined. The cathodic processes on a platinum electrode in the presence of ozone are more intense by a factor of 1.5 than in the presence of oxygen. Ozone increases the possibility of chemical reaction with the help of generation of OH− ions. In addition, we have proposed a scheme for the action of ozone in acid media.
Similar content being viewed by others
REFERENCES
B. A. Gru, G. O. Tatarchenko, N. F. Tyupalo, and V. S. Kuzub, “Effect of ozonization and some additives on the corrosion-electrochemical behavior of 12Kh18N10T steel in sulfuric acid,” Fiz.-Khim. Mekh. Mater., 27, No.2, 5–9 (1991).
G. Tatarchenko, Zh. Makarova, and A. Kuzyukov, “Specific features of the corrosion-electrochemical behavior of titanium and its alloys with aluminum in hydrochloric acid solutions,” Fiz.-Khim. Mekh. Mater., 38, No.3, 97–101 (2002).
G. O. Tatarchenko, I. N. Shapovalova, and N. F. Tyupalo, “Selection of corrosion-proof structural materials in ozonized acetic acid solutions,” Fiz.-Khim. Mekh. Mater., 38, Special Issue No. 3, 143–147 (2002).
G. Tatarchenko, K. Cherkas, and A. Kuzyukov, “Corrosion-electrochemical behavior of nickel in ozonized sulfuric acid solutions,” Fiz.-Khim. Mekh. Mater., 38, No.1, 98–100 (2002).
G. Tatarchenko, K. Cherkas, and M. V. Pavlova, “On the passivation of chromium in ozonized sulfuric acid solutions,” Fiz.-Khim. Mekh. Mater., 37, Special Issue No. 2, 97–101 (2001).
V. V. Skorchelletti, Theoretical Foundations of Metallic Corrosion [in Russian], Khimiya, Leningrad (1973).
B. E. Conway and S. Gottesfeld, “Real condition of oxidized platinum electrodes,” J. Chem. Soc. Faraday Trans., Ser. I, 69, 1090–1107 (1973).
Ya. M. Kolotyrkin, G. M. Florianovich, and A. I. Kasperovich, “Application of oxygen for the protection of structural metallic materials against corrosion in aqueous media,” in: VINITI Series in Corrosion and Corrosion Protection [in Russian], Vol. 8, VINITI, Moscow (1981), pp. 3–45.
Yu. N. Mikhailovskii, “New ideas of the electrochemical mechanism of corrosion inhibition by oxygen-containing inorganic oxidizers,” Zashch. Met., 20, No.2, 179–190 (1984).
S. D. Razumovskii, Oxygen: Its Elementary Forms and Properties [in Russian], Khimiya, Moscow (1979).
L. I. Antropov, Theoretical Electrochemistry [in Russian], Vysshaya Shkola, Moscow (1984).
G. M. Florianovich, “Theory of the corrosion of metals and alloys as a basis of optimization and their protection under service conditions,” in: Trans. Fifth Session “Engineering and Chemical Science for Advanced Technologies” [in Russian], Vol. 1, International School for Advanced Training, Moscow (1999), pp. 231–251.
V. A. Marichev, “A study of the contact electric resistance of platinum in aqueous solutions,” Zashch. Met., 38, No.5, 488–495 (2002).
Yu. M. Tyurin, V. I. Naumov, A. L. Galkin, and V. V. Izotova, “Charged and uncharged films on platinum in hydrochloric acid solutions and their role in the reactions of liberation of chlorine and oxygen,” Elektrokhimiya, 26, No.10, 1324–1332 (1990).
Author information
Authors and Affiliations
Additional information
__________
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol. 40, No. 5, pp. 43–47, September– October, 2004.
Rights and permissions
About this article
Cite this article
Tatarchenko, G.O. Parameters of Oxidation-Reduction Processes in Ozonized Acid Media. Mater Sci 40, 617–622 (2004). https://doi.org/10.1007/s11003-005-0090-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11003-005-0090-1