Abstract
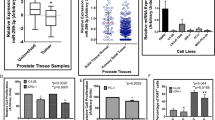
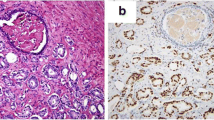
Previous studies have shown that androgen receptor (AR) is involved in the progression of prostate cancer (CaP) by several mechanisms. However, how AR is regulated has not been fully understood. In this study, miR-185 was found to be down-regulated in clinical CaP samples. Targets prediction revealed that AR had putative complementary sequences to miR-185, which was confirmed by the following dual luciferase reporter assay. Overexpression of miR-185 could reduce the expression of AR protein but not mRNA in LNCaP cells. The proliferation of LNCaP cells was inhibited by overexpression of miR-185. Cell cycle analysis revealed cell cycle arrest at G0/G1 phase. The invasive and migration abilities of cells could also be suppressed by miR-185. Furthermore, miR-185 inhibited tumorigenicity in a CaP xenografts model. CDC6, one target of AR and an important regulatory molecule for cell cycle, was found to be down-regulated by overexpression of miR-185. Our findings suggest that miR-185 could function as a tumor-suppressor gene in CaP by directly targeting AR, and act as a potential therapeutic target for CaP.






Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ (2007) Cancer statistics, 2007. CA Cancer J Clin 57:43–66
Corona G, Baldi E, Maggi M (2011) Androgen regulation of prostate cancer: where are we now? J Endocrinol Invest 34:232–243
Feldman BJ, Feldman D (2001) The development of androgen-independent prostate cancer. Nat Rev Cancer 1:34–45
Nadiminty N, Tummala R, Lou W, Zhu Y, Zhang J, Chen X, eVere White RW, Kung HJ, Evans CP, Gao AC (2012) MicroRNA let-7c suppresses androgen receptor expression and activity via regulation of Myc expression in prostate cancer cells. J Biol Chem 287:1527–1537
Klotz L (2011) Active surveillance for prostate cancer: a review. Arch Esp Urol 64:806–814
Suzuki H, Ueda T, Ichikawa T, Ito H (2003) Androgen receptor involvement in the progression of prostate cancer. Endocr Relat Cancer 10:209–216
Gao W (2010) Androgen receptor as a therapeutic target. Adv Drug Deliv Rev 62:1277–1284
Attard G, Richards J, de Bono JS (2011) New strategies in metastatic prostate cancer: targeting the androgen receptor signaling pathway. Clin Cancer Res 17:1649–1657
Lai EC (2002) Micro RNAs are complementary to 3′ UTR sequence motifs that mediate negative post-transcriptional regulation. Nat Genet 30:363–364
Budd WT, Weaver DE, Anderson J, Zehner ZE (2012) MicroRNA dysregulation in prostate cancer: network analysis reveals preferential regulation of highly connected nodes. Chem Biodivers 9:857–867
Bushati N, Cohen SM (2007) MicroRNA functions. Annu Rev Cell Dev Biol 23:175–205
Zhang Y, Wang Z, Chen M, Peng L, Wang X, Ma Q, Ma F, Jiang B (2012) MicroRNA-143 targets MACC1 to inhibit cell invasion and migration in colorectal cancer. Mol Cancer 11:23
Nadiminty N, Tummala R, Lou W, Zhu Y, Shi XB, Zou JX, Chen H, Zhang J, Chen X, Luo J, deVere White RW, Kung HJ, Evans CP and Gao AC (2012) MicroRNA let-7c is downregulated in prostate cancer and suppresses prostate cancer growth. PLoS One 7:e32832
Bousquet M, Nguyen D, Chen C, Shields L, Lodish HF (2012) MicroRNA-125b transforms myeloid cell lines by repressing multiple mRNA. Haematologica 97:1713–1721
Imam JS, Buddavarapu K, Lee-Chang JS, Ganapathy S, Camosy C, Chen Y, Rao MK (2010) MicroRNA-185 suppresses tumor growth and progression by targeting the Six1 oncogene in human cancers. Oncogene 29:4971–4979
Kirby M, Hirst C, Crawford ED (2011) Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract 65:1180–1192
Lamont KR, Tindall DJ (2010) Androgen regulation of gene expression. Adv Cancer Res 107:137–162
Zegarra-Moro OL, Schmidt LJ, Huang H, Tindall DJ (2002) Disruption of androgen receptor function inhibits proliferation of androgen-refractory prostate cancer cells. Cancer Res 62:1008–1013
Wu CT, Altuwaijri S, Ricke WA, Huang SP, Yeh S, Zhang C, Niu Y, Tsai MY, Chang C (2007) Increased prostate cell proliferation and loss of cell differentiation in mice lacking prostate epithelial androgen receptor. Proc Natl Acad Sci U S A 104:12679–12684
Dehm SM, Tindall DJ (2006) Molecular regulation of androgen action in prostate cancer. J Cell Biochem 99:333–344
Lin B, Wang J, Hong X, Yan X, Hwang D, Cho JH, Yi D, Utleg AG, Fang X, Schones DE, Zhao K, Omenn GS, Hood L (2009) Integrated expression profiling and ChIP-seq analyses of the growth inhibition response program of the androgen receptor. PLoS ONE 4:e6589
Ngan S, Stronach EA, Photiou A, Waxman J, Ali S, Buluwela L (2009) Microarray coupled to quantitative RT-PCR analysis of androgen-regulated genes in human LNCaP prostate cancer cells. Oncogene 28:2051–2063
Visakorpi T, Hyytinen E, Koivisto P, Tanner M, Keinanen R, Palmberg C, Palotie A, Tammela T, Isola J, Kallioniemi OP (1995) In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat Genet 9:401–406
Shiota M, Yokomizo A, Naito S (2011) Increased androgen receptor transcription: a cause of castration-resistant prostate cancer and a possible therapeutic target. J Mol Endocrinol 47:R25–R41
Koochekpour S (2010) Androgen receptor signaling and mutations in prostate cancer. Asian J Androl 12:639–657
Marques RB, Dits NF, Erkens-Schulze S, van Ijcken WF, van Weerden WM, Jenster G (2011) Modulation of androgen receptor signaling in hormonal therapy-resistant prostate cancer cell lines. PLoS ONE 6:e23144
Ueda T, Bruchovsky N, Sadar MD (2002) Activation of the androgen receptor N-terminal domain by interleukin-6 via MAPK and STAT3 signal transduction pathways. J Biol Chem 277:7076–7085
Lamont KR, Tindall DJ (2011) Minireview: alternative activation pathways for the androgen receptor in prostate cancer. Mol Endocrinol 25:897–907
Golias C, Iliadis I, Peschos D, Charalabopoulos K (2009) Amplification and co-regulators of androgen receptor gene in prostate cancer. Exp Oncol 31:3–8
Gregory CW, He B, Johnson RT, Ford OH, Mohler JL, French FS, Wilson EM (2001) A mechanism for androgen receptor-mediated prostate cancer recurrence after androgen deprivation therapy. Cancer Res 61:4315–4319
Wang D, Qiu C, Zhang H, Wang J, Cui Q, Yin Y (2010) Human MicroRNA oncogenes and tumor suppressors show significantly different biological patterns: from functions to targets. PLoS One 5 30:5(9). 10.1371/journal.pone.0013067
Catto JW, Alcaraz A, Bjartell AS, De Vere White R, Evans CP, Fussel S, Hamdy FC, Kallioniemi O, Mengual L, Schlomm T, Visakorpi T (2011) MicroRNA in prostate, bladder, and kidney cancer: a systematic review. Eur Urol 59:671–681
Fendler A, Stephan C, Yousef GM, Jung K (2011) MicroRNAs as regulators of signal transduction in urological tumors. Clin Chem 57:954–968
Liu M, Lang N, Chen X, Tang Q, Liu S, Huang J, Zheng Y, Bi F (2011) miR-185 targets RhoA and Cdc42 expression and inhibits the proliferation potential of human colorectal cells. Cancer Lett 301:151–160
Liao JM, Lu H (2011) Autoregulatory suppression of c-Myc by miR-185-3p. J Biol Chem 286:33901–33909
Ayad NG (2005) CDKs give Cdc6 a license to drive into S phase. Cell 122:825–827
Blow JJ, Gillespie PJ (2008) Replication licensing and cancer–a fatal entanglement? Nat Rev Cancer 8:799–806
Bai VU, Cifuentes E, Menon M, Barrack ER, Reddy GP (2005) Androgen receptor regulates Cdc6 in synchronized LNCaP cells progressing from G1 to S phase. J Cell Physiol 204:381–387
Mallik I, Davila M, Tapia T, Schanen B, Chakrabarti R (2008) Androgen regulates Cdc6 transcription through interactions between androgen receptor and E2F transcription factor in prostate cancer cells. Biochim Biophys Acta 1783:1737–1744
Jin F, Fondell JD (2009) A novel androgen receptor-binding element modulates Cdc6 transcription in prostate cancer cells during cell-cycle progression. Nucleic Acids Res 37:4826–4838
Balk SP, Knudsen KE (2008) AR, the cell cycle, and prostate cancer. Nucl Recept Signal 6:e001
Fu M, Wang C, Li Z, Sakamaki T, Pestell RG (2004) Minireview: cyclin D1: normal and abnormal functions. Endocrinology 145:5439–5447
Xu Y, Chen SY, Ross KN, Balk SP (2006) Androgens induce prostate cancer cell proliferation through mammalian target of rapamycin activation and post-transcriptional increases in cyclin D proteins. Cancer Res 66:7783–7792
Acknowledgments
This work was supported by grants from National Natural Science Foundation of China (No. 30973006), National Natural Science Foundation of China for Youths (No. 81001136), and Shanghai Committee of Science and Technology General Program for Medicine (No. 10411963600).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qu, F., Cui, X., Hong, Y. et al. MicroRNA-185 suppresses proliferation, invasion, migration, and tumorigenicity of human prostate cancer cells through targeting androgen receptor. Mol Cell Biochem 377, 121–130 (2013). https://doi.org/10.1007/s11010-013-1576-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1576-z