Abstract
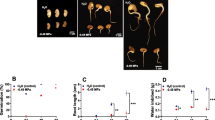
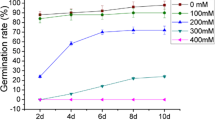
Many of the world’s important crops such as soybean are now widely distributed, beyond their original zones of natural selection and chilling temperature is one of the major abiotic environmental factors which limit their growth and yield. Various molecular mechanisms, underlying chilling stress and plant responses are yet to be discovered. Chilling temperatures impart maximum damage to the soybean seeds, sown in wet soils, during imbibition, causing poor germination rate, reduced seedling emergence, decreased seedling vigor, and ultimately severe loss in yield. Here we report the changes in proteome of soybean seeds exposed to low temperature (4°C) during imbibition in a known chilling-resistant soybean cultivar Z22, using two-dimensional gel electrophoresis (2-DE) and Matrix-Assisted Laser Ionization Time of Flight Mass Spectrometry (MALDI-TOF/MS). Total 40 protein spots were differentially expressed in response to low temperature, in which, 25 protein spots were up-regulated and fifteen protein spots were down-regulated. According to the search in primary databases, these proteins were a part of many metabolic pathways including, cell defense, energy, protein synthesis, cell growth/division, storage, transcription and transport. To investigate the change in mRNA levels during imbibition at 4°C, quantitative RT-PCR of nine up-regulated protein genes was carried out, which clearly demonstrated an increase in the transcript levels of five genes, whereas no change was observed for other four genes. Results of this study provide some invaluable insights about the response of soybean seeds to low temperature imbibition which include; (1) alcohol dehydrogenase I and RAB21 might contribute in decreasing the effect of anoxia, resulting from water up-take during imbibition, (2) stress-related proteins such as LEA and GST24 probably played a pivotal role in confronting low temperature stress and (3) expression of some crucial enzymes (malate dehydrogenase and phosphoenolpyruvate carboxylase) involved in TCA cycle was enhanced, which might be beneficial for seeds, in stress conditions during germination.






Similar content being viewed by others
Abbreviations
- 2-DE:
-
Two-dimensional gel electrophoresis
- ACN:
-
Acetonitrile
- ADH I:
-
Alcohol dehydrogenase I
- CBB:
-
Coomassie brilliant blue
- CHAPS:
-
3-[(3-Cholamidopropyl) dimethylammonio]-1-propanesulfonate
- DREB:
-
Dehydration responsive element
- DTT:
-
Dithiothreitol
- IPTG:
-
Isopropyl-β-d-thiogalactopyranoside
- LEA:
-
Late embryogenesis abundant
- MALDI-TOF/MS:
-
Matrix-Assisted Laser Ionization Time of Flight Mass Spectrometer
- NCBI:
-
National Center for Biotechnology Information
- PMSF:
-
Phenylmethylsulphonyl fluoride
- SBP:
-
Sucrose binding protein
- SCOF-1:
-
Cold-inducible zinc finger protein-1
- SDS–PAGE:
-
Sodium dodecyl sulphate–polyacrylamide gel electrophoresis
- TCA cycle:
-
Tricarboxylic acid cycle
- M r :
-
Relative molecular mass
- pI:
-
Isoelectric point
References
Agarwal PK, Agarwal P, Reddy MK, Sopory SK (2006) Role of DREB transcription factors in abiotic and biotic stress tolerance in plants. Plant Cell Rep 25:1263–1274
Auld DL, Bettis BL, Crock JE, Kephart D (1988) Planting date and temperature effects on germination, and seed yield of Chickpea. Agron J 80:909–914
Becker B et al (2006) Influence of the photoperiod on redox regulation and stress responses in Arabidopsis thaliana L. (Heynh.) plants under long- and short-day conditions. Planta 224:380–393
Blackman SA, Wettlaufer SH, Obendorf RL, Leopold AC (1991) Maturation proteins associated with desiccation tolerance in soybean. Plant Physiol 96:868–874
Bonk T, Humeny A (2001) MALDI-TOF-MS analysis of protein and DNA. Neuroscientist 7:6. doi:10.1177/107385840100700104
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Bungard RA, Mcneil D, Morton JD (1997) Effects of chilling, light and nitrogen-containing compounds on germination, rate of germination and seed imbibition of Clematis vitalba L. Ann Bot 79:643–650
Cardol P, Gonzalez-Halphen D, Reyes-Prieto A, Baurain D, Matagne RF, Remacle C (2005) The mitochondrial oxidative phosphorylation proteome of Chlamydomonas reinhardtii deduced from the genome sequencing project. Plant Physol 137:447–459
Chen GA, Gharib TG, Huang CC, Taylor JMG, Misek DE, Kardia SLR, Giordano TJ, Iannettoni MD, Orringer MB, Hanash SM, Beer DG (2002) Discordant protein and mRNA expression in lung adenocarcinomas. Mol Cell Proteomics 1:304–313
Christie AH, Allen GG, Gregory JT (2001) Induction of vacuolar ATPase and mitochondrial ATP synthase by aluminum in an aluminum-resistant cultivar of wheat. Plant Physiol 125:2068–2077
Collins MO, Yu L, Choudhary JS (2007) Analysis of protein phosphorylation on a proteome-scale. Proteomics 7:2751–2768
Downie B, Gurusinghe S, Dahal P, Thacker RR, Snyder JC, Nonogaki H, Yim K, Fukanaga K, Alvarado V, Bradford KJ (2003) Expression of a galactinol synthase gene in tomato seeds is up-regulated before maturation desiccation and again after imbibition whenever radicle protrusion is prevented. Plant Physiol 131:1347–1359
Gietl C (1992) Malate dehydrogenase isoenzymes: cellular locations and role in the flow of metabolites between the cytoplasm and cell organelles. Biochem Biophys Acta 1100:217–234
Ginger A, Swire-Clark WR, Marcotte JR (1999) The wheat LEA protein Em functions as an osmoprotective molecule in Saccharomyces cerevisia. Plant Mol Biol 39:117–128
Goh CH, Nam HG, Park YS (2004) Stress memory in plants: a negative regulation of stomatal response and transient induction of rd22 gene to light in abscisic acid-entrained Arabidopsis plants. Plant J 36:240–255
Gonzalez DO, Vodkin LO (2007) Specific elements of the glyoxylate pathway play a significant role in the functional transition of the soybean cotyledon during seedling development. BMC Genomics 8:468–490
Greaves JA (1996) Improving suboptimal temperature tolerance in maize—the search for variation. J Exp Bot 296:307–323
Hameister S et al (2007) Transcriptional regulation of the NADP dependent malate dehydrogenase: comparative genetics and identification of DNA-binding proteins. J Mol Evol 65:437–455
Hamilton CA, Taylor GJ, Good AG (2002) Vacuolar H+ -ATPase, but not mitochondrial F1F0-ATPase, is required for NaCl tolerance in Saccharomyces cerevisiae. FEMS Microbiol Lett 208:227–232
Hobbs PR, Obendorf RL (1972) Interaction of initial seed moisture and inhibitional temperature on germination and productivity of soybean. Crop Sci 12:664–667
Hurkman WJ, Tanaka CK (1986) Solubilization of plant membrane proteins for analysis by two-dimensional gel electrophoresis. Plant Physiol 81:802–806
Hurkman WJ, Tanaka CK (2007) High-resolution two-dimensional gel electrophoresis: a cornerstone of plant proteomics. In: Samaj J, Thelen J (eds) Plant proteomics. Springer-Verlag, Berlin, pp 14–28
Imai L, Chang A, Otha EA, Bray TM (1996) A lea-class gene of tomato confers salt and freezing tolerance when expressed in Saccharomyces cerevisiae. Gene 170:243–248
Ismail AM, Hall AE (2002) Variation in traits associated with chilling tolerance during emergence in cowpea germplasm. Field Crops Res 77:99–113
Ismail AM, Hall AE, Close T (1999) Allelic variation of a dehydrin gene cosegregates with chilling tolerance during seedling emergence. PNAS 96:13566–13570
Jennings P, Saltveit ME (1994) Temperature effect on imbibition and germination of cucumber (Cucumis sativus) seed. J Am Soc Hortic Sci 119:464–467
Jung SH, Lee JY, Lee DH (2003) Use of SAGE technology to reveal changes in gene expression in Arabidopsis leaves undergoing cold stress. Plant Mol Biol 52:553–567
Kim JC, Lee SH, Cheong YH, Yoo CM, Lee SI, Chun HJ, Yun DJ, Hong JC, Lee SY, Lim CO, Cho MJ (2001) A novel cold-inducible zinc finger protein from soybean, SCOF-1, enhances cold tolerance in transgenic plants. Plant J 25:247–259
Lai SLN, Grlyani VH, Khurana PJ (2008) Overexpression of HVA1 gene from barley generates tolerance to salinity and water stress in transgenic mulberry (Morus indica). Transgenic Res 17:651–663
Leopold AC (1980) Temperature effect on soybean imbibition and leakage. Plant Physiol 65:1096–1098
Marrs KA (1996) The functions and regulation of glutathione S-transferases in plants. Ann Rev Plant Physiol Plant Mol Biol 47:127–158
McDonald MB (1988) Soybean seed imbibition: water absorption by seed parts. Crop Sci 28:993–997
Moons A (2005) Regulatory and functional interactions of plant growth regulators and plant glutathione S-transferases (GSTs). Vitam Horm 72:155–202
Newsholme SJ, Maleeft BF, Steiner S, Anderson NL, Schwartz LW (2000) Two-dimensional electrophoresis of liver proteins: characterization of a drug-induced hepatomegaly in rats. Electrophoresis 21:2122–2128
Nijsse J, Walther P, Hoekstra FA (2004) Cold-induced imbibition damage of lettuce embryos: a study using cryo-scanning electron microscopy. Seed Sci Res 14:117–126
Overvoorde PJ, Chao WS, Grimes HD (1997) Plasma membrane sucrose-binding protein that mediates sucrose uptakes structural and sequence similarity with seed storage proteins but remains functionally distinct. J Biol Chem 272:15898–15904
Parrish DJ, Leopold AC (1977) Transient changes during soybean imbibition. Plant Physiol 59:1111–1115
Perkins DN, Pappinl DJC, Creasy DM, Cottrell JS (1999) Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 20:3551–3567
Posmyk MM, Corbineau F, Vinel D, Bailly C, Côme D (2001) Osmoconditioning reduces physiological and biochemical damage induced by chilling in soybean seeds. Physiol Plant 111:473–482
Poysti NJ, Oresnik IJ (2007) Characterization of Sinorhizobium meliloti triose phosphate isomerase genes. J Bacteriol 189:3445–3451
Pradet-Balade B, Boulme F, Beug H, Mullner EW, Garcia-Sanz JA (2001) Translation control: bridging the gap between genomics and proteomics? Trends Biochem Sci 26:225–229
Rattan SI (2006) Theories of biological aging: genes, proteins, and free radicals. Free Bio Res 12:1230–1238
Rohde P, Hincha DK, Heyer AG (2004) Heterosis in the freezing tolerance of crosses between two Arabidopsis thaliana accessions (Columbia-0 and C24) that show differences in non-acclimated and acclimated freezing tolerance. Plant J 38:790–799
Roxas VP, Smith RK, Allen ER, Allen RD (1997) Overexpression of glutathione S-transferase/glutathione peroxidase enhances the growth of transgenic tobacco seedlings during stress. Nat Biotechnol 15:988–991
Roxas VP, Lodhi SA, Garrett DK, Mahan JR, Allen RD (2000) Stress tolerance in transgenic tobacco seedlings that overexpress glutathione S-transferase/glutathione peroxidase. Plant Cell Physiol 41:1229–1234
Savitch LV, Barker-Astrom J, Ivanov AG, Hurry V, Oquist G, Huner NPA (2001) Cold acclimation of Arabidopsis thaliana results in incomplete recovery of photosynthetic capacity, associated with an increased reduction of the chloroplast stroma. Planta 214:295–303
Sawyer TK, Shakespeare WC, Wang Y, Sundaramoorthi R, Huang WS, Metcalf CA III, Thomas M, Lawrence BM, Rozamus L, Noehre J, Zhu X, Narula S, Bohacek RS, Weigele M, Dalgarno DC (2005) Protein phosphorylation and signal transduction modulation: chemistry perspectives for small-molecule drug discovery. Med Chem 1:293–319
Scheibe R (2004) Malate valves to balance cellular energy supply. Physiol Plant 120:21–26
Shiao TL, Ellis MH, Dolferus R, Dennis ES, Doran PM (2002) Overexpression of alcohol dehydrogenase or pyruvate decarboxylase improves growth of hairy roots at reduced oxygen concentrations. Biotechnol Bioeng 77:455–461
Smirnoff N (1993) The role of active oxygen in the response of plants to water deficit and desiccation. New Phytol 125:27–58
Tetlow LJ, Wait R, Lu ZX, Akkasaeng R, Bowsher CG, Esposito S, Kosar-Hashemi B, Morell MK, Emes MJ (2004) Protein phosphorylation in amyloplasts regulates starch branching enzyme activity and protein-protein interactions. Plant Cell 16:694–708
Thomashow MF (2001) So what’s new in the field of plant cold acclimation? Lots!. Plant Physiol 125:89–93
Vertucci CW, Leopold CA (1987) Water binding in legume seeds. Plant Physiol 85:224–231
Walker JE, Saraste M, Gay NJ (1984) The UNC operon nucleotide sequence, regulation and structure of ATP synthase. Biochim Biophys Acta 768:164–200
Watson P, Morris G (1987) Cold shock injury in animal cells. Symp Soc Exp Biol 41:311–340
Wilson KA, Rightmire BR, Chen JC, Tan-Wilson AL (1986) Differential proteolysis of glycinin and β-conglycinin polypeptides during soybean germination and seedling growth. Plant Physiol 82:71–76
Xiao H, Siddiqua M, Braybrook S, Nassuth A (2006) Three grape CBF/DREB1 genes respond to low temperature, drought and abscisic acid. Plant Cell Environ 29:1410–1421
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 57:781–803
Zhang L, Ohta A, Takagi M, Imai R (2000) Expression of plant group 2 and group 3 lea genes in Saccharomyces cerevisiae revealed functional divergence among LEA proteins. J Biochem 127:611–616
Zhang X, Leung SM, Morris CR, Shigenaga MK (2004) Evaluation of a Novel, integrated approach using functionalized magnetic beads, bench-top MALDI-TOF-MS with prestructured sample supports, and pattern recognition software for profiling potential biomarkers in human plasma. J Biomol Tech 15:167–175
Acknowledgments
We are grateful to Dr. Shihua Shen for critical discussion and advice. We also extend our thanks to all the members of Seed Physiology and Biology Laboratory in the Institute of Botany (Chinese Academy of Sciences, Beijing), for providing equipment for spectrometry.
Author information
Authors and Affiliations
Corresponding author
Additional information
Libao Cheng and Xuan Gao have contributed to this work equally.
Rights and permissions
About this article
Cite this article
Cheng, L., Gao, X., Li, S. et al. Proteomic analysis of soybean [Glycine max (L.) Meer.] seeds during imbibition at chilling temperature. Mol Breeding 26, 1–17 (2010). https://doi.org/10.1007/s11032-009-9371-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-009-9371-y