Abstract
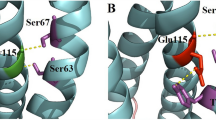
Clinical heterogeneity is commonly observed in Wilson disease (WD), including cases with identical ATP7B mutations. It is thought to be an outcome of impairment in other genes involved in cellular copper homeostasis in addition to the mutations in the ATP7B gene. ATOX1, a copper chaperone that delivers copper to ATP7B, is a potential genetic modifier of WD. In the present study, we analyzed the genetic variations in the ATOX1 gene in 50 WD patients and 60 controls. We identified four novel variants, of which, the coding region variant c.40G > A, p.(Gly14Ser) was observed in 2% alleles. Interestingly, p.(Gly14Ser) was seen with an early onset age, reduced serum ceruloplasmin level and manifestations of liver and brain in a WD patient unlike the other having identical ATP7B mutation but normal ATOX1 alleles. Further, computational analysis predicted that p.(Gly14Ser) substitution, in the critical copper binding motif (MXCXG14C) of the protein, affects the protein–protein interaction involved in copper sharing and transfer between ATOX1 and ATP7B-MBD4. Our findings suggest that p.(Gly14Ser) variant of ATOX1 might play a role as a genetic modifier leading to phenotypic variation in WD.



Similar content being viewed by others
References
Gao J, Brackley S, Mann JP (2018) The global prevalence of Wilson disease from next-generation sequencing data. Genet Med. https://doi.org/10.1038/s41436-018-0309-9
Hahn SH (2014) Population screening for Wilson’s disease. Ann N Y Acad Sci 1315:64–69. https://doi.org/10.1111/nyas.12423
Wu ZY, Wang N, Lin MT, Fang L, Murong SX, Yu L (2001) Mutation analysis and the correlation between genotype and phenotype of Arg778Leu mutation in chinese patients with Wilson disease. Arch Neurol 58:971–976
Okada T, Shiono Y, Hayashi H, Satoh H, Sawada T, Suzuki A et al (2000) Mutational analysis of ATP7B and genotype-phenotype correlation in Japanese with Wilson’s disease. Hum Mutat 15:454–462. https://doi.org/10.1002/(SICI)1098-1004(200005)15:5%3c454:AID-HUMU7%3e3.0.CO;2-J
Chang IJ, Hahn SH (2017) The genetics of Wilson disease. Handb Clin Neurol 142:19–34. https://doi.org/10.1016/B978-0-444-63625-6.00003-3
Medici V, Weiss KH (2017) Genetic and environmental modifiers of Wilson disease. Handb Clin Neurol 142:35–41. https://doi.org/10.1016/B978-0-444-63625-6.00004-5
Roy S, Ganguly K, Pal P, Ghosh S, Das SK, Gangopadhyay PK et al (2018) Influence of apolipoprotein E polymorphism on susceptibility of Wilson disease. Ann Hum Genet 82:53–59. https://doi.org/10.1111/ahg.12223
Stuehler B, Reichert J, Stremmel W, Schaefer M (2004) Analysis of the human homologue of the canine copper toxicosis gene MURR1 in Wilson disease patients. J Mol Med (Berl) 82:629–634. https://doi.org/10.1007/s00109-004-0557-9
Matson Dzebo M, Arioz C, Wittung-Stafshede P (2016) Extended functional repertoire for human copper chaperones. Biomol Concepts 7:29–39. https://doi.org/10.1515/bmc-2015-0030
Hamza I, Faisst A, Prohaska J, Chen J, Gruss P, Gitlin JD (2001) The metallochaperone Atox1 plays a critical role in perinatal copper homeostasis. Proc Natl Acad Sci 98:6848–6852. https://doi.org/10.1073/pnas.111058498
Miyayama T, Suzuki KT, Ogra Y (2009) Copper accumulation and compartmentalization in mouse fibroblast lacking metallothionein and copper chaperone, Atox1. Toxicol Appl Pharmacol 237:205–213. https://doi.org/10.1016/j.taap.2009.03.024
Hatori Y, Inouye S, Akagi R (2017) Thiol-based copper handling by the copper chaperone Atox1. IUMB Life 69:245–254. https://doi.org/10.1002/iub.1620
Banci L, Bertini I, Cantini F, Massaqni C, Migliardi M, Rosato A (2009) An NMR study of the interaction of the N-terminal cytoplasmic tail of the Wilson disease protein with copper (I)-HAH1. J Biol Chem 284:9354–9360. https://doi.org/10.1074/jbc.M805981200
Rodriguez-Granillo A, Crespo A, Estrin DA, Wittung-Stafshede P (2010) Copper-transfer mechanism from the human chaperone Atox1 to a metal-binding domain of Wilson disease protein. J Phys Chem B 114:3698–3706. https://doi.org/10.1021/jp911208z
Walker JM, Tsivkovskii R, Lutsenko S (2002) Metallochaperone Atox1 transfers copper to the NH2-terminal domain of Wilson’s disease protein and regulates its catalytic activity. J Biol Chem 277:27953–27959. https://doi.org/10.1074/jbc.M203845200
Hamza I, Schaefer M, Klomp LW, Giltin D (1999) Interaction of the copper chaperone HAH1 with the Wilson disease protein is essential for copper homeostasis. Proc Natl Acad Sci 96:13363–13368
Kumari N, Kumar A, Thapa BR, Modi M, Pal A, Prasad R (2018) Characterization of mutation spectrum and identification of novel mutations in ATP7B gene from a cohort of Wilson disease patients: functional and therapeutic implications. Hum Mutat. https://doi.org/10.1002/humu.23614
Daly AK, Steen VM, Fairbrother KS, Idle JR (1996) CYP2D6 multiallelism. Methods Enzymol 272:199–210
Simon I, Schaefer M, Reichert J, Stremmel W (2008) Analysis of the human Atox 1 homologue in Wilson patients. World J Gastroenterol 14:2383–2387
Anastassopoulou I, Banci L, Bertini I, Cantini F, Katsari E, Rosato A (2004) Solution structure of the apo and copper(I)-loaded human metallochaperone HAH1. Biochemistry 43:13046–13053. https://doi.org/10.1021/bi0487591
Wernimont AK, Huffman DL, Lamb AL, O’Halloran TV, Rosenzweig AC (2000) Structural basis for copper transfer by the metallochaperone for the Menkes/Wilson disease proteins. Nat Struct Mol Biol 7:766–771. https://doi.org/10.1038/78999
Banci L, Bertini I, Cantini F, Rosenzweig AC, Yatsunyk LA (2008) Metal binding domains 3 and 4 of the Wilson disease protein: solution structure and interaction with the copper(I) chaperone HAH1. Biochemistry 47:7423–7429. https://doi.org/10.1021/bi8004736
Biasini M, Bienert S, Waterhouse A, Arnold K, Studer G, Schmidt T, Kiefer F, Gallo Cassarino T, Bertoni M, Bordoli L, Schwede T (2014) SWISS-MODEL: modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res 42:W252–W258. https://doi.org/10.1093/nar/gku340
Agarwal V, Bell GW, Nam J, Bartel DP (2015) Predicting effective microRNA target sites in mammalian mRNAs. eLife 4:e05005. https://doi.org/10.7554/elife.05005
Bost M, Piguet-Lacroix G, Parant F, Wilson CM (2012) Molecular analysis of Wilson patients: direct sequencing and MLPA analysis in the ATP7B gene and Atox1 and COMMD1 gene analysis. J Trace Elem Med Biol 26:97–101. https://doi.org/10.1016/j.jtemb.2012.04.024
Skeeles LE, Fleming JL, Kimberly L, Toland AE (2013) The impact of 3′ UTR variants on differential expression of candidate cancer susceptibility genes. PLoS ONE 8:e58609. https://doi.org/10.1371/journal.pone.0058609
Hatori Y, Lutsenko S (2013) An expanding range of functions for the copper chaperone/antioxidant protein Atox1. Antioxid Redox Signal 19:945–957. https://doi.org/10.1089/ars.2012.5086
Acknowledgements
We thank the patients and families for their kind participation and interest in this study. The work was supported by grants from the Indian Council of Medical Research, New Delhi, India (Grant Nos. 54/1/2014-BMS and 3/1/3/JRF-2012/HRD-53).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumari, N., Kumar, A., Pal, A. et al. In-silico analysis of novel p.(Gly14Ser) variant of ATOX1 gene: plausible role in modulating ATOX1–ATP7B interaction. Mol Biol Rep 46, 3307–3313 (2019). https://doi.org/10.1007/s11033-019-04791-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-04791-x