Abstract
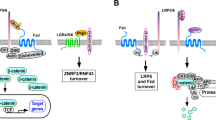
The canonical Wnt pathway contributes to the regulation of neuronal survival and homeostasis in the CNS. Recent evidence suggests that an increased expression of Dickkopf-1 (Dkk-1), a secreted protein that negatively modulates the canonical Wnt pathway, is causally related to processes of neurodegeneration in a number of CNS disorders, including Alzheimer’s disease (AD), brain ischemia and temporal lobe epilepsy (TLE). Dkk-1 induction precedes neuronal death in cellular and animal models of excitotoxicity, β-amyloid toxicity, transient global ischemia, and kainate-induced epilepsy. In addition, Dkk-1, which is barely visible in the healthy brain, is strongly induced in brain tissue from AD patients or from patients with TLE associated with hippocampal sclerosis. These data raise the attractive possibility that Dkk-1 antagonists or neutralizing antibodies behave as neuroprotective agents by rescuing the activity of the canonical Wnt pathway.



Similar content being viewed by others
References
Dale TC (1998) Signal transduction by the Wnt family of ligands. Biochem J 329:209–223
He X, Semenov M, Tamai K et al (2004) LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: arrows point the way. Development 131:1663–1677
Miller JR, Hocking AM, Brown JD et al (1999) Mechanism and function of signal transduction by the Wnt/beta-catenin and Wnt/Ca2+ pathways. Oncogene 18:7860–7872
Prunier C, Hocevar BA, Howe PH (2004) Wnt signaling: physiology and pathology. Growth Factors 22:141–150
Miller JR (2002) The Wnts. Genome Biol 3(1):3001.1–3001.15
Moon RT, Bowerman B, Boutros M et al (2002) The promise and perils of Wnt signaling through β-catenin. Science 296:1644–1646
Mlodzik M (2002) Planar cell polarization: do the same mechanisms regulate Drosophila tissue polarity and vertebrate gastrulation? Trends Genet 18:564–571
Kühl M, Sheldahl LC, Park M et al (2000) The Wnt/Ca2+pathway: a new vertebrate Wnt signaling pathway takes shape. Trends Genet 16:279–283
Tamai K, Semenov M, Kato Y et al (2000) LDL-receptor-related proteins in Wnt signal transduction. Nature 407:530–535
Wehrli M, Dougan ST, Caldwell K et al (2000) Arrow encodes an LDL-receptor-related protein essential for Wingless signalling. Nature 407:527–530
Liu C, Li Y, Semenov M et al (2002) Control of β-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell 108:837–847
Aberle H, Bauer A, Stappert J et al (1997) Beta-catenin is a target for the ubiquitin–proteasome pathway. EMBO J 16:3797–3804
Willert K, Nusse R (1998) Beta-catenin: a key mediator of Wnt signaling. Curr Opin Genet Dev 8:95–102
Willert K, Jones KA (2006) Wnt signaling: is the party in the nucleus? Genes Dev 20:1394–1404
Salinas PC (1999) Wnt factors in axonal remodelling and synaptogenesis. Biochem Soc Symp 65:101–109
Yu X, Malenka RC (2003) Beta-catenin is critical for dendritic morphogenesis. Nat Neurosci 6:1169–1177
Murase S, Mosser E, Schuman EM (2002) Depolarization drives beta-catenin into neuronal spines promoting changes in synaptic structure and function. Neuron 35:91–105
Niehrs C (2006) Function and biological roles of the Dickkopf family of Wnt modulators. Oncogene 25:7469–7481
Krupnik VE, Sharp JD, Jiang C et al (1999) Functional and structural diversity of the human Dickkopf gene family. Gene 238:301–313
Brott BK, Sokol SY (2002) Regulation of Wnt/LRP signalling by distinct domains of Dickkopf proteins. Mol Cell Biol 22:6100–6110
Diep DB, Hoen N, Backman M et al (2004) Characterisation of the Wnt antagonists and their response to conditionally activated Wnt signalling in the developing mouse forebrain. Brain Res Dev Brain Res 153:261–270
Grotewold L, Ruther U (2002) The Wnt antagonist Dickkopf-1 is 675 regulated by Bmp signaling and c-Jun and modulates programmed cell death. EMBO J 21:966–975
Glinka A, Wu W, Delius H et al (1998) Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 391:357–362
Zorn AM (2001) Wnt signalling: antagonistic Dickkopfs. Curr Biol 11:R592–R595
Mao B, Niehrs C (2003) Kremen2 modulates Dickkopf2 activity during Wnt/LRP6 signaling. Gene 302:179–183
Wang J, Shou J, Chen X (2000) Dickkopf-1, an inhibitor of the Wnt signaling pathway, is induced by p53. Oncogene 19:1843–1848
Slee EA, O’Connor DJ, Lu X (2004) To die or not to die: how does p53 decide? Oncogene 23:2809–2818
Choi DW (1994) Calcium and excitotoxic neuronal injury. Ann N Y Acad Sci 747:162–171
Zipfel GJ, Babcock DJ, Lee JM et al (2000) Neuronal apoptosis after CNS injury: the roles of glutamate and calcium. J Neurotrauma 17:857–869
Bruno V, Battaglia G, Copani A et al (2001) Metabotropic glutamate receptor subtypes as targets for neuroprotective drugs. J Cereb Blood Flow Metab 21:1013–1033
Cappuccio I, Calderone A, Busceti CL et al (2005) Induction of Dickkopf-1, a negative modulator of the Wnt pathway, is required for the development of ischemic neuronal death. J Neurosci 25:2647–2657
Jope RS (2003) Lithium and GSK-3: one inhibitor, two inhibitory actions, multiple outcomes. Trends Pharmacol Sci 24:441–443
Scali C, Caraci F, Gianfriddo M et al (2006) Inhibition of Wnt signaling, modulation of Tau phosphorylation and induction of neuronal cell death by DKK1. Neurobiol Dis 24:254–265
Oddo S, Caccamo A, Kitazawa M et al (2003) Amyloid deposition precedes tangle formation in a triple transgenic model of Alzheimer’s disease. Neurobiol Aging 24:1063–1070
Blurton-Jones M, Laferla FM (2006) Pathways by which Abeta facilitates tau pathology. Curr Alzheimer Res 3:437–448
Takashima A, Honda T, Yasutake K et al (1998) Activation of tau protein kinase I/glycogen synthase kinase-3beta by amyloid beta peptide (25–35) enhances phosphorylation of tau in hippocampal neurons. J Neurosci Res 31:317–323
Takashima A (2006) GSK-3 is essential in the pathogenesis of Alzheimer’s disease. J Alzheimers Dis 9:309–317
Zhou J, Liyanage U, Medina M et al (1997) Presenilin 1 interaction in the brain with a novel member of the Armadillo family. NeuroReport 8:1489–1494
Takashima A, Murayama M, Murayama O et al (1998) Presenilin 1 associates with glycogen synthase kinase-3beta and its substrate tau. Proc Natl Acad Sci USA 95:9637–9641
Kang DE, Soriano S, Xia X et al (2002) Presenilin couples the paired phosphorylation of beta-catenin independent of axin: implications for beta-catenin activation in tumorigenesis. Cell 110:751–762
Zhang Z, Hartmann H, Do VM et al (1998) Destabilization of beta-catenin by mutations in presenilin-1 potentiates neuronal apoptosis. Nature 395:698–702
De Ferrari GV, Moon RT (2006) The ups and downs of Wnt signaling in prevalent neurological disorders. Oncogene 25:7545–7553
De Ferrari GV, Inestrosa NC (2000) Wnt signaling function in Alzheimer’s disease. Brain Res Brain Res Rev 33:1–12
De Ferrari GV, Chacón MA, Barría MI et al (2003) Activation of Wnt signaling rescues neurodegeneration and behavioral impairments induced by beta-amyloid fibrils. Mol Psychiatry 8:195–208
Copani A, Condorelli F, Caruso A et al (1999) Mitotic signaling by beta-amyloid causes neuronal death. FASEB J 13:2225–2234
Copani A, Caraci F, Hoozemans JJ et al (2007) The nature of the cell cycle in neurons: focus on a “non-canonical” pathway of DNA replication causally related to death. Biochim Biophys Acta 1772:409–412
Caricasole A, Copani A, Caraci F et al (2004) Induction of Dickkopf-1, a negative modulator of the Wnt pathway, is associated with neuronal degeneration in Alzheimer’s brain. J Neurosci 24:6021–6027
De Ferrari GV, Papassotiropoulos A, Biechele T et al (2007) Common genetic variation within the low-density lipoprotein receptor-related protein 6 and late-onset Alzheimer’s disease. Proc Natl Acad Sci USA 104:9434–9439
Biagioni F, Mastroiacovo F, Busceti CL et al (2007) Induction of transient focal brain ischemia induces the expression of the Wnt inhibitor, Dickkopf-1, in neurons of the perifocal regions in rats. Soc Neurosci Abstr Program No. 493.22
Honavar M, Meldrum BS (1997) Epilepsy. In: Graham DI, Lantos PI (eds) Greenfield’s neuropathology, 6th edn. Arnold, London, pp 931–971
Busceti CL, Biagioni F, Aronica E et al (2007) Induction of the Wnt inhibitor, Dickkopf-1, is associated with neurodegeneration related to temporal lobe epilepsy. Epilepsia 48:694–705
Busceti CL, Biagioni F, Riozzi et al (2007) 3,4-Methylendioxymethamphetamine (ecstasy) induces the expression of the wnt inhibitor, dickkopf-1, and tau protein phosphorylation in the mouse hippocampus. Soc Neurosci Abstr Program No. 66.4
Zakzanis KK, Campbell Z (2006) Memory impairment in now abstinent MDMA users and continued users: a longitudinal follow-up. Neurology 66:740–741
Wu D, Zhang Y, Liu P et al (2005) Compositions and methods for bone formation and remodeling. US Patent 2005/0196349
Desbois-Mouthon C, Cadoret A, Blivet-Van Eggelpoël MJ et al (2001) Insulin and IGF-1 stimulate the beta-catenin pathway through two signalling cascades involving GSK-3beta inhibition and Ras activation. Oncogene 20:252–259
Caraci F, Copani A, Battaglia G et al (2006) A dual mechanism of neuroprotection by transforming growth factor-β1 against β-amyloid neurotoxicity. Soc Neurosci Abstr Program No. 171.4
Torres Aleman I (2005) Role of insulin-like growth factors in neuronal plasticity and neuroprotection. Adv Exp Med Biol 567:243–258
Vivien D, Ali C (2006) Transforming growth factor-beta signalling in brain disorders. Cytokine Growth Factor Rev 17:121–128
Huang HC, Klein PS (2006) Multiple roles for glycogen synthase kinase-3 as a drug target in Alzheimer’s disease. Curr Drug Targets 7:1389–1397
Wada A, Yokoo H, Yanagita T et al (2005) Lithium: potential therapeutics against acute brain injuries and chronic neurodegenerative diseases. J Pharmacol Sci 99:307–321
Nunes PV, Forlenza OV, Gattaz WF (2007) Lithium and risk for Alzheimer’s disease in elderly patients with bipolar disorder. Br J Psychiatry 190:359–360
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Anna Maria Giuffrida-Stella.
Agata Copani and Ferdinando Nicoletti—Co-senior authors.
Filippo Caraci—PhD Program in Neuropharmacology.
Rights and permissions
About this article
Cite this article
Caraci, F., Busceti, C., Biagioni, F. et al. The Wnt Antagonist, Dickkopf-1, as a Target for the Treatment of Neurodegenerative Disorders. Neurochem Res 33, 2401–2406 (2008). https://doi.org/10.1007/s11064-008-9710-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9710-0