Abstract
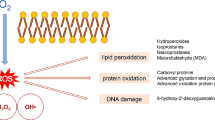
Normobaric hyperoxia (NBO) is applied for treatment of various clinical conditions related to hypoxia, but it can potentially also induce generation of reactive oxygen species, causing cellular damage. In this study, we examined the effects of 60 h NBO treatment on lipid and protein oxidative damage and activity of superoxide dismutase (Mn-SOD) in brain mitochondria of guinea pigs. Despite significant stimulation of Mn-SOD expression and activity the NBO treatment resulted in accumulation of markers of oxidative lesions, including lipid peroxidation (conjugated dienes, thiobarbituric acid reactive substances) and protein modification (bityrosines, adducts with lipid peroxidation products, oxidized thiols). When inhaled O2 was enriched with oxygen cation, O •+2 , the Mn-SOD expression and activity were stimulated to similar extend, but lipid peroxidation and protein oxidation were prevented. These results suggest that long-term NBO treatment causes oxidative stress, but enrichment of inhaled oxygen by oxygen cation can protect the brain again adverse effects of hyperoxia.




Similar content being viewed by others
References
Singhal AB (2007) A review of oxygen therapy in ischemic stroke. Neurol Res 29:173–183
Kumaria A, Tolias CM (2009) Normobaric hyperoxia therapy for traumatic brain injury and stroke: a review. Br J Neurosurg 26:576–584
Chen Y, Nadi NS, Chavko M et al (2009) Microarray analysis of gene expression in rat cortical neurons exposed to hyperbaric air and oxygen. Neurochem Res 34:1047–1056
Matchett GA, Martin RD, Zhang JH (2009) Hyperbaric oxygen therapy and cerebral ischemia: neuroprotective mechanisms. Neurol Res 31:114–121
Brutus NA, Hanley S, Ashraf QM et al (2009) Effect of hyperoxia on serine phosphorylation of apoptotic proteins in mitochondrial membranes of the cerebral cortex of newborn piglets. Neurochem Res 34:1219–1225
Mudduluru M, Zubrow AB, Ashraf QM et al (2010) Tyrosine phosphorylation of apoptotic proteins during hyperoxia in mitochondria of the cerebral cortex of newborn piglets Neurochem Res 35:1003–1009
Thom SR (2009) Oxidative stress is fundamental to hyperbaric oxygen therapy. J Appl Physiol 106:988–995
Henseley K, Howard BJ, Carney JM et al (1995) Membrane protein alterations in rodent erythrocytes and synaptosomes due to aging and hyperoxia. Biochim Biophys Acta 1270:203–206
Dundar K, Topal T, Ay H et al (2005) Protective effects of exogenously administrated or endogenously produced melatonin on hyperbaric oxygen-induced oxidative stress in the rat brain. Clin Exp Pharmacol Physiol 32:926–930
Bader N, Bosy-Westphal A, Koch A et al (2007) Effect of hyperbaric oxygen and vitamin C and E supplementation on biomarkers of oxidative stress in healthy men. Br J Nutr 98:826–833
Brozmanova M, Plevkova J, Bartos V et al (2008) The interaction of dietary antioxidant vitamins and oxidative stress on cough reflex in guinea-pigs after long term oxygen therapy J Physiol Pharmacol 57:45–54
Calderón Guzmán D, Trujillo Jiménez F, Hernández García E et al (2007) Assessment of antioxidant effect of 2, 5-dihydroxybenzoic acid and vitamin A in brains of rats with induced hyperoxia. Neurochem Res 32:1036–1040
Mollaoglu H, Topal T, Ozler M et al (2007) Antioxidant effects of melatonin in rats during chronic exposure to hyperbaric oxygen. J Pineal Res 42:50–54
Calkovska A, Engler I, Mokra D et al (2008) Differences in oxidative status, lung function, and pulmonary surfactant during long-term inhalation of medical oxygen and partially ionized oxygen in guinea pigs. J Physiol Pharmacol 59:173–181
Kaplan P, Tatarkova Z, Engler I et al (2009) Effects of long-term oxygenation treatments on α-ketoglutarate dehydrogenase activity and oxidative modifications in mitochondria of guinea pig heart. Eur J Med Res 14:116–120
Engler I, Atzmüeller C, Donic V et al (2009) Reactive oxygen species, especially O +•2 in cancer mechanisms. J Exp Therapeut Oncol 8(2):157–165
Babusikova E, Hatok J, Dobrota D et al (2007) Age-related oxidative modifications of proteins and lipids in rat brain. Neurochem Res 32:1351–1356
Dodd PR, Hardy JA, Oakley AE (1981) A rapid method for preparing synaptosomes: comparison with alternative procedures. Brain Res 226:107–118
Kaplan P, Babusikova E, Lehotsky J et al (2003) Free radical-induced protein modification and inhibition of Ca2+-ATPase of cardiac sarcoplasmic reticulum. Mol Cell Biochem 248:41–47
Gonzales R, Auclair C, Voisin E et al (1984) Superoxide dismutase, catalase, and glutathione peroxidase in red blood cells from patients with malignant diseases. Cancer Res 44:4137–4139
Powell CS, Jackson RM (2003) Mitochondrial complex I, aconitase, and succinate dehydrogenase during hypoxia-reoxygenation: Modulation of enzyme activities by MnSOD. Am J Physiol Lung Cell Mol Physiol 285:L189–L198
Hink J, Jansen E (2001) Are superoxide and/or hydrogen peroxide responsible for some of the beneficial effects of hyperbaric oxygen therapy? Med Hypotheses 57:764–769
Ay H, Topal T, Özler M et al (2007) Persistence of hyperbaric oxygen-induced oxidative effects after exposure in rat brain cortex tissue. Life Sci 80:2025–2029
Nemoto EM, Betterman K (2007) Basic physiology of hyperbaric oxygen in brain. Neurol Res 29:116–126
Xue L, Yu Q, Zhang H et al (2008) Effect of large dose hyperbaric oxagenation therapy on prognosis and oxidative stress of acute pemanet cerebral ischemic stroke in rats. Neurol Res 30:389–393
Singhal AB, Wang X, Sumii T et al (2002) Effect of normobaric hyperoxia in a rat moedl of focal cerebral ischemia-reperfusion. J Cereb Blood Flow Metab 22:861–868
Puccio AM, Hoffman LA, Bayir H et al (2009) Effect of short periods of normobaric hyperoxia on local brain tissue oxygenation and cerebrospinal fluid oxidative stress markers in severe traumatic brain injury. J Neurotrauma 26:1241–1249
Liu W, Sood R, Chen Q et al (2008) Normobaric hyperoxia inhibits NADPH oxidase-mediated matrix metalloproteinase-9 induction in cerebral microvessels in experimental stroke. J Neurochem 107:1196–1205
Ahotupa M, Mantyla E, Peltola V et al (1992) Pro-oxidant effects of normobaric hyperoxia in rat tissues. Acta Physiol Scand 145:151–157
Bigdeli MR, Rahnema M (2009) The effect of normobaric hyperoxia on superoxide dismutase activity and neurological deficits in an animal model of Huntington disease. Physiol Pharmacol 13:18–27
Boveris A, Chance B (1973) The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J 134:707–716
McLennan HR, Esposti MD (2000) The contribution of mitochondrial respiratory complexes to the production of reactive oxygen species. J. Bioenerg Biomembr 32:153–162
Lenaz G (2002) The mitochondrial production of reactive oxygen species: Mechanisms and implications in human pathology. IUBMB Life 52:159–164
Tang X, Liu KJ, Ramu J et al (2010) Inhibition of gp91phox contributes towards normobaric hyperoxia afforded neuroprotection in focal cerebral ischemia. Brain Res 1348:174–180
Acknowledgments
This work was partially supported by grants VEGA 1/0028/11 and VVCE APVV 0064/07 from the Ministry of Education and Science of the Slovak Republic and project “Identification of Novel Markers in Diagnostic Panel of Neurological Diseases”, code:26220220114 co-financed from EC sources and European Regional Development Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tatarkova, Z., Engler, I., Calkovska, A. et al. Effect of Long-Term Normobaric Hyperoxia on Oxidative Stress in Mitochondria of the Guinea Pig Brain. Neurochem Res 36, 1475–1481 (2011). https://doi.org/10.1007/s11064-011-0473-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0473-7