Abstract
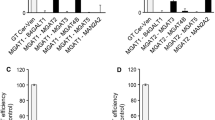
The functional link between glycolipid glycosyltransferases (GT) relies on the ability of these proteins to form organized molecular complexes. The organization, stoichiometry and composition of these complexes may impact their sorting properties, sub-Golgi localization, and may determine relative efficiency of GT in different glycolipid biosynthetic pathways. In this work, by using Förster resonance energy transfer microscopy in live CHO-K1 cells, we investigated homo- and hetero-complex formation by different GT as well as their spatial organization and molecular stoichiometry on Golgi membranes. We find that GalNAcT and GalT2 Ntd are able to form hetero-complexes in a 1:2 molar ratio at the trans-Golgi network and that GalT2 but not GalNAcT forms homo-complexes. Also, GalNAcT/GalT2 complexes exhibit a stable behavior reflected by its clustered lateral organization. These results reveals that particular topological organization of GTs may have functional implications in determining the composition of glycolipids in cellular membranes.




Similar content being viewed by others
References
Yu RK, Bieberich E, Xia T, Zeng G (2004) Regulation of ganglioside biosynthesis in the nervous system. J Lipid Res 45(5):783–793
Maxzud MK, Daniotti JL, Maccioni HJ (1995) Functional coupling of glycosyl transfer steps for synthesis of gangliosides in Golgi membranes from neural retina cells. J Biol Chem 270(34):20207–20214
Maccioni HJ (2007) Glycosylation of glycolipids in the Golgi complex. J Neurochem 103(Suppl 1):81–90
de Graffenried CL, Bertozzi CR (2004) The roles of enzyme localisation and complex formation in glycan assembly within the Golgi apparatus. Curr Opin Cell Biol 16(4):356–363
Young WW Jr (2004) Organization of Golgi glycosyltransferases in membranes: complexity via complexes. J Membr Biol 198(1):1–13
Giraudo CG, Daniotti JL, Maccioni HJ (2001) Physical and functional association of glycolipid N-acetyl-galactosaminyl and galactosyl transferases in the Golgi apparatus. Proc Natl Acad Sci USA 98(4):1625–1630
Giraudo CG, Maccioni HJ (2003) Ganglioside glycosyltransferases organize in distinct multienzyme complexes in CHO-K1 cells. J Biol Chem 278(41):40262–40271
Hassinen A, Pujol FM, Kokkonen N, Pieters C, Kihlstrom M, Korhonen K, Kellokumpu S (2011) Functional organization of Golgi N- and O-glycosylation pathways involves pH-dependent complex formation that is impaired in cancer cells. J Biol Chem 286(44):38329–38340
Kremers GJ, Goedhart J, van Munster EB, Gadella TW Jr (2006) Cyan and yellow super fluorescent proteins with improved brightness, protein folding, and FRET Forster radius. Biochemistry 45(21):6570–6580
Elangovan M, Wallrabe H, Chen Y, Day RN, Barroso M, Periasamy A (2003) Characterization of one- and two-photon excitation fluorescence resonance energy transfer microscopy. Methods 29(1):58–73
Trenchi A, Gomez GA, Daniotti JL (2009) Dual acylation is required for trafficking of growth-associated protein-43 (GAP-43) to endosomal recycling compartment via an Arf6-associated endocytic vesicular pathway. Biochem J 421(3):357–369
Jares-Erijman EA, Jovin TM (2003) FRET imaging. Nat Biotechnol 21(11):1387–1395
Lippincott-Schwartz J, Snapp E, Kenworthy A (2001) Studying protein dynamics in living cells. Nat Rev Mol Cell Biol 2(6):444–456
Wallrabe H, Periasamy A (2005) Imaging protein molecules using FRET and FLIM microscopy. Curr Opin Biotechnol 16(1):19–27
Hoppe A, Christensen K, Swanson JA (2002) Fluorescence resonance energy transfer-based stoichiometry in living cells. Biophys J 83(6):3652–3664
Uliana AS, Giraudo CG, Maccioni HJ (2006) Cytoplasmic tails of SialT2 and GalNAcT impose their respective proximal and distal Golgi localization. Traffic 7(5):604–612
Kenworthy AK, Edidin M (1998) Distribution of a glycosylphosphatidylinositol-anchored protein at the apical surface of MDCK cells examined at a resolution of <100 Å using imaging fluorescence resonance energy transfer. J Cell Biol 142(1):69–84
Wallrabe H, Elangovan M, Burchard A, Periasamy A, Barroso M (2003) Confocal FRET microscopy to measure clustering of ligand-receptor complexes in endocytic membranes. Biophys J 85(1):559–571
Gordon GW, Berry G, Liang XH, Levine B, Herman B (1998) Quantitative fluorescence resonance energy transfer measurements using fluorescence microscopy. Biophys J 74(5):2702–2713
Förster T (1948) Zwischenmolekulare Energiewanderung und Fluoreszenz. Ann Physik 6(2):55–75
Giraudo CG, Rosales Fritz VM, Maccioni HJ (1999) GA2/GM2/GD2 synthase localizes to the trans-golgi network of CHO-K1 cells. Biochem J 342(Pt 3):633–640
Panzetta P, Maccioni HJ, Caputto R (1980) Synthesis of retinal gangliosides during chick embryonic development. J Neurochem 35(1):100–108
Yu RK, Macala LJ, Taki T, Weinfield HM, Yu FS (1988) Developmental changes in ganglioside composition and synthesis in embryonic rat brain. J Neurochem 50(6):1825–1829
Maccioni HJ, Quiroga R, Ferrari ML (2011) Cellular and molecular biology of glycosphingolipid glycosylation. J Neurochem 117(4):589–602
Galperin E, Verkhusha VV, Sorkin A (2004) Three-chromophore FRET microscopy to analyze multiprotein interactions in living cells. Nat Methods 1(3):209–217
Pohlentz G, Klein D, Schwarzmann G, Schmitz D, Sandhoff K (1988) Both GA2, GM2, and GD2 synthases and GM1b, GD1a, and GT1b synthases are single enzymes in Golgi vesicles from rat liver. Proc Natl Acad Sci USA 85(19):7044–7048
Martina JA, Daniotti JL, Maccioni HJ (2000) GM1 synthase depends on N-glycosylation for enzyme activity and trafficking to the Golgi complex. Neurochem Res 25(5):725–731
Li J, Yen TY, Allende ML, Joshi RK, Cai J, Pierce WM, Jaskiewicz E, Darling DS, Macher BA, Young WW Jr (2000) Disulfide bonds of GM2 synthase homodimers. Antiparallel orientation of the catalytic domains. J Biol Chem 275(52):41476–41486
Bieberich E, MacKinnon S, Silva J, Li DD, Tencomnao T, Irwin L, Kapitonov D, Yu RK (2002) Regulation of ganglioside biosynthesis by enzyme complex formation of glycosyltransferases. Biochemistry 41(38):11479–11487
Hassinen A, Rivinoja A, Kauppila A, Kellokumpu S (2010) Golgi N-glycosyltransferases form both homo- and heterodimeric enzyme complexes in live cells. J Biol Chem 285(23):17771–17777
Acknowledgments
This work was supported in part by Grants from Agencia Nacional de Promoción Científica y Tecnológica (PICT·2006-01239), Universidad Nacional de Córdoba, y MinCyT Córdoba. We thank the technical assistance of G. Schachner and S. Deza with cell cultures and of C. Mas and M. C. Sampedro with confocal microscopy. M.L.F. and G.A.G. were recipients of CONICET Fellowships and H.J.F.M. is Career Investigator of CONICET (Argentina).
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: In Honor of Bob Ledeen
This work is dedicated to Bob Ledeen, a pioneer in the study of the biochemistry and neurochemistry of glycolipids, great friend and enthusiastic and clever investigator.
Rights and permissions
About this article
Cite this article
Ferrari, M.L., Gomez, G.A. & Maccioni, H.J.F. Spatial Organization and Stoichiometry of N-Terminal Domain-Mediated Glycosyltransferase Complexes in Golgi Membranes Determined by Fret Microscopy. Neurochem Res 37, 1325–1334 (2012). https://doi.org/10.1007/s11064-012-0741-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-012-0741-1