Abstract
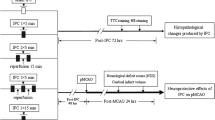
Whether ischemic postconditioning (IPC) can significantly alleviate ischemic injury hinges on the appropriate measure. In this study, the expression RGMa and IL-1β, IL-6 are investigated to estimate the therapeutic benefits of various postconditioning strategies after cerebral ischemia/reperfusion. The study consists of the sham-operated group and five treatment groups: ischemia/reperfusion (I/R), two proximate ischemic postconditioning (IPC-S and IPC-M), remote postconditioning (RIPC) and delayed postconditioning (DIPC) groups. We find that rats in IPC and RIPC groups exhibit significantly less neural deficit and lower infarct volume than that in I/R and DIPC groups after ischemia/reperfusion. Moreover, in ischemic cortex and hippocampus, the mRNA level of RGMa is much lower in IPC and RIPC groups. Immunohistochemical analysis indicates that the expression of RGMa, IL-1β and IL-6 are reduced in IPC and RIPC groups (especially in IPC-S group). Furthermore, neurofilament staining reveals that the rats in IPC and RIPC groups have less axonal injury than that in I/R and DIPC groups. Our studies suggest that the optimal strategy to attenuate cerebral ischemia/reperfusion is achieved by early, short-term, and multiple cycles of proximal IPC. The cerebral protective effect of IPC may be associated with the decreased expression of RGMa and inflammation mediators.






Similar content being viewed by others
References
Donnan GA, Fisher M, Macleod M, Davis SM (2008) Stroke. Lancet 371(9624):1612–1623. doi:10.1016/S0140-6736(08)60694-7
Chen H, Song YS, Chan PH (2009) Inhibition of NADPH oxidase is neuroprotective after ischemia-reperfusion. J Cereb Blood Flow Metab 29(7):1262–1272. doi:10.1038/jcbfm.2009.47
Fang J, Chen L, Fan L, Wu L, Chen X, Li W, Lin Y, Wang W (2011) Enhanced therapeutic effects of mesenchymal stem cells on myocardial infarction by ischemic postconditioning through paracrine mechanisms in rats. J Mol Cell Cardiol 51(5):839–847. doi:10.1016/j.yjmcc.2011.06.013
Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, Vinten-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285(2):H579–H588. doi:10.1152/ajpheart.01064.2002
Vinten-Johansen J, Yellon DM, Opie LH (2005) Postconditioning: a simple, clinically applicable procedure to improve revascularization in acute myocardial infarction. Circulation 112(14):2085–2088. doi:10.1161/CIRCULATIONAHA.105.569798
Zhao H, Sapolsky RM, Steinberg GK (2006) Interrupting reperfusion as a stroke therapy: ischemic postconditioning reduces infarct size after focal ischemia in rats. J Cereb Blood Flow Metab 26(9):1114–1121. doi:10.1038/sj.jcbfm.9600348
Gao X, Ren C, Zhao H (2008) Protective effects of ischemic postconditioning compared with gradual reperfusion or preconditioning. J Neurosci Res 86(11):2505–2511. doi:10.1002/jnr.21703
Zhou C, Tu J, Zhang Q, Lu D, Zhu Y, Zhang W, Yang F, Brann DW, Wang R (2011) Delayed ischemic postconditioning protects hippocampal CA1 neurons by preserving mitochondrial integrity via Akt/GSK3beta signaling. Neurochem Int 59(6):749–758. doi:10.1016/j.neuint.2011.08.008
Zhang W, Miao Y, Zhou S, Jiang J, Luo Q, Qiu Y (2011) Neuroprotective effects of ischemic postconditioning on global brain ischemia in rats through upregulation of hippocampal glutamine synthetase. J Clin Neurosci 18(5):685–689. doi:10.1016/j.jocn.2010.08.027
Penna C, Tullio F, Merlino A, Moro F, Raimondo S, Rastaldo R, Perrelli MG, Mancardi D, Pagliaro P (2009) Postconditioning cardioprotection against infarct size and post-ischemic systolic dysfunction is influenced by gender. Basic Res Cardiol 104(4):390–402. doi:10.1007/s00395-008-0762-8
Ren C, Gao X, Niu G, Yan Z, Chen X, Zhao H (2008) Delayed postconditioning protects against focal ischemic brain injury in rats. PLoS ONE 3(12):e3851. doi:10.1371/journal.pone.0003851
Zhao H (2009) Ischemic postconditioning as a novel avenue to protect against brain injury after stroke. J Cereb Blood Flow Metab 29(5):873–885. doi:10.1038/jcbfm.2009.13
Zhou Y, Fathali N, Lekic T, Ostrowski RP, Chen C, Martin RD, Tang J, Zhang JH (2011) Remote limb ischemic postconditioning protects against neonatal hypoxic-ischemic brain injury in rat pups by the opioid receptor/Akt pathway. Stroke 42(2):439–444. doi:10.1161/STROKEAHA.110.592162
Pignataro G, Meller R, Inoue K, Ordonez AN, Ashley MD, Xiong Z, Gala R, Simon RP (2008) In vivo and in vitro characterization of a novel neuroprotective strategy for stroke: ischemic postconditioning. J Cereb Blood Flow Metab 28(2):232–241. doi:10.1038/sj.jcbfm.9600559
Endo M, Yamashita T (2009) Inactivation of Ras by p120GAP via focal adhesion kinase dephosphorylation mediates RGMa-induced growth cone collapse. J Neurosci 29(20):6649–6662. doi:10.1523/JNEUROSCI.0927-09.2009
Schnichels S, Heiduschka P, Julien S (2011) Different spatial and temporal protein expressions of repulsive guidance molecule a and neogenin in the rat optic nerve after optic nerve crush with and without lens injury. J Neurosci Res 89(4):490–505. doi:10.1002/jnr.22584
Kitayama M, Ueno M, Itakura T, Yamashita T (2011) Activated microglia inhibit axonal growth through RGMa. PLoS ONE 6(9):e25234. doi:10.1371/journal.pone.0025234
Satoh J, Tabunoki H, Ishida T, Saito Y, Arima K (2012) Accumulation of a repulsive axonal guidance molecule RGMa in amyloid plaques: a possible hallmark of regenerative failure in Alzheimer’s disease brains. Neuropathol Appl Neurobiol. doi:10.1111/j.1365-2990.2012.01281.x
Hata K, Fujitani M, Yasuda Y, Doya H, Saito T, Yamagishi S, Mueller BK, Yamashita T (2006) RGMa inhibition promotes axonal growth and recovery after spinal cord injury. J Cell Biol 173(1):47–58. doi:10.1083/jcb.200508143
Schwab JM, Monnier PP, Schluesener HJ, Conrad S, Beschorner R, Chen L, Meyermann R, Mueller BK (2005) Central nervous system injury-induced repulsive guidance molecule expression in the adult human brain. Arch Neurol 62(10):1561–1568. doi:10.1001/archneur.62.10.1561
Zhang G, Zhang JH, Feng J, Li Q, Wu X, Qin X (2011) Electrical stimulation of olfactory bulb downregulates RGMa expression after ischemia/reperfusion injury in rats. Brain Res Bull 86(3–4):254–261. doi:10.1016/j.brainresbull.2011.08.002
The State Science and Technology Commission of the People’s Republic of China (1988-11-14) Regulations for the administration of affairs concerning experimental animals. Decree No. 2
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20(1):84–91
Langhauser F, Gob E, Kraft P, Geis C, Schmitt J, Brede M, Gobel K, Helluy X, Pham M, Bendszus M, Jakob P, Stoll G, Meuth SG, Nieswandt B, McCrae KR, Kleinschnitz C (2012) Kininogen deficiency protects from ischemic neurodegeneration in mice by reducing thrombosis, blood-brain barrier damage, and inflammation. Blood 120(19):4082–4092. doi:10.1182/blood-2012-06-440057
Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M, Chopp M (2001) Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke 32(4):1005–1011
Jiang B, Liu X, Chen H, Liu D, Kuang Y, Xing B, Chen Z (2010) Ischemic postconditioning attenuates renal ischemic/reperfusion injury in mongrel dogs. Urology 76(6):1519e1–1519e7. doi:10.1016/j.urology.2010.06.055
Wang ZF, Wang NP, Harmouche S, Philip T, Pang XF, Bai F, Zhao ZQ (2013) Postconditioning promotes the cardiac repair through balancing collagen degradation and synthesis after myocardial infarction in rats. Basic Res Cardiol 108(1):318. doi:10.1007/s00395-012-0318-9
Knudsen AR, Kannerup AS, Dich R, Funch-Jensen P, Gronbaek H, Kruhoffer M, Mortensen FV (2012) Ischemic pre- and postconditioning has pronounced effects on gene expression profiles in the rat liver after ischemia/reperfusion. Am J Physiol Gastrointest Liver Physiol 303(4):G482–G489. doi:10.1152/ajpgi.00337.2011
Kim M, Park SW, D’Agati VD, Lee HT (2012) Isoflurane post-conditioning protects against intestinal ischemia-reperfusion injury and multiorgan dysfunction via transforming growth factor-beta1 generation. Ann Surg 255(3):492–503. doi:10.1097/SLA.0b013e3182441767
Staat P, Rioufol G, Piot C, Cottin Y, Cung TT, L’Huillier I, Aupetit JF, Bonnefoy E, Finet G, Andre-Fouet X, Ovize M (2005) Postconditioning the human heart. Circulation 112(14):2143–2148. doi:10.1161/CIRCULATIONAHA.105.558122
Heusch G (2012) Cardioprotection: chances and challenges of its translation to the clinic. Lancet. doi:10.1016/S0140-6736(12)60916-7
Yellon DM, Hausenloy DJ (2005) Realizing the clinical potential of ischemic preconditioning and postconditioning. Nat Clin Pract Cardiovasc Med 2(11):568–575. doi:10.1038/ncpcardio0346
Kloner RA, Rezkalla SH (2006) Preconditioning, postconditioning and their application to clinical cardiology. Cardiovasc Res 70(2):297–307. doi:10.1016/j.cardiores.2006.01.012
Burda J, Danielisova V, Nemethova M, Gottlieb M, Matiasova M, Domorakova I, Mechirova E, Ferikova M, Salinas M, Burda R (2006) Delayed postconditionig initiates additive mechanism necessary for survival of selectively vulnerable neurons after transient ischemia in rat brain. Cell Mol Neurobiol 26(7–8):1141–1151. doi:10.1007/s10571-006-9036-x
Ren C, Yan Z, Wei D, Gao X, Chen X, Zhao H (2009) Limb remote ischemic postconditioning protects against focal ischemia in rats. Brain Res 1288:88–94. doi:10.1016/j.brainres.2009.07.029
Conrad S, Genth H, Hofmann F, Just I, Skutella T (2007) Neogenin-RGMa signaling at the growth cone is bone morphogenetic protein-independent and involves RhoA, ROCK, and PKC. J Biol Chem 282(22):16423–16433. doi:10.1074/jbc.M610901200
Kubo T, Tokita S, Yamashita T (2012) Repulsive guidance molecule-a and demyelination: implications for multiple sclerosis. J Neuroimmun Pharmacol 7(3):524–528. doi:10.1007/s11481-011-9334-z
Liu P-W, Chen M-F, Tsai AP-Y, Lee TJF (2012) STAT1 mediates oroxylin A inhibition of iNOS and pro-inflammatory cytokines expression in microglial BV-2 cells. PLoS ONE 7(12):e50363. doi:10.1371/journal.pone.0050363
Block ML, Zecca L, Hong JS (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8(1):57–69. doi:10.1038/nrn2038
Acknowledgments
This reported work was supported in part by research grants from the National Science Foundation of China (NSFC No. 30770762, 30970987) and Medical Science Foundation of the First Affiliated Hospital of Chongqing Medical University (No. YXJJ2009-02, YXJJ2009-14).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kong, Y., Rogers, M.R. & Qin, X. Effective Neuroprotection by Ischemic Postconditioning is Associated with a Decreased Expression of RGMa and Inflammation Mediators in Ischemic Rats. Neurochem Res 38, 815–825 (2013). https://doi.org/10.1007/s11064-013-0984-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-0984-5