Abstract
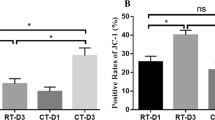
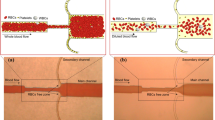
Growing evidence has suggested the feasibility and effectiveness of blood coagulation with low temperature plasma (LTP) at atmospheric pressure. In the present study we examined morphological changes and contents released out from platelet after LTP treatment to investigate the mechanism of LTP accelerate blood coagulation. Changes of isolated rat platelets treated with LTP had been detected by flow cytometry, transmission electron microscopy (TEM) and scanning electron microscopy (SEM). Additionally, a scheme that the mixture of different color fluorescent stained platelets treated by LTP and then detected by flow cytometry had proposed to quantitatively assess plasma triggered platelets aggregation. Furthermore, we had used label free quantitative mass spectrometry to identify changes in the supernatant proteome released from LTP treated platelets. On the surface of LTP treatment hastened whole rat blood clots, a layer of semi-transparent film had formed, which had been demonstrated to form from platelet-like membrane structure by ultrastructural analysis with TEM. Flow cytometry combined with fluorescent staining confirmed that LTP treatment promoted platelet aggregation. The observation of plasma treated stained platelets under fluorescence confocal microscopy also confirmed this judgment. We separated the supernatant from the platelets treated with LTP, and then analyzed their differences using mass spectrometry. Compared with control group, Gene ontology (GO) analysis showed that the concentration of 16 protein molecules in the experimental group was increased. LTP treatment can promotes platelet aggregation and the release of platelet activating proteins, which is the potential mechanism of speeding up blood coagulation.







Similar content being viewed by others
References
Rndt S, Unger P, Wacker E et al (2013) Cold atmospheric plasma (CAP) changes gene expression of key molecules of the wound healing machinery and improves wound healing in vitro and in vivo. PLoS ONE. https://doi.org/10.1371/journal.pone.0079325
Laroussi M, Lu X, Keidar M (2017) Perspective: the physics, diagnostics, and applications of atmospheric pressure low temperature plasma sources used in plasma medicine. J Appl Phys 122:1–19. https://doi.org/10.1063/1.4993710
Laroussi M (2005) Low temperature plasma-based sterilization: overview and state-of-the-art. Plasma Process Polym 2:391–400. https://doi.org/10.1002/ppap.200400078
McCombs G, Darby M (2010) New discoveries and directions for medical, dental and dental hygiene research: low temperature atmospheric pressure plasma. Int J Dent Hyg 8:10–15. https://doi.org/10.1111/j.1601-5037.2009.00386.x
Heinlin J, Morfill G, Landthaler M et al (2010) Plasma medicine: possible applications in dermatology. JDDG J Deutsch Dermatol Ges 8:968–976. https://doi.org/10.1111/j.1610-0387.2010.07495.x
Barekzi N, Laroussi M (2013) Effects of low temperature plasmas on cancer cells. Plasma Process Polym 10:1039–1050
Daeschlein G, Napp M, von Podewils S et al (2014) In vitro susceptibility of multidrug resistant skin and wound pathogens against low temperature atmospheric pressure plasma jet (APPJ) and dielectric barrier discharge plasma (DBD). Plasma Process Polym 11:175–183. https://doi.org/10.1002/ppap.201300070
Yan K, Jin Q, Zheng C et al (2018) Pulsed cold plasma-induced blood coagulation and its pilot application in stanching bleeding during rat hepatectomy. Plasma Sci Technol. https://doi.org/10.1088/2058-6272/aa9b79
Beardsley DJS, Tang C, Chen BG et al (2003) The disulfide-rich region of platelet glycoprotein (GP) IIIa contains hydrophilic peptide sequences that bind anti-GPIIIa autoantibodies from patients with immune thrombocytopenic purpura (ITP). Biophys Chem 105:503–515. https://doi.org/10.1016/S0301-4622(03)00111-X
Nomura Y, Takamatsu T, Kawano H et al (2017) Investigation of blood coagulation effect of nonthermal multigas plasma jet in vitro and in vivo. J Surg Res 219:302–309. https://doi.org/10.1016/j.jss.2017.06.055
García-Alcantara E, López-Callejas R, Morales-Ramírez PR et al (2013) Accelerated mice skin acute wound healing in vivo by combined treatment of argon and helium plasma needle. Arch Med Res 44:169–177. https://doi.org/10.1016/j.arcmed.2013.02.001
Miyamoto K, Ikehara S, Sakakita H, Ikehara Y (2017) Low temperature plasma equipment applied on surgical hemostasis and wound healings. J Clin Biochem Nutr 60:25–28. https://doi.org/10.3164/jcbn.16-60
Ueda M, Yamagami D, Watanabe K et al (2015) Cover picture: plasma process. polym. 12∕2015. Plasma Process Polym 12:1329–1329. https://doi.org/10.1002/ppap.201570043
Graves DB (2017) Mechanisms of plasma medicine: coupling plasma physics, biochemistry, and biology. IEEE Trans Radiat Plasma Med Sci 1:281–292. https://doi.org/10.1109/trpms.2017.2710880
Yadav S, Storrie B (2017) The cellular basis of platelet secretion: emerging structure/function relationships. Platelets 28:108–118. https://doi.org/10.1080/09537104.2016.1257786
Broos K, Feys HB, De Meyer SF et al (2011) Platelets at work in primary hemostasis. Blood Rev 25:155–167. https://doi.org/10.1016/J.BLRE.2011.03.002
Kalghatgi SU, Fridman G, Cooper M et al (2007) Mechanism of blood coagulation by nonthermal atmospheric pressure dielectric barrier discharge plasma. IEEE Trans Plasma Sci 35:1559–1566. https://doi.org/10.1109/TPS.2007.905953
Kuo SP, Tarasenko O, Chang J et al (2009) Contribution of a portable air plasma torch to rapid blood coagulation as a method of preventing bleeding. New J Phys. https://doi.org/10.1088/1367-2630/11/11/115016
Yousfi M, Merbahi N, Pathak A, Eichwald O (2014) Low-temperature plasmas at atmospheric pressure: toward new pharmaceutical treatments in medicine. Fundam Clin Pharmacol 28:123–135. https://doi.org/10.1111/fcp.12018
Bekeschus S, Brüggemeier J, Hackbarth C et al (2017) Platelets are key in cold physical plasma-facilitated blood coagulation in mice. Clin Plasma Med 7–8:58–65. https://doi.org/10.1016/J.CPME.2017.10.001
Bekeschus S, Brüggemeier J, Hackbarth C et al (2018) The feed gas composition determines the degree of physical plasma-induced platelet activation for blood coagulation. Plasma Sour Sci Technol. https://doi.org/10.1088/1361-6595/aaaf0e
Liu Z, Zheng C, Yan K et al (2016) A microsecond-pulsed cold plasma jet for medical application. Plasma Med 6:179–191. https://doi.org/10.1615/plasmamed.2016019251
Wang XF, Fang QQ, Jia B et al (2020) Potential effect of non-thermal plasma for the inhibition of scar formation: a preliminary report. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-57703-6
Cuyper IM de, Meinders M, Vijver E van de et al (2013) A novel flow cytometry–based platelet aggregation assay. 121:70–81. https://doi.org/10.1182/blood-2012-06-437723. I.M.D.C
Mi H, Huang X, Muruganujan A et al (2017) PANTHER version 11: expanded annotation data from gene ontology and reactome pathways, and data analysis tool enhancements. Nucl Acids Res 45:D183–D189. https://doi.org/10.1093/nar/gkw1138
Miyamoto K, Ikehara S, Takei H et al (2016) Red blood cell coagulation induced by low-temperature plasma treatment. Arch Biochem Biophys 605:95–101. https://doi.org/10.1016/J.ABB.2016.03.023
Takamatsu T, Uehara K, Sasaki Y et al (2014) Investigation of reactive species using various gas plasmas. RSC Adv 4:39901–39905. https://doi.org/10.1039/C4RA05936K
Hristov M, Zernecke A, Bidzhekov K et al (2007) Importance of CXC chemokine receptor 2 in the homing of human peripheral blood endothelial progenitor cells to sites of arterial injury. Circ Res 100:590–597. https://doi.org/10.1161/01.RES.0000259043.42571.68
Ikehara S, Sakakita H, Ishikawa K et al (2015) Plasma blood coagulation without involving the activation of platelets and coagulation factors. Plasma Process Polym 12(12):1348–1353
Ke Z, Huang Q (2016) Haem-assisted dityrosine-cross-linking of fibrinogen under non-thermal plasma exposure: one important mechanism of facilitated blood coagulation. Sci Rep 6:1–8. https://doi.org/10.1038/srep26982
Bekeschus S, Poschkamp B, van der Linde J (2020) Medical gas plasma promotes blood coagulation via platelet activation. Biomaterials. https://doi.org/10.1016/j.biomaterials.2020.120433
Kim J, Kim JH, Chang B et al (2016) Hemorheological alterations of red blood cells induced by non-thermal dielectric barrier discharge plasma. Appl Phys Lett. https://doi.org/10.1063/1.4967451
Boyanova D, Nilla S, Birschmann I et al (2012) PlateletWeb: a systems biologic analysis of signaling networks in human platelets. Blood 119:e22–e34. https://doi.org/10.1182/blood-2011-10-387308.secretome
Maynard DM, Heijnen HFG, Horne MK et al (2007) Proteomic analysis of platelet α-granules using mass spectrometry. J Thromb Haemost 5:1945–1955. https://doi.org/10.1111/j.1538-7836.2007.02690.x
Schmidt A, Bekeschus S, Wende K et al (2017) A cold plasma jet accelerates wound healing in a murine model of full-thickness skin wounds. Exp Dermatol 26:156–162. https://doi.org/10.1111/exd.13156
Lu P, Ziuzina D, Cullen PJ, Bourke P (2018) Cover picture plasma process polym. 12/2018. Plasma Process Polym. https://doi.org/10.1002/ppap.201870026
Acknowledgements
Thanks for the technical support by the Core Facilities, Zhejiang University School of Medicine.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jia, B., Liu, J., Yin, S. et al. Low Temperature Plasma Treatment of Rat Blood is Accompanied by Platelet Aggregation. Plasma Chem Plasma Process 41, 955–972 (2021). https://doi.org/10.1007/s11090-021-10176-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-021-10176-5