Abstract
Purpose
Monitoring of reactive oxygen species (ROS) generation from photoirradiated compounds would be effective for the prediction of the phototoxic potential. The aim of this investigation was to clarify the possible role of biomimetic vehicle systems on the photochemical properties of phototoxic compounds, focusing on the singlet oxygen generation.
Materials and Methods
Nine phototoxic and one non-phototoxic compounds (200 μM), dissolved in Tween 20, sodium laurate, or sodium dodecyl sulfate (SDS) micellar solution, were exposed to UVA/B light (250 W/m2), and singlet oxygen generation was monitored by RNO bleaching methodology. Photochemical properties of photosensitizers were also evaluated by UV measurement, and the interaction of photosensitizers with surfactant micelles was assessed by Z-potential and NMR spectroscopic analyses.
Results
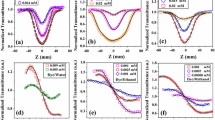
All phototoxic compounds tended to generate singlet oxygen under light exposure in the all micellar solutions tested. There appeared to be some differences in photoreactivity of both cationic and anionic photosensitizers among the micelles tested, whereas ROS data on anthracene, dissolved in three micellar solutions, were found to be quite similar. Photosensitizers exhibited no significant changes in UV spectral patterns among the dissolving micellar solutions. Addition of cationic photosensitizer at the final concentration of 100 μM into 100 mM SDS solution resulted in the 20 mV increase of zeta potential and transition of NMR spectral pattern, which would reflect the electrostatic interaction with anionic micelles.
Conclusion
Based on the data obtained, the photoreactivity of photosensitizing molecules, especially cationic and anionic photosensitizers, strongly depends on the physicochemical properties of the microenvironment.






Similar content being viewed by others
Abbreviations
- DNA:
-
deoxyribonucleic acid
- DMSO:
-
dimethyl sulfoxide
- NMR:
-
nuclear magnetic resonance
- NRU:
-
neutral red uptake
- NSAIDs:
-
non-steroidal anti-inflammatory drugs
- ROS:
-
reactive oxygen species
- SDS:
-
sodium dodecyl sulfate
- UV:
-
ultraviolet
References
A. M. Kligman and R. Breit. The identification of phototoxic drugs by human assay. J. Invest. Dermatol. 51:90–99 (1968).
N. J. Wainwright, P. Collins, and J. Ferguson. Photosensitivity associated with antibacterial agents. Drug Saf. 9:437–440 (1993).
J. A. Wiebe and D. E. Moore. Oxidation photosensitized by tetracyclines. J. Pharm. Sci. 66:186–189 (1977).
F. Vargas, I. Martinez Volkmar, J. Sequera, H. Mendez, J. Rojas, G. Fraile, M. Velasquez, and R. Medina. Photodegradation and phototoxicity studies of furosemide. Involvement of singlet oxygen in the photoinduced hemolysis and lipid peroxidation. J. Photochem. Photobiol. B 42:219–225 (1998).
L. Becker, B. Eberlein-Konig, and B. Przybilla. Phototoxicity of non-steroidal anti-inflammatory drugs: in vitro studies with visible light. Acta Derm. Venereol. 76:337–340 (1996).
P. Ball and G. Tillotson. Tolerability of fluoroquinolone antibiotics. Past, present and future. Drug Saf. 13:343–358 (1995).
G. Viola, G. Miolo, D. Vedaldi, and F. Dall’Acqua. In vitro studies of the phototoxic potential of the antidepressant drugs amitriptyline and imipramine. Farmaco 55:211–218 (2000).
N. Canudas and C. Contreras. Isolation and identification of the photodegradation products of the photosensitizing antidepressant drug clomipramine. Phototoxicity studies on erythrocytes. Pharmazie 57:405–408 (2002).
J. H. Epstein and B. U. Wintroub. Photosensitivity due to drugs. Drugs 30:42–57 (1985).
C. Arlett, L. Earl, J. Ferguson, N. Gibbs, J. Hawk, L. Henderson, B. Johnson, W. Lovell, H. D. Menage, S. Navaratnam, et al. British Photodermatology Group Workshop. Predictive in vitro methods for identifying photosensitizing drugs: a report. Br. J. Dermatol. 132:271–274 (1995).
E. Selvaag. Evaluation of phototoxic properties of oral antidiabetics and diuretics. Photohemolysis model as a screening method for recognizing potential photosensitizing drugs. Arzneimittelforschung 47:1031–1034 (1997).
J. C. Beani, R. Gautron, P. Amblard, F. Bastrenta, L. Harrouch, P. Jardon, and J. L. Reymond. Screening for drug photosensitization activity by measuring the variations in oxygen consumption of Bacillus subtilis. Photodermatology 2:101–106 (1985).
P. Portes, M. J. Pygmalion, E. Popovic, M. Cottin, and M. Mariani. Use of human reconstituted epidermis Episkin for assessment of weak phototoxic potential of chemical compounds. Photodermatol. Photoimmunol. Photomed. 18:96–102 (2002).
H. Spielmann, M. Liebsch, B. Doring, and F. Moldenhauer. First results of an EC/COLIPA validation project of in vitro phototoxicity testing methods. Altex 11:22–31 (1994).
S. Onoue and Y. Tsuda. Analytical studies on the prediction of photosensitive/phototoxic potential of pharmaceutical substances. Pharm. Res. 23:156–164 (2006).
C. S. Foote. Definition of type I and type II photosensitized oxidation. Photochem. Photobiol. 54:659 (1991).
D. E. Moore. Drug-induced cutaneous photosensitivity: incidence, mechanism, prevention and management. Drug Saf. 25:345–372 (2002).
A. M. Sautereau, C. Tournaire, M. Suares, J. F. Tocanne, and N. Paillous. Interactions of amiodarone with model membranes and amiodarone-photoinduced peroxidation of lipids. Biochem. Pharmacol. 43:2559–2566 (1992).
M. Budai, Z. Szabo, A. Zimmer, M. Szogyi, and P. Grof. Studies on molecular interactions between nalidixic acid and liposomes. Int. J. Pharm. 279:67–79 (2004).
I. Kraljic and S. E. Mohsni. A new method for the detection of singlet oxygen in aqueous solutions. Photochem. Photobiol. 28:577–581 (1978).
C. O. Rangel-Yagui, A. Pessoa Jr., and L. C. Tavares. Micellar solubilization of drugs. J. Pharm. Pharm. Sci. 8:147–165 (2005).
M. Monici, N. Mulinacci, P. Baglioni, and F. F. Vincieri. Flavone photoreactivity. UV-induced reactions in organic solvents and micellar systems. J. Photochem. Photobiol. B 20:167–172 (1993).
J. E. Weinstein and K. D. Polk. Phototoxicity of anthracene and pyrene to glochidia of the freshwater mussel Utterbackia imbecillis. Environ. Toxicol. Chem. 20:2021–2028 (2001).
S. Encinas, F. Bosca, and M. A. Miranda. Phototoxicity associated with diclofenac: a photophysical, photochemical, and photobiological study on the drug and its photoproducts. Chem. Res. Toxicol. 11:946–952 (1998).
G. P. Thami, S. Kaur, and A. J. Kanwar. Delayed reactivation of haloperidol induced photosensitive dermatitis by methotrexate. Postgrad. Med. J. 78:116–117 (2002).
J. Jagger. Solar-UV actions on living cells. Praeger Scientific, New York, 1985.
Y. N. Abdiche and D. G. Myszka. Probing the mechanism of drug/lipid membrane interactions using Biacore. Anal. Biochem. 328:233–243 (2004).
S. Kobayashi, K. Takeshima, C. B. Park, S. C. Kim, and K. Matsuzaki. Interactions of the novel antimicrobial peptide buforin 2 with lipid bilayers: proline as a translocation promoting factor. Biochemistry 39:8648–8654 (2000).
P. W. Wertz. Epidermal lipids. Semin. Dermatol. 11:106–113 (1992).
H. J. Yardley and R. Summerly. Lipid composition and metabolism in normal and diseased epidermis. Pharmacol. Ther. 13:357–383 (1981).
M. Fresta, S. Guccione, A. R. Beccari, P. M. Furneri, and G. Puglisi. Combining molecular modeling with experimental methodologies: mechanism of membrane permeation and accumulation of ofloxacin. Bioorg. Med. Chem. 10:3871–3889 (2002).
J. P. Martin Jr., K. Colina, and N. Logsdon. Role of oxygen radicals in the phototoxicity of tetracyclines toward Escherichia coli B. J. Bacteriol. 169:2516–2522 (1987).
K. Hirakawa, S. Kawanishi, and T. Hirano. The mechanism of guanine specific photooxidation in the presence of berberine and palmatine: activation of photosensitized singlet oxygen generation through DNA-binding interaction. Chem. Res. Toxicol. 18:1545–1552 (2005).
Acknowledgement
We are grateful to Ms. Satsuki Segawa for her excellent technical assistance throughout this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Onoue, S., Yamauchi, Y., Kojima, T. et al. Analytical Studies on Photochemical Behavior of Phototoxic Substances; Effect of Detergent Additives on Singlet Oxygen Generation. Pharm Res 25, 861–868 (2008). https://doi.org/10.1007/s11095-007-9383-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-007-9383-4