Abstract
Purpose
In overt hypercortisolism, growth hormone (GH) secretion is decreased and normalizes after surgery. In subclinical hypercortisolism (SH), GH secretion has been scarcely investigated. We assessed GH reserve in patients with and without SH and, in the former, also after recovery.
Methods
We enrolled 24 patients with adrenal adenomas, 12 with SH (SH+, 8 females, 58.3 ± 6.5 years) and 12 without SH (SH−; 11 females, 61.8 ± 10.6 years). SH was diagnosed in the presence of ≥2 out of: 1 mg overnight dexamethasone suppression test >83 nmol/L, urinary free cortisol (UFC) >193 nmol/day and ACTH levels <2.2 pmol/L. GH secretion was assessed by GHRH + Arginine test (GHRH–ARG) and age-adjusted serum IGF-I levels, expressed as SDS (IGF-I SDS). Eight SH+ patients were re-evaluated after the recovery from SH.
Results
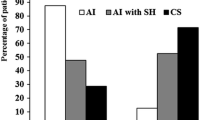
Age, gender, body mass index (BMI) and IGF-I SDS were comparable between SH+ and SH− patients. After GHRH–ARG the mean GH peak levels (GH-P) and GH response (as Area Under Curve, GH-AUC) were lower in SH+ than in SH− patients (15.2 ± 8.1 vs 44.5 ± 30.9 μg/L, P = 0.004 and 1,418 ± 803 vs 4,028 ± 2,476 μg/L/120 min, P = 0.002, respectively), after adjusting for age and BMI. The GH-AUC and GH-P levels were negatively associated with UFC after adjusting for age and BMI (β = −0.39, P = 0.02 and β = −0.4, P = 0.020 respectively). After recovery, GH-P levels and GH-AUC increased as compared to baseline (23.7 ± 16.3 vs 15.8 ± 10.2 μg/L, P = 0.036 and 2,549 ± 1,982 vs 1,618 ± 911 μg/L/120 min, P = 0.012, respectively).
Conclusions
GH secretion reserve is decreased in SH patients and increases after the recovery.



Similar content being viewed by others
References
Burguera B, Muruais C, Peñalva A, Dieguez C, Casanueva FF (1990) Dual and selective actions of glucocorticoids upon basal and stimulated growth hormone release in man. Neuroendocrinology 51:51–58
Smals AE, Pieters GF, Smals AG, Benraad TJ, Kloppenborg PW (1986) Human pancreatic growth hormone releasing hormone fails to stimulate human growth hormone both in Cushing’s disease and in Cushing’s syndrome due to adrenocortical adenoma. Clin Endocrinol (Oxf) 24:401–407
Leal-Cerro A, Pumar A, Garcia–Garcia E, Dieguez C, Casanueva FF (1994) Inhibition of growth hormone release after the combined administration of GHRH and GHRP-6 in patients with Cushing’s syndrome. Clin Endocrinol (Oxf) 41:649–654
Mazziotti G, Giustina A (2013) Glucocorticoids and the regulation of growth hormone secretion. Nat Rev Endocrinol. doi:10.1038/nrendo.2013.5
Wehrenberg WB, Bergman PJ, Stagg L, Ndon J, Giustina A (1990) Glucocorticoid inhibition of growth in rats: partial reversal with somatostatin antibodies. Endocrinology 127:2705–2708
Leal-Cerro A, Soto A, Martínez MA, Alvarez P, Isidro L, Casanueva FF, Dieguez C, Cordido F (2002) Effect of withdrawal of somatostatin plus growth hormone (GH)-releasing hormone as a stimulus of GH secretion in Cushing’s syndrome. Clin Endocrinol (Oxf) 57:745–749
Giustina A, Veldhuis JD (1998) Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Endocr Rev 19:717–797
Tzanela M, Karavitaki N, Stylianidou C, Tsagarakis S, Thalassinos NC (2004) Assessment of GH reserve before and after successful treatment of adult patients with Cushing’s syndrome. Clin Endocrinol (Oxf) 60:309–314
Mukherjee A, Murray RD, Teasdale GM, Shalet SM (2004) Acquired prolactin deficiency (APD) after treatment for Cushing’s disease is a reliable marker of irreversible severe GHD but does not reflect disease status. Clin Endocrinol (Oxf) 60:476–483
Boscaro M, Barzon L, Fallo F, Sonino N (2001) Cushing’s syndrome. Lancet 357:783–791
Chiodini I (2011) Clinical review: diagnosis and treatment of subclinical hypercortisolism. J Clin Endocrinol Metab 96:1223–1236
Terzolo M, Pia A, Alì A, Osella G, Reimondo G, Bovio S, Daffara F, Procopio M, Paccotti P, Borretta G, Angeli A (2002) Adrenal incidentaloma: a new cause of the metabolic syndrome? J Clin Endocrinol Metab 87:998–1003
Chiodini I, Morelli V, Masserini B, Salcuni AS, Eller-Vainicher C, Viti R, Coletti F, Guglielmi G, Battista C, Carnevale V, Iorio L, Beck-Peccoz P, Arosio M, Ambrosi B, Scillitani A (2009) Bone mineral density, prevalence of vertebral fractures and bone quality in patients with adrenal incidentalomas with and without subclinical hypercortisolism: an Italian Multicenter Study. J Clin Endocrinol Metab 94:3207–3214
Chiodini I, Torlontano M, Scillitani A, Arosio M, Bacci S, Di Lembo S, Epaminonda P, Augello G, Enrini R, Ambrosi B, Adda G, Trischitta V (2005) Association of subclinical hypercortisolism with type 2 diabetes mellitus: a case-control study in hospitalized patients. Eur J Endocrinol 15:837–844
Terzolo M, Osella G, Alì A, Borretta G, Cesario F, Paccotti P, Angeli A (1998) Subclinical Cushing’s syndrome in adrenal incidentaloma. Clinical Endocrinol (Oxf) 48:89–97
Terzolo M, Bossoni S, Alí A, Doga M, Reimondo G, Milani G, Peretti P, Manelli F, Angeli A, Giustina A (2000) Growth hormone (GH) responses to GH-releasing hormone alone or combined with arginine in patients with adrenal incidentaloma: evidence for enhanced somatostatinergic tone. J Clin Endocrinol Metab 85:1310–1315
Tzanela M, Zianni D, Stylianidou Ch, Karavitaki N, Tsagarakis S, Thalassinos NC (2005) Evaluation of GH reserve in patients with adrenal incidentalomas and biochemical evidence of subclinical autonomous glucocorticoid hypersecretion. Clin Endocrinol (Oxf) 62:597–602
Murray RD, Adams JE, Shalet SM (2004) Adults with partial growth hormone deficiency have an adverse body composition. J Clin Endocrinol Metab 89:1586–1591
Molitch ME, Clemmons DR, Malozowski S, Merriam GR, Vance ML (2011) Evaluation and treatment of adult growth hormone deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 96:1587–1609
NIH state-of-the-science statement on management of the clinically inapparent adrenal mass (“incidentaloma”) (2002) NIH Consens. State Sci Statements 19: 1–25
Ho KK (2007) 2007 GH deficiency consensus workshop participants consensus guidelines for the diagnosis and treatment of adults with GH deficiency II: a statement of the GH research society in association with the European society for Pediatric Endocrinology, Lawson Wilkins society, European Society of Endocrinology, Japan Endocrine Society, and Endocrine Society of Australia. Eur J Endocrinol 157:695–700
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentration in men. Diabetologia 28:412–419
Toniato A, Merante-Boschin I, Opocher G, Pelizzo MR, Schiavi F, Ballotta E (2009) Surgical versus conservative management for subclinical Cushing syndrome in adrenal incidentalomas: a prospective randomized study. Ann Surg 249:388–391
Chiodini I, Morelli V, Salcuni AS, Eller-Vainicher C, Torlontano M, Coletti F, Iorio L, Cuttitta A, Ambrosio A, Vicentini L, Pellegrini F, Copetti M, Beck-Peccoz P, Arosio M, Ambrosi B, Trischitta V, Scillitani A (2010) Beneficial metabolic effects of prompt surgical treatment in patients with an adrenal incidentaloma causing biochemical hypercortisolism. J Clin Endocrinol Metab 95:2736–2745
Eller-Vainicher C, Morelli V, Ulivieri FM, Palmieri S, Zhukouskaya VV, Cairoli E, Pino R, Naccarato R, Scillitani A, Beck-Peccoz P, Chiodini I (2012) Bone quality as measured by trabecular bone score (TBS) in patients with adrenal incidentalomas with and without subclinical hypercortisolism. J Bone Miner Res 27:2223–2230
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Standards
The study was conducted in accordance with applicable laws.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palmieri, S., Morelli, V., Salcuni, A.S. et al. GH secretion reserve in subclinical hypercortisolism. Pituitary 17, 470–476 (2014). https://doi.org/10.1007/s11102-013-0528-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-013-0528-7