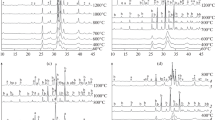
Octacalcium phosphate (OCP) is synthesized by pyrolysis of brushite CaHPO4 · H2O in an acetate buffer solution at 60 °C and pH = 7.5. OCP thermolysis is performed in the range 200 – 700 °C. The starting OCP and powder obtained is studied by IR-spectroscopy, electron microscopy, and x-ray-phase analyses. Thermolysis of OCP, prepared by this method proceeds at higher temperature than provided in publications. It is demonstrated for the first time in this work that the main feature of OCP thermolysis is formation of an intermediate apatite-like phase having within its composition, apart from orthophosphate and hydroxide ions, also pyrophosphate ions. Crystals of this intermediate phase retain the original OCPlamellar morphology entirely.




Similar content being viewed by others
References
M. Navarro, A. Michiardi, O. Castan, et al., “Biomaterials in orthopaedics, J. R. Soc. Interface., No. 5, 1137 – 1158 (2008).
T. Kanazawa, Inorganic Phosphate Materials [Russian translation], Naukova Dumka, Kiev (1998).
T. V. Safronova, V. I. Putlyaev, M. A. Shekhirev, and Yu. D. Tret’yakov, RF Patent 2391316 C1, method for preparing ceramic biodegradable material consisting of calcium pyrophosphate and tricalcium phosphate, Claim, 12.30.08, Publ. 06 – 10.2010.
T. V. Safronov, V. I. Pumlyaev,M. A. Shekhirev, et al., “Composite material containing bioresorbable phase,” Steklo Keram., NO. 3, 31 – 35 (2007).
V. I. Putlyaev, T. V. Safronova, E. V. Kukueva, and Yu. D. Tret’yakov, RF Patent 245653 C2, Method of charge preparation for obtaining ceramic biodegradable material, Claim 06.24.10, Publ. 07.20. 2012.
E. Boanini, M. Gazzano, K. Rubini, et al., “Collapsed octacalcium phosphate stabilized by ionic substitutions,” Crystal Growth and Design, No. 10, 3612 – 3617 (2010).
Author information
Authors and Affiliations
Additional information
Translated from Novye Ogneupory, No. 10, pp. 52 – 56, October, 2013.
Rights and permissions
About this article
Cite this article
Putlyaev, V.I., Kukueva, E.V., Safronova, T.V. et al. Features of Octacalcium Phosphate Thermolysis. Refract Ind Ceram 54, 420–424 (2014). https://doi.org/10.1007/s11148-014-9624-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11148-014-9624-0