Abstract
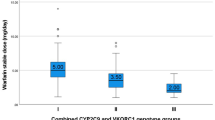
The dosage of warfarin is restricted due to its narrow therapeutic index, so, the required dose must be adapted individually to each patient. Variations in warfarin dosage are influenced by genetic factors, the changes in patient diet, anthropometric and clinical parameters. To determine whether VKORC1 G3730A and CYP4F2 G1347A genotypes contribute to warfarin dosage in patients during initiation and long-term anticoagulation treatment after heart valve surgery. From totally 307 patients, who underwent heart valve surgery, 189 patients (62 %) who had been treated with warfarin more than 3 months, were included into the study. A hierarchical stepwise multivariate linear regression model showed, that during initiation clinical factors can explain 17 % of the warfarin dose variation. The addition of CYP2C9 and VKORC1 G-1639A genotype raises the accuracy about twice—to 32 %. The CYP4F2 G1347A genotype can add again about 2–34 %. During long-term treatment clinical factors explain about 26 % of warfarin dose variation. If the CYP2C9 *2, *3, VKORC1*2 alleles are detected, model can explain about 49 % in dose variation. The *3 allele of VKORC1 raises the accuracy by 1–50 %. The carriers of CYP4F2 A1347A genotype required higher daily warfarin doses during initiation of warfarin therapy after heart valve surgery than comparing to G/G and G/A carriers, but during the longer periods of warfarin use, the dosage of warfarin depended significantly on VKORC1 *3 allele (G3730A polymorphism) and on the thyroid stimulating hormone level in the blood plasma.
Similar content being viewed by others
References
Moreau C, Loriot MA, Siguret V (2012) Vitamin K antagonists: from discovery to pharmacogenetics. Ann Biol Clin 70:539–551
Rieder MJ, Reiner AP, Gage BF, Nickerson DA, Eby CS, McLeod HL, Blough DK, Thummel KE, Veenstra DL, Rettie AE (2005) Effect of VKORC1 haplotypes on transcriptional regulation and warfarin dose. N Engl J Med 352:2285–2293
Lipinski S, Bremer L, Lammers T, Thieme F, Schreiber S, Rosenstiel P (2011) Coagulation and inflammation. Molecular insights and diagnostic implications. Hamostaseologie 31:94–102
Schalekamp T, de Boer A (2010) Pharmacogenetics of oral anticoagulant therapy. Curr Pharm Des 16:187–203
Cini M, Legnani C, Cosmi B, Guazzaloca G, Valdrè L, Frascaro M, Palareti G (2012) A new warfarin dosing algorithm including VKORC1 3730 G>A polymorphism: comparison with results obtained by other published algorithms. Eur J Clin Pharmacol 68:1167–1174
Gage BF, Eby C, Johnson JA, Deych E, Rieder MJ, Ridker PM, Milligan PE, Grice G, Lenzini P, Rettie AE, Aquilante CL, Grosso L, Marsh S, Langaee T, Farnett LE, Voora D, Veenstra DL, Glynn RJ, Barrett A, McLeod HL (2008) Use of pharmacogenetic and clinical factors to predict the therapeutic dose of warfarin. Clin Pharmacol Ther 84:326–331
Ozer N, Cam N, Tangurek B, Ozer S, Uyarel H, Oz D, Guney MR, Ciloglu F (2010) The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements in an adult Turkish population. Heart Vessels 25:155–162
Miao L, Yang J, Huang C, Shen Z (2007) Contribution of age, body weight, and CYP2C9 and VKORC1 genotype to the anticoagulant response to warfarin: proposal for a new dosing regimen in Chinese patients. Eur J Clin Pharmacol 63:1135–1141
Sconce EA, Khan TI, Wynne HA, Avery P, Monkhouse L, King BP, Wood P, Kesteven P, Daly AK, Kamali F (2005) The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements: proposal for a new dosing regimen. Blood 106:2329–2333
Caldwell MD, Awad T, Johnson JA, Gage BF, Falkowski M, Gardina P, Hubbard J, Turpaz Y, Langaee TY, Eby C, King CR, Brower A, Schmelzer JR, Glurich I, Vidaillet HJ, Yale SH, Qi Zhang K, Berg RL, Burmester JK (2008) CYP4F2 genetic variant alters required warfarin dose. Blood 111:4106–4112
McDonald MG, Rieder MJ, Nakano M, Hsia CK, Rettie AE (2009) CYP4F2 is a vitamin K1 oxidase: an explanation for altered warfarin dose in carriers of the V433M variant. Mol Pharmacol 75:1337–1346
Bejarano-Achache I, Levy L, Mlynarsky L, Bialer M, Muszkat M, Caraco Y (2012) Effects of CYP4F2 polymorphism on response to warfarin during induction phase: a prospective, open-label, observational cohort study. Clin Ther 34:811–823
Müllenbach R, Lagoda PJ, Welter C (1989) An efficient salt-chloroform extraction of DNA from blood and tissues. Trends Genet 5:391
Yoo SH, Nah HW, Jo MW, Kang DW, Kim JS, Koh JY, Kwon SU (2009) Age and body weight adjusted warfarin initiation program for ischaemic stroke patients. Eur J Neurol 16:1100–1105
Loun B, Hage DS (1995) Characterization of thyroxine-albumin binding using high-performance affinity chromatography. II. Comparison of the binding of thyroxine, triiodothyronines and related compounds at the warfarin and indole sites of human serum albumin. J Chromatogr B 665:303–314
Ascenzi P, Fasano M (2010) Allostery in a monomeric protein: the case of human serum albumin. Biophys Chem 148:16–22
Klotz U (2007) Antiarrhythmics: elimination and dosage considerations in hepatic impairment. Clin Pharmacokinet 46:985–996
Miners JO, Birkett DJ (1998) Cytochrome P4502C9: an enzyme of major importance in human drug metabolism. Br J Clin Pharmacol 45:525–538
Malerba M, Ragnoli B (2008) Ambroxol in the 21st century: pharmacological and clinical update. Expert Opin Drug Metab Toxicol 4:1119–1129
De Mey C, Peil H, Kölsch S, Bubeck J, Vix JM (2008) Efficacy and safety of ambroxol lozenges in the treatment of acute uncomplicated sore throat. EBM-based clinical documentation. Arzneimittelforschung 58:557–568
Wang XM, Hamza M, Gordon SM, Wahl SM, Dionne RA (2008) COX inhibitors downregulate PDE4D expression in a clinical model of inflammatory pain. Clin Pharmacol Ther 84:39–42
Orme M, Breckenridge A, Brooks RV (1972) Interactions of benzodiazepines with warfarin. Br Med J 3:611–614
Domenici E, Bertucci C, Salvadori P, Wainer IW (1991) Use of a human serum albumin-based high-performance liquid chromatography chiral stationary phase for the investigation of protein binding: detection of the allosteric interaction between warfarin and benzodiazepine binding sites. J Pharm Sci 80:164–166
Bird J, Carmona C (2008) Probable interaction between warfarin and torsemide. Ann Pharmacother 42:1893–1898
Sanderson S, Emery J, Higgins J (2005) CYP2C9 gene variants, drug dose, and bleeding risk in warfarin-treated patients: a HuGEnet systematic review and meta-analysis. Genet Med 7:97–104
Du L, Yin H, Morrow JD, Strobel HW, Keeney DS (2009) 20-Hydroxylation is the CYP-dependent and retinoid-inducible leukotriene B4 inactivation pathway in human and mouse skin cells. Arch Biochem Biophys 484:80–86
Kim HS, Lee SS, Oh M, Jang YJ, Kim EY, Han IY, Cho KH, Shin JG (2009) Effect of CYP2C9 and VKORC1 genotypes on early-phase and steady-state warfarin dosing in Korean patients with mechanical heart valve replacement. Pharmacogenet Genomics 19:103–112
Huang SW, Chen HS, Wang XQ, Huang L, Xu DL, Hu XJ, Huang ZH, He Y, Chen KM, Xiang DK, Zou XM, Li Q, Ma LQ, Wang HF, Chen BL, Li L, Jia YK, Xu XM (2009) Validation of VKORC1 and CYP2C9 genotypes on interindividual warfarin maintenance dose: a prospective study in Chinese patients. Pharmacogenet Genomics 19:226–234
Tatarunas V, Lesauskaite V, Veikutiene A, Grybauskas P, Jakuska P, Benetis R (2012) The combined effects of clinical factors and CYP2C9 and VKORC1 gene polymorphisms on initiating warfarin treatment in patients after cardiac valve surgery. J Heart Valve Dis 21:628–635
Acknowledgments
These studies were funded by a Grant (No. LIG-26/2010) from the Research Council of Lithuania.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tatarunas, V., Lesauskaite, V., Veikutiene, A. et al. The effect of CYP2C9, VKORC1 and CYP4F2 polymorphism and of clinical factors on warfarin dosage during initiation and long-term treatment after heart valve surgery. J Thromb Thrombolysis 37, 177–185 (2014). https://doi.org/10.1007/s11239-013-0940-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-013-0940-x