Abstract
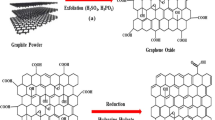
The adsorption capacity of graphene for removing phenol from aqueous solutions was evaluated and compared to those obtained for three different commercial activated carbons. In this study, graphene, W-35 activated carbon, RB2H2 activated carbon, and Nuchar granular activated carbon were investigated. Various techniques such as X-ray diffraction, BET surface analysis, and scanning electron microscopy were used to characterize the adsorbents. The adsorption of phenol onto graphene was investigated under different experimental conditions including temperature, solution pH, adsorbent dosage, and initial concentration of phenol. For comparison, adsorption experiments of phenol onto the activated carbons were also conducted. The adsorption of phenol onto graphene and activated carbons could be described by the pseudo-second-order kinetic model and the Langmuir equilibrium isotherm. The maximum adsorption capacities predicted by the Langmuir isotherm for graphene, W-35, RB2H2, and Nuchar granular activated carbons were found to be 233, 200, 91, and 167 mg/g, respectively. The thermodynamic study demonstrated that the adsorption of phenol onto graphene and activated carbons is a spontaneous and exothermic process. Regeneration of graphene was found to be possible using sodium hydroxide or methanol as eluents over five adsorption-desorption cycles.









Similar content being viewed by others
References
Abd Gami, A., et al. (2014). Phenol and phenolic compounds toxicity. Journal of Environmental Microbiology and Toxicology, 2(1), 11–23.
Ahmaruzzaman, M. (2008). Adsorption of phenolic compounds on low-cost adsorbents: a review. Advances in Colloid and Interface Science, 143(1–2), 48–67.
Akgerman, A., & Zardkoohi, M. (1996). Adsorption of phenolic compounds on fly ash. Journal of Chemical & Engineering Data, 41(2), 185–187.
Al Zarooni, M., & Elshorbagy, W. (2006). Characterization and assessment of Al Ruwais refinery wastewater. Journal of Hazardous Materials, 136(3), 398–405.
Alhamed, Y. A. (2009). Adsorption kinetics and performance of packed bed adsorber for phenol removal using activated carbon from dates’ stones. Journal of Hazardous Materials, 170(2–3), 763–770.
Alshehri, S. M., et al. (2014). Synthesis, characterization of curcumin based ecofriendly antimicrobial bio-adsorbent for the removal of phenol from aqueous medium. Chemical Engineering Journal, 254, 181–189.
Bazrafshan, E., Kord Mostafapour, F., & Jafari Mansourian, H. (2013). Phenolic compounds: health effects and its removal from aqueous environments by low cost adsorbents. Health Scope, 2(2), 65–66.
Bianco, A., et al. (2013). All in the graphene family—a recommended nomenclature for two-dimensional carbon materials. Carbon, 65, 1–6.
Bradder, P., et al. (2011). Dye adsorption on layered graphite oxide. Journal of Chemical & Engineering Data, 56(1), 138–141.
Bustillo-Lecompte, C. F., Knight, M., & Mehrvar, M. (2015). Assessing the performance of UV/H2O2 as a pretreatment process in toc removal of an actual petroleum refinery wastewater and its inhibitory effects on activated sludge. The Canadian Journal of Chemical Engineering, 93(5), 798–807.
Chakraborty, S., et al. (2010). Biodegradation of phenol by native microorganisms isolated from coke processing wastewater. Journal of Environmental Biology, 31(3), 293.
Coughlin, R. W., & Ezra, F. S. (1968). Role of surface acidity in the adsorption of organic pollutants on the surface of carbon. Environmental Science & Technology, 2(4), 291–297.
Djebbar, M., et al. (2012). Adsorption of phenol on natural clay. Applied Water Science, 2(2), 77–86.
Dursun, A. Y., & Kalayci, Ç. S. (2005). Equilibrium, kinetic and thermodynamic studies on the adsorption of phenol onto chitin. Journal of Hazardous Materials, 123(1–3), 151–157.
Ekpete, O., & Horsfall, M. J. (2011). Column removal of phenol and chlorophenol using a commercial activated carbon. Advances in Environmental Biology, 5(1), 194–200.
El-Naas, M. H., Al-Zuhair, S., & Alhaija, M. A. (2010). Removal of phenol from petroleum refinery wastewater through adsorption on date-pit activated carbon. Chemical Engineering Journal, 162(3), 997–1005.
Freundlich, H. M. F. (1906). Uber die adsorption in losungen. Zeitschrift für Physikalische Chemie, 57(A), 385.
Girods, P., et al. (2009). Activated carbons prepared from wood particleboard wastes: characterisation and phenol adsorption capacities. Journal of Hazardous Materials, 166(1), 491–501.
Gundogdu, A., et al. (2012). Adsorption of phenol from aqueous solution on a low-cost activated carbon produced from tea industry waste: equilibrium, kinetic, and thermodynamic study. Journal of Chemical & Engineering Data, 57(10), 2733–2743.
Gupta, V., Ali, I., & Saini, V. (2004). Removal of chlorophenols from wastewater using red mud: an aluminum industry waste. Environmental Science & Technology, 38(14), 4012–4018.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34(5), 451–465.
Ho, Y. S., & McKay, G. (2000). The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Research, 34(3), 735–742.
Hu, H., et al. (2015). Metal-free graphene-based catalyst—insight into the catalytic activity: a short review. Applied Catalysis, A: General, 492, 1–9.
Huang, J., Jin, X., & Deng, S. (2012). Phenol adsorption on an n-methylacetamide-modified hypercrosslinked resin from aqueous solutions. Chemical Engineering Journal, 192, 192–200.
Kazmi, M., et al. (2013). Removal of phenol from wastewater using activated waste tea leaves. Polish Journal of Chemical Technology, 15(2), 1–6.
Khalid, M., et al. (2004). Removal of phenol from water by adsorption using zeolites. Industrial & Engineering Chemistry Research, 43(17), 5275–5280.
Kulkarni, S. J., et al. (2013). Adsorption of phenol from wastewater in fluidized bed using coconut shell activated carbon. Procedia Engineering, 51, 300–307.
Langmuir, I. (1916). The constitution and fundamental properties of solids and liquids. Journal of the American Chemical Society, 38(11), 2221–2295.
Li, Y., et al. (2011). Adsorption of fluoride from aqueous solution by graphene. Journal of Colloid and Interface Science, 363(1), 348–354.
Li, Y., et al. (2012). Equilibrium, kinetic and thermodynamic studies on the adsorption of phenol onto graphene. Materials Research Bulletin, 47(8), 1898–1904.
Li, C., et al. (2017). Synergy of adsorption and photosensitization of graphene oxide for improved removal of organic pollutants. RSC Advances, 7(26), 16204–16209.
Lian, P., et al. (2010). Large reversible capacity of high quality graphene sheets as an anode material for lithium-ion batteries. Electrochimica Acta, 55(12), 3909–3914.
Lin, K., et al. (2009). Study the adsorption of phenol from aqueous solution on hydroxyapatite nanopowders. Journal of Hazardous Materials, 161(1), 231–240.
Liu, T., et al. (2012). Adsorption of methylene blue from aqueous solution by graphene. Colloids and Surfaces. B, Biointerfaces, 90, 197–203.
Ma, Y., et al. (2013). Removal of phenol by powdered activated carbon adsorption. Frontiers of Environmental Science & Engineering, 7(2), 158–165.
Mattson, J., et al. (1969). Surface chemistry of active carbon: specific adsorption of phenols. Journal of Colloid and Interface Science, 31, 116–130.
Mehrizad, A. & Gharbani, P. (2014). Decontamination of 4-chloro-2-nitrophenol from aqueous solution by graphene adsorption: equilibrium, kinetic, and thermodynamic studies. Polish Journal of Environmental Studies, 23(26), 2111–2116.
Moradi, O., et al. (2010). The studies of equilibrium and thermodynamic adsorption of Pb(II), Cd(II) and Cu(II) ions from aqueous solution onto SWCNTs and SWCNT–COOH surfaces. Fullerenes, Nanotubes, and Carbon Nanostructures, 18(3), 285–302.
Moreno-Castilla, C. (2004). Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon, 42(1), 83–94.
Mukherjee, R., & De, S. (2014). Adsorptive removal of phenolic compounds using cellulose acetate phthalate–alumina nanoparticle mixed matrix membrane. Journal of Hazardous Materials, 265, 8–19.
Păcurariu, C., et al. (2013). Adsorption of phenol and p-chlorophenol from aqueous solutions on poly (styrene-co-divinylbenzene) functionalized materials. Chemical Engineering Journal, 222, 218–227.
Pigatto, G., et al. (2013). Chitin as biosorbent for phenol removal from aqueous solution: equilibrium, kinetic and thermodynamic studies. Chemical Engineering and Processing: Process Intensification, 70, 131–139.
Qin, G., et al. (2013). Preparation of hydrophobic granular silica aerogels and adsorption of phenol from water. Applied Surface Science, 280, 806–811.
Qiu, H., et al. (2009). Critical review in adsorption kinetic models. Journal of Zhejiang University. Science. A, 10(5), 716–724.
Roostaei, N., & Tezel, F. H. (2004). Removal of phenol from aqueous solutions by adsorption. Journal of Environmental Management, 70(2), 157–164.
Saha, P. D., Srivastava, J., & Chowdhury, S. (2013). Removal of phenol from aqueous solution by adsorption onto seashells: equilibrium, kinetic and thermodynamic studies. J Water Reuse Desal, 3(2), 119–127.
Shang, H., et al. (2015). Preparing high surface area porous carbon from biomass by carbonization in a molten salt medium. RSC Advances, 5(92), 75728–75734.
Singh, D. K., & Srivastava, B. (2002). Removal of phenol pollutants from aqueous solutions using various adsorbents. Journal of Scientific and Industrial Research, 61, 208–218.
Srivastava, V. C., et al. (2006). Adsorptive removal of phenol by bagasse fly ash and activated carbon: equilibrium, kinetics and thermodynamics. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 272(1–2), 89–104.
Sulaymon, A. H., & Ahmed, K. W. (2008). Competitive adsorption of furfural and phenolic compounds onto activated carbon in fixed bed column. Environmental Science & Technology, 42(2), 392–397.
Tancredi, N., et al. (2004). Phenol adsorption onto powdered and granular activated carbon, prepared from eucalyptus wood. Journal of Colloid and Interface Science, 279(2), 357–363.
Thommes, M., et al. (2015). Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC technical report). Pure and Applied Chemistry, 87(9–10), 1051–1069.
Tor, A., et al. (2006). Removal of phenol from aqueous phase by using neutralized red mud. Journal of Colloid and Interface Science, 300(2), 498–503.
Tran, H. N., You, S.-J., & Chao, H.-P. (2016). Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: a comparison study. Journal of Environmental Chemical Engineering, 4(3), 2671–2682.
Wang, G., et al. (2008). Facile synthesis and characterization of graphene nanosheets. Journal of Physical Chemistry C, 112(22), 8192–8195.
Wang, S., et al. (2013). Adsorptive remediation of environmental pollutants using novel graphene-based nanomaterials. Chemical Engineering Journal, 226, 336–347.
Worsley, M. A., et al. (2010). Synthesis of graphene aerogel with high electrical conductivity. Journal of the American Chemical Society, 132(40), 14067–14069.
Wu, T., et al. (2011). Adsorption characteristics of acrylonitrile, p-toluenesulfonic acid, 1-naphthalenesulfonic acid and methyl blue on graphene in aqueous solutions. Chemical Engineering Journal, 173(1), 144–149.
Xu, J., Wang, L., & Zhu, Y. (2012). Decontamination of bisphenol A from aqueous solution by graphene adsorption. Langmuir, 28(22), 8418–8425.
Yang, C., et al. (2006). Solvent extraction process development and on-site trial-plant for phenol removal from industrial coal-gasification wastewater. Chemical Engineering Journal, 117(2), 179–185.
Zhang, J. (2013). Phenol removal from water with potassium permanganate modified granular activated carbon. Journal of Environmental Protection, 04(05), 7.
Zhang, Y., et al. (2014). Recyclable removal of bisphenol a from aqueous solution by reduced graphene oxide–magnetic nanoparticles: adsorption and desorption. Journal of Colloid and Interface Science, 421, 85–92.
Zhao, G., Li, J., & Wang, X. (2011). Kinetic and thermodynamic study of 1-naphthol adsorption from aqueous solution to sulfonated graphene nanosheets. Chemical Engineering Journal, 173(1), 185–190.
Acknowledgements
The authors are grateful to the Research Office at The Petroleum Institute, Abu Dhabi for funding project [no. 14513]. The contribution of Dr. Pal Priyabrata is appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gaber, D., Abu Haija, M., Eskhan, A. et al. Graphene as an Efficient and Reusable Adsorbent Compared to Activated Carbons for the Removal of Phenol from Aqueous Solutions. Water Air Soil Pollut 228, 320 (2017). https://doi.org/10.1007/s11270-017-3499-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3499-x