Abstract
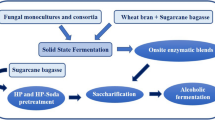
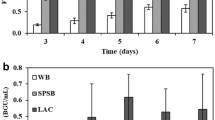
Pretreatment of lignocellulosic biomasses, the first step in their conversion to utilizable molecules requires very high energy (steam and electricity), corrosion resistant high-pressure reactors and high temperatures. These severe conditions not only add to the cost component of the entire process but also lead to the loss of sugars to the side reactions. Microbial pretreatments have been reported to be associated with reducing the cost factors as well as the severities of the reactions. Eight bioagents, including fungi and bacteria, were screened for their pretreatment effects on sugarcane trash. They narrowed down the C:N ratio of trash from 108:1 to a varying range of approximately 42:1 to 60:1.The maximum drop in C:N ratio of 61% was observed using Aspergillus terreus followed by Cellulomonas uda (52%) and Trichoderma reesei and Zymomonas mobilis (49%). The bioagents helped in degradation of sugarcane trash by production of cellulases, the maximum being produced by A. terreus, (12 fold) followed by C. uda (10 fold), Cellulomonas cartae (9 fold) and Bacillus macerans (8 fold). The microbial pretreatment of trash rendered the easy accessibility of sugars for enzymatic hydrolysis, which can be directed for production of alcohol.


Similar content being viewed by others
References
Akin DE, Sethuraman A, Morrison WH, Martin SA, Eriksson KE (1993) Microbial delignification with white rot fungi improves forage digestibility. Appl Environ Microbiol 59:4274–4282
Chandler JA, Jewel WJ, Gossett JM, van Soest PJ, Robertson JB (1980) Predicting methane fermentation biodegradability. Biotechnol Bioeng 10:93–107
Demian AL, Solomon NA (eds) (1986) Manual of industrial microbiology and biotechnology. American Society for Microbiology, Washington, DC
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 26:350–356
Eiland S, Leth M, Klamr M, Lind AM, Jensen HEK (2001) C and N turnover and lignocellulose degradation during composting of Miscanthus straw and liquid pig manure. Compost Sci Util 9:886–896
Eriksson KE, Grunewald A, Nilsson T, Valander L (1980) A scanning electron microscopy study of the growth and attack on wood by three white rot fungi and their cellulase less mutants. Hotz Forschung 34:204–213
Eriksson KE, Vallander L (1982) Properties of pulps from thermo-mechanical pulping of chips pretreated with fungi. Svensk Papperstidning 6:33–38
Fan LT, Gharpuray MM, Lee YH (1987) Design and economic evaluation of cellulose hydrolysis, Cellulose hydrolysis. Springer Verlag, New York
Filiminova M (1997) Microbiological activity in native soils. Field Notes Newsl. 23. Kerr Centre for Sustainable Agriculture, Poteau
Guerra G, Casado GRM, Arguelles J, Sacchez TMA, Manzano MA, Guzman T (2006) Cellulase production with sugarcane straw by Trichoderma citrinoviride on solid bed. Sugar Tech 8:30–35
Howarth WR, Elliot LF, Churchill DB (1995) Mechanisms regulating composting of high carbon to nitrogen ratio grass straw. Compost Sci Util 3:14–21
Horwitz W (1980) Lignin estimation, official methods of analysis (edition 13). Association of Analytical Chemists, pp 48–49
Kayhanian M, Tchobanoglous G (1992) Computation of C:N ratios for various organic fractions. Biocycle 33:58–60
Keller AF, Hamilton JE, Nguyen QA (2003) Microbial pretreatment of biomass: potential for reducing the severity of thermochemical biomass pretreatment. Appl Biochem Biotechnol 101:105–108
Lynd LR, Cushman JH, Nicholas RJ, Wyman CE (1991) Fuel ethanol from cellulosic biomass. Science 251:1318–1323
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Mandels M, Reese ET (1964) Fungal cellulases and the microbial decomposition of cellulosic fabric, Developments in Industrial Microbiology, vol 5. Society for Industrial Microbiology, Washington, DC, pp 5–20
Mandels M, Adreotti R, Roche C (1975) Measurements of saccharifying cellulase. Biotechnol Bioeng 6:1471–1493
Mosier N, Wyman C, Dale B, Elander R, Lee YY, Holtzapple M, Ladisch M (2005) Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour Technol 96:673–686
Margolles E, Hayes C, Harman G, Pentiila M (1996) Improved production of Trichoderma harzianum endochitanase by expression in Trichoderma reesei. Appl Environ Microbiol 62:2145–2151
Miyomoto K (1997) Cellulase production. Renewable biological systems for alternative sustainable energy production, FAO, Agricultural Services Bulletin, 128. Osaka University, Osaka, Japan
Page AL, Miller RH, Keeney DR (1982) Methods of soil analysis. Part 2, 2nd edn. Agron. Monogr. 9. ASA and SSSA, Madison, WI, pp 903–947
Parisi F (1989) Advances in lignocellulosic hydrolysis and in the utilization of hydrolysates. Adv Biochem Eng Biotechnol:38:53–87
Playne MJ (1984) Increased digestibility of bagasse by pretreatment with alkali and steam explosion. Biotechnol Bioeng 26:420
Ramos LP (2003) The chemistry involved in the steam treatment of lignocellulosic materials. Quim Nova 26:863–873
Reese ET, Siu RGH, Levinson HS (1950) The biological degradation of soluble cellulose derivatives and its relationship to the mechanism of cellulose hydrolysis. J Bacteriol 59:485–497
Sawada T, Kuwahara M, Nakamura Y, Suda H (1987) Effect of process of explosion for the effective utilization of biomass. Int Chem Eng 27:686–693
Sawada T, Nakamura Y, Kobayashi F, Kuwahara M, Watanabe T (1995) Effect of fungal pretreatment and steam explosion pretreatment on the enzymatic saccharification of the plant biomass. Biotechnol Bioeng 48:719–724
Schwald W, Smaridge T, Chan M, Breuil C, Saddler JN (1989) The influence of SO2 impregnation and fractionation on the product recovery and enzymatic hydrolysis of steam treated spruce wood. In: Coughlan MP (ed) Enzyme systems for lignocellulose degradation. Proceedings of the COST 84 bis Workshop, Ireland. Elsevier Applied Science, London, pp 231–242
Sluiter A, Hames B, Raymond R, Christopher S, Justin S, Templeton D (2005) Determination of total solids in biomass. National Renewable Energy Laboratory, National Bioenergy Centre, USA
Snedecor GW, Cochran WG (1967) Statistical methods. Oxford and IBH Publ., India
Somogyi N (1945). A new reagent for determining sugars. J Biol Chem 160:61–68
Tomatii U, Galli E, Pasetti L, Volterra E (1995) Bioremediation of olive mill waste waters by composting. Waste Manage Res 13:509–518
Updegroff DM (1969) Determination of cellulose in biological materials. Anal Biochem 32:420–424
Wichern F, Miiller T, Joergensen RG, Buerkert A (2004) Effect of manure quality and application forms on soil C and N turnover of a subtropical oasis soil under laboratory conditions. Biol Fertil Soils 39:165–171
Wood TM, Saddler JN (1988) Increasing the availability of cellulose in biomass material. Methods Enzymol 160:3–11
Acknowledgements
Authors thank the Director, Indian Institute of Sugarcane Research for his support and encouragement. Financial assistance by Indian Council of Agriculture (ICAR) through APCess project is dually acknowledged. Thanks are also due to National Center for Industrial Microorganisms (NCIM), National Chemical Laboratory (NCL), Pune, India for providing some microbial isolates.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, P., Suman, A., Tiwari, P. et al. Biological pretreatment of sugarcane trash for its conversion to fermentable sugars. World J Microbiol Biotechnol 24, 667–673 (2008). https://doi.org/10.1007/s11274-007-9522-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-007-9522-4