Abstract
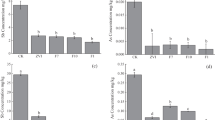
The role of indigenous bacteria in mobilization of sediment bound arsenic (As) into groundwater is investigated using subsurface sediment from Brahmaputra River Basin (BRB) and the Bacillus sp. strain IIIJ3-1, an indigenous species to BRB. Anaerobic sediment microcosms with varying organic carbon sources and terminal electron acceptors (TEAs) are used to illustrate the role of the test bacterium in As mobilization. The aquifer sediment shows an asymmetric distribution of As and Fe in its different phases. Among the TEAs added, NO3 amendment promotes higher cell growth, oxalic acid production and maximum release of sediment bound As. X-ray diffraction analysis further suggests that weathering of As bearing secondary minerals through bacterial action enhances As bioavailability, followed by dissimilatory reduction and thus promotes its mobilization into aqueous phase. Co-release pattern of other elements from the sediment indicates that release of As is decoupled from that of Fe. This study confirms that microbe-mediated mineral weathering followed by respiratory reduction of As facilitates mobilization of sediment hosted As into aqueous phase, and provides a better insight into the catabolic ability of groundwater bacteria in mobilization of sediment hosted As in BRB region.
Graphic abstract






Similar content being viewed by others
References
Ahmed KM, Bhattacharya P, Hasan MA, Akhter SH, Alam SM, Bhuyian MH, Imam MB, Khan AA, Sracek O (2004) Arsenic enrichment in groundwater of the alluvial aquifers in Bangladesh: an overview. Appl Geochem 19(2):181–200. https://doi.org/10.1016/j.apgeochem.2003.09.006
Alexandratos VG, Elzinga EJ, Reeder RJ (2007) Arsenate uptake by calcite: macroscopic and spectroscopic characterization of adsorption and incorporation mechanisms. Geochim Cosmochim Act 71(17):4172–4187. https://doi.org/10.1016/j.gca.2007.06.055
Aminuddin M, Nicholas DJ (1973) Sulphide oxidation linked to the reduction of nitrate and nitrite in Thiobacillus denitrificans. BBA-Bioenerget 325(1):81–93
Asta MP, Wang Y, Frutschi M, Viacava K, Loreggian L, Le Pape P, Le Vo P, Fernandez AM, Morin G, Bernier-Latmani R (2019) Microbially-mediated release of As from Mekong Delta peat sediments. Environ Sci Technol 53(17):10208–10217. https://doi.org/10.1021/acs.est.9b02887
Baig JA, Kazi TG, Arain MB, Shah AQ, Sarfraz RA, Afridi HI, Kandhro GA, Jamali MK, Khan S (2009) Arsenic fractionation in sediments of different origins using BCR sequential and single extraction methods. J Hazard Mater 167(1):745–751. https://doi.org/10.1016/j.jhazmat.2009.01.040
Baltrus JP, Granite EJ, Pennline HW, Stanko D, Hamilton H, Rowsell L, Poulston S, Smith A, Chu W (2010) Surface characterization of palladium–alumina sorbents for high-temperature capture of mercury and arsenic from fuel gas. Fuel 89(6):1323–1325. https://doi.org/10.1016/j.fuel.2009.09.030
Bauer M, Blodau C (2006) Mobilization of arsenic by dissolved organic matter from iron oxides, soils and sediments. Sci Total Environ 354(2):179–190. https://doi.org/10.1016/j.scitotenv.2005.01.027
Baviskar S, Choudhury R, Mahanta C (2015) Dissolved and solid-phase arsenic fate in an arsenic-enriched aquifer in the river Brahmaputra alluvial plain. Environ Monit Assess 187(3):1–14. https://doi.org/10.1007/s10661-015-4277-0
Baviskar SM, Mahanta C, Choudhary R (2011) Leaching of iron, controlling factors and implication to arsenic mobilization in an aquifer of the Brahmaputra Floodplain. In: Annual International Conference 2011 Programme RGS-IBM, London
Baziramakenga R, Simard RR, Leroux GD (1995) Determination of organic acids in soil extracts by ion chromatography. Soil Biol Biochem 27(3):349–356. https://doi.org/10.1016/0038-0717(94)00178-4
Benz M, Brune A, Schink B (1998) Anaerobic and aerobic oxidation of ferrous iron at neutral pH by chemoheterotrophic nitrate-reducing bacteria. Arch Microbiol 169(2):159–165. https://doi.org/10.1007/s002030050555
Berg M, Trang PT, Stengel C, Buschmann J, Viet PH, Van Dan N, Giger W, Stüben D (2008) Hydrological and sedimentary controls leading to arsenic contamination of groundwater in the Hanoi area, Vietnam: the impact of iron-arsenic ratios, peat, river bank deposits, and excessive groundwater abstraction. ChemGeol 249(1):91–112. https://doi.org/10.1016/j.chemgeo.2007.12.007
Bhattacharya P, Jacks G, Jana J, Sracek A, Gustafsson JP, Chatterjee D (2001) Geochemistry of the Holocene alluvial sediments of Bengal Delta Plain from West Bengal, India: implications on arsenic contamination in groundwater. Groundwater Arsenic Contam Bengal Delta Plain Bangladesh 3084:21–40
Blum JS, Bindi AB, Buzzelli J, Stolz JF, Oremland RS (1998) Bacillus arsenicoselenatis, sp. nov., and Bacillus selenitireducens, sp. nov.: two haloalkaliphiles from Mono Lake, California that respire oxyanions of selenium and arsenic. Arch Microbiol 171(1):19–30. https://doi.org/10.1007/s002030050673
Burnol A, Garrido F, Baranger P, Joulian C, Dictor MC, Bodénan F, Morin G, Charlet L (2007) Decoupling of arsenic and iron release from ferrihydrite suspension under reducing conditions: a biogeochemical model. Geochem Trans 8(1):1. https://doi.org/10.1186/1467-4866-8-12
Cavalca L, Corsini A, Zaccheo P, Andreoni V, Muyzer G (2013) Microbial transformations of arsenic: perspectives for biological removal of arsenic from water. Future Microbiol 8(6):753–768. https://doi.org/10.2217/fmb.13.38
Chakraborti D, Singh EJ, Das B, Shah BA, Hossain MA, Nayak B, Ahamed S, Singh NR (2008) Groundwater arsenic contamination in Manipur, one of the seven North-Eastern Hill states of India: a future danger. Environ Geol 56(2):381–390. https://doi.org/10.1007/s00254-007-1176-x
Charlet L, Chakraborty S, Appelo CA, Roman-Ross G, Nath B, Ansari AA, Lanson M, Chatterjee D, Mallik SB (2007) Chemodynamics of an arsenic “hotspot” in a West Bengal aquifer: a field and reactive transport modeling study. Appl Geochem 22(7):1273–1292. https://doi.org/10.1016/j.apgeochem.2006.12.022
Chetia M, Singh SK, Bora K, Kalita H, Saikia LB, Goawami DC, Srivastava RB, Sarma HP (2008) Groundwater arsenic contamination in three blocks of Golaghat district of Assam. J Indian Water Works Assoc 40(2):150–154
Crognale S, Amalfitano S, Casentini B, Fazi S, Petruccioli M, Rossetti S (2017) Arsenic-related microorganisms in groundwater: a review on distribution, metabolic activities and potential use in arsenic removal processes. Rev Environ Sci Bio/Technol 16(4):647–665
Cullen WR, Reimer KJ (1989) Arsenic speciation in the environment. Chem Rev 89(4):713–764. https://doi.org/10.1021/cr00094a002
Das S, Barooah M (2018) Characterization of siderophore producing arsenic-resistant Staphylococcus sp. strain TA6 isolated from contaminated groundwater of Jorhat, Assam and its possible role in arsenic geocycle. BMC Microbiol 18(1):104. https://doi.org/10.1186/s12866-018-1240-6
Devi NL, Ch YI, Shihua QI (2009) Recent status of arsenic contamination in groundwater of Northeastern India–a review. Kathmandu Univ J Sci Eng Technol 2(1):1–11
Edition F (2011) Guidelines for drinking-water quality. WHO Chron 38(4):104–108
Fang Y, Du Y, Feng H, Hu LF, Shen DS, Long YY (2015a) Sulfide oxidation and nitrate reduction for potential mitigation of H2S in landfills. Biodegradation 26(2):115–126. https://doi.org/10.1007/s10532-015-9720-y
Frey B, Rieder SR, Brunner I, Plötze M, Koetzsch S, Lapanje A, Brandl H, Furrer G (2010) Weathering-associated bacteria from the Damma glacier forefield: physiological capabilities and impact on granite dissolution. Appl Environ Microb 76(14):4788–4796. https://doi.org/10.1128/AEM.00657-10
Gallegos-Garcia M, Ramírez-Muñiz K, Song S (2012) Arsenic removal from water by adsorption using iron oxide minerals as adsorbents: a review. Miner Process Extr Metall Rev 33(5):301–315. https://doi.org/10.1080/08827508.2011.584219
Ghosh S (2018) Characterization of bacterial communities in arsenic contaminated groundwater of Brahmaputra River Basin (Doctoral dissertation, IIT, Kharagpur).
Ghosh S, Gupta A, Sarkar J, Verma S, Mukherjee A, Sar P (2019) Enrichment of indigenous arsenate reducing anaerobic bacteria from arsenic rich aquifer sediment of Brahmaputra river basin and their potential role in as mobilization. J Environ Sci Health A 54(7):635–647. https://doi.org/10.1080/10934529.2019.1579524
Ghosh S, Sar P (2013) Identification and characterization of metabolic properties of bacterial populations recovered from arsenic contaminated ground water of North East India (Assam). Water Res 47(19):6992–7005. https://doi.org/10.1016/j.watres.2013.08.044
Héry M, Van Dongen BE, Gill F, Mondal D, Vaughan DJ, Pancost RD, Polya DA, Lloyd JR (2010) Arsenic release and attenuation in low organic carbon aquifer sediments from West Bengal. Geobiology 8(2):155–168. https://doi.org/10.1111/j.1472-4669.2010.00233.x
IS 10500: (2012) Drinking Water—Specification; Bureau of Indian Standards; https://cgwb.gov.in/Documents/WQ-standards.pdf
Islam FS, BoothmanC GAG, Polya DA, Lloyd JR (2005) Potential role of the Fe(III)-reducing bacteria Geobacter and Geothrix in controlling arsenic solubility in Bengal delta sediments. Mineral Mag 69:865–875. https://doi.org/10.1180/0026461056950294
Islam FS, Gault AG, Boothman C, Polya DA, Charnock JM, Chatterjee D, Lloyd JR (2004) Role of metal-reducing bacteria in As release from Bengal delta sediments. Nature 430:68–71. https://doi.org/10.1038/nature02638
Jönsson J, Sherman DM (2008) Sorption of As (III) and As (V) to siderite, green rust (fougerite) and magnetite: implications for arsenic release in anoxic groundwaters. Chem Geol 255(1):173–181. https://doi.org/10.1016/j.chemgeo.2008.06.036
Kim Y, Kim C, Choi I, Rengaraj S, Yi J (2004) Arsenic removal using mesoporous alumina prepared via a templating method. Environ Sci Technol 38(3):924–931. https://doi.org/10.1021/es0346431
Kirk MF, Holm TR, Park J, Jin Q, Sanford RA, Fouke BW, Bethke CM (2004) Bacterial sulfate reduction limits natural arsenic contamination in groundwater. Geology 32(11):953–956
Kudo K, Yamaguchi N, Makino T, Ohtsuka T, Kimura K, Dong DT, Amachi S (2013) Release of arsenic from soil by a novel dissimilatory arsenate-reducing bacterium, Anaeromyxobacter sp. strain PSR-1. Appl Environ Microbiol 79(15):4635–4642
Macy JM, Nunan K, Hagen KD, Dixon DR, Harbour PJ, Cahill M, Sly LI (1996) Chrysiogenes arsenatis gen. nov., sp. nov., a new arsenate-respiring bacterium isolated from gold mine wastewater. Int J Syst Evol Microbiol 46(4):1153–1157. https://doi.org/10.1099/00207713-46-4-1153
Mailloux BJ, Alexandrova E, Keimowitz AR, Wovkulich K, Freyer GA, Herron M, Stolz JF, Kenna TC, Pichler T, Polizzotto ML, Dong H (2009) Microbial mineral weathering for nutrient acquisition releases arsenic. Appl Environ Microb 75:2558–2565. https://doi.org/10.1128/AEM.02440-07
McArthur JM, Banerjee DM, Hudson-Edwards KA, Mishra R, Purohit R, Ravenscroft P, Cronin A, Howarth RJ, Chatterjee A, Talukder T, Lowry D (2004) Natural organic matter in sedimentary basins and its relation to arsenic in anoxic ground water: the example of West Bengal and its worldwide implications. Appl Geochem 19(8):1255–1293. https://doi.org/10.1016/j.apgeochem.2004.02.001
McArthur JM, Ravenscroft P, Safiulla S, Thirlwall MF (2001) Arsenic in groundwater: testing pollution mechanisms for sedimentary aquifers in Bangladesh. Water Resour Res 37(1):109–117
Mohapatra B, Kazy SK, Sar P (2019) Comparative genome analysis of arsenic reducing, hydrocarbon metabolizing groundwater bacterium Achromobacter sp. KAs 3–5T explains its competitive edge for survival in aquifer environment. Genomics 111(6):1604–1619
Mohapatra B, Sarkar A, Joshi S, Chatterjee A, Kazy SK, Maiti MK, Satyanarayana T, Sar P (2017) An arsenate-reducing and alkane-metabolizing novel bacterium, Rhizobium arsenicireducens sp. nov., isolated from arsenic-rich groundwater. Archiv Microbiol 199(2):191–201
Mohapatra B, Satyanarayana T, Sar P (2018) Molecular and eco-physiological characterization of arsenic (As)-transforming Achromobacter sp. KAs 3–5T from As-contaminated groundwater of West Bengal, India. J Environ Sci Health Part A 53(10):915–924
Mukherjee A, von Brömssen M, Scanlon BR, Bhattacharya P, Fryar AE, Hasan MA, Ahmed KM, Chatterjee D, Jacks G, Sracek O (2008) Hydrogeochemical comparison and effects of overlapping redox zones on groundwater arsenic near the Western (Bhagirathi sub-basin, India) and Eastern (Meghna sub-basin, Bangladesh) margins of the Bengal Basin. J Contam Hydrol 99(1–4):31–48. https://doi.org/10.1016/j.jconhyd.2007.10.005
Nath B, Stüben D, Mallik SB, Chatterjee D, Charlet L (2008) Mobility of arsenic in West Bengal aquifers conducting low and high groundwater arsenic. Part I: comparative hydrochemical and hydrogeological characteristics. Appl Geochem 23(5):977–995. https://doi.org/10.1016/j.apgeochem.2007.11.016
Nickson RT, McArthur JM, Ravenscroft P, Burgess WG, Ahmed KM (2000) Mechanism of arsenic release to groundwater, Bangladesh and West Bengal. Appl Geochem 15:403–413. https://doi.org/10.1016/S0883-2927(99)00086-4
Omoregie EO, Couture RM, Van Cappellen P, Corkhill CL, Charnock JM, Polya DA, Vaughan D, Vanbroekhoven K, Lloyd JR (2013) Arsenic bioremediation by biogenic iron oxides and sulfides. Appl Environ Microbiol 79(14):4325–4335
Omoregie EO, Couture RM, Van Cappellen P, Corkhill CL, Charnock JM, Polya DA, Vaughan D, Vanbroekhoven K, Lloyd JR (2013) Arsenic bioremediation by biogenic iron oxides and sulfides. Appl Environ Microbiol 79(14):4325–4335. https://doi.org/10.1128/AEM.00683-13
Oremland RS, Stolz JF (2005) Arsenic, microbes and contaminated aquifers. Trends Microbiol 13(2):45–49. https://doi.org/10.1016/j.tim.2004.12.002
Osborne TH, McArthur JM, Sikdar PK, Santini JM (2015) Isolation of an arsenate-respiring bacterium from a redox front in an arsenic-polluted aquifer in West Bengal. Bengal Basin. Environ Sci Technol 49(7):4193–4199. https://doi.org/10.1021/es504707x
Paul D, Kazy SK, Banerjee TD, Gupta AK, Pal T, Sar P (2015a) Arsenic biotransformation and release by bacteria indigenous to arsenic contaminated groundwater. Bioresource Technol 188:14–23. https://doi.org/10.1016/j.biortech.2015.02.039
Paul D, Kazy SK, Gupta AK, Pal T, Sar P (2015b) Diversity, metabolic properties and arsenic mobilization potential of indigenous bacteria in arsenic contaminated groundwater of West Bengal. India. PLoS ONE 10(3):e0118735. https://doi.org/10.1371/journal.pone.0118735
Pedersen HD, Postma D, Jakobsen R (2006) Release of arsenic associated with the reduction and transformation of iron oxides. GeochimCosmochim Ac 70(16):4116–4129. https://doi.org/10.1016/j.gca.2006.06.1370
Rahdar S, Taghavi M, Khaksefidi R, Ahmadi S (2019) Adsorption of arsenic (V) from aqueous solution using modified saxaul ash: isotherm and thermodynamic study. Appl Water Sci 9(4):87. https://doi.org/10.1007/s13201-019-0974-0
Ravenscroft P, McArthur JM, Hoque BA (2001) Geochemical and palaeohydrological controls on pollution of groundwater by arsenic. Arsenic exposure and health effects IV5. Elsevier Science Ltd., Oxford pp. 1–20
Reza AS, Jean JS, Yang HJ, Lee MK, Woodall B, Liu CC, Lee JF, Luo SD (2010) Occurrence of arsenic in core sediments and groundwater in the Chapai-Nawabganj District, northwestern Bangladesh. Water Res 44(6):2021–2037. https://doi.org/10.1016/j.watres.2009.12.006
Rhine ED, Garcia-Dominguez E, Phelps CD, Young LY (2005) Environmental microbes can speciate and cycle arsenic. Environ Sci Technol 39(24):9569–9573. https://doi.org/10.1021/es051047t
Roden EE (2006) Geochemical and microbiological controls on dissimilatory iron reduction. Comptes Rendus Geosci 338(6):456–467. https://doi.org/10.1016/j.crte.2006.04.009
Rowland HA, Pederick RL, Polya DA, Pancost RD, Van Dongen BE, Gault AG, Vaughan DJ, Bryant C, Anderson B, Lloyd JR (2007) The control of organic matter on microbially mediated iron reduction and arsenic release in shallow alluvial aquifers Cambodia. Geobiology 5(3):281–292. https://doi.org/10.1111/j.1472-4669.2007.00100.x
Sailo L, Mahanta C (2014) Arsenic mobilization in the Brahmaputra plains of Assam: groundwater and sedimentary controls. Environ Monit Assess 186(10):6805–6820. https://doi.org/10.1007/s10661-014-3890-7
Sarkar A, Kazy SK, Sar P (2013) Characterization of arsenic resistant bacteria from arsenic rich groundwater of West Bengal India. Ecotoxicology 22(2):363–376. https://doi.org/10.1007/s10646-012-1031-z
Sarkar A, Kazy SK, Sar P (2014) Studies on arsenic transforming groundwater bacteria and their role in arsenic release from subsurface sediment. Environ Sci Pollut Res 21(14):8645–8662. https://doi.org/10.1007/s11356-014-2759-1
Schmalenberger A, Duran AL, Bray AW, Bridge J, Bonneville S, Benning LG, Romero-Gonzalez ME, Leake JR, Banwart SA (2015) Oxalate secretion by ectomycorrhizal Paxillus involutus is mineral-specific and controls calcium weathering from minerals. Sci Rep-UK 5:12187. https://doi.org/10.1038/srep12187
Senn DB, Hemond HF (2002) Nitrate controls on iron and arsenic in an urban lake. Science 296(5577):2373–2376. https://doi.org/10.1126/science.1072402
Singh AK (2004) Arsenic contamination in groundwater of North Eastern India. In: Proceedings of 11th national symposium on hydrology with focal theme on water quality. National Institute of Hydrology, Roorkee, pp 255–262
Sø HU, Postma D, Jakobsen R, Larsen F (2008) Sorption and desorption of arsenate and arsenite on calcite. Geochimica et Cosmochimica Acta 72(24):5871–5884
Stillings LL, Drever JI, Brantley SL, Sun Y, Oxburgh R (1996) Rates of feldspar dissolution at pH 3–7 with 0–8 m M oxalic acid. Chem Geol 132(1–4):79–89
Straub KL, Benz M, Schink B, Widdel F (1996) Anaerobic, nitrate-dependent microbial oxidation of ferrous iron. Appl Environ Microbiol 62(4):1458–1460
Sugiyama N, Taniguchi N (1997) Evaluation of the role of lactate dehydrogenase in oxalate synthesis. Phytochemistry 44(4):571–574. https://doi.org/10.1016/S0031-9422(96)00629-2
Tessier A, Campbell PG, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51(7):844–851. https://doi.org/10.1021/ac50043a017
USEPA (1997) Exposure factors handbook. Environmental Protection Agency Office of Health and Environmental Assessment, Washington, DC
Van Geen A, Rose J, Thoral S, Garnier JM, Zheng Y, Bottero JY (2004) Decoupling of As and Fe release to Bangladesh groundwater under reducing conditions. Part II: evidence from sediment incubations. GeochimCosmochim Act 68(17):3475–3486. https://doi.org/10.1016/j.gca.2004.02.014
Verma S, Mukherjee A, Choudhury R, Mahanta C (2015) Brahmaputra river basin groundwater: solute distribution, chemical evolution and arsenic occurrences in different geomorphic settings. J Hydrol 4:131–153. https://doi.org/10.1016/j.ejrh.2015.03.001
Viollier E, Inglett PW, Hunter K, Roychoudhury AN, Van Cappellen P (2000) The ferrozine method revisited: Fe (II)/Fe (III) determination in natural waters. Appl Geochem 15(6):785–790. https://doi.org/10.1016/S0883-2927(99)00097-9
Weber KA, Achenbach LA, Coates JD (2006) Microorganisms pumping iron: anaerobic microbial iron oxidation and reduction. Nat Rev Microbiol 4(10):752–764. https://doi.org/10.1038/nrmicro1490
Yamamura S, Yamashita M, Fujimoto N, Kuroda M, Kashiwa M, Sei K, Fujita M, Ike M (2007) Bacillus selenatarsenatis sp. nov., a selenate-and arsenate-reducing bacterium isolated from the effluent drain of a glass-manufacturing plant. Int J Syst Evol Microbiol 57(5):1060–1064. https://doi.org/10.1099/ijs.0.64667-0
Yunus FM, Khan S, Chowdhury P, Milton AH, Hussain S, Rahman M (2016) A review of groundwater arsenic contamination in Bangladesh: the millennium development goal era and beyond. Int J Environ Res Public Health 13(2):215. https://doi.org/10.3390/ijerph13020215
Acknowledgements
This work was funded by Department of Biotechnology, India, North-East Region Twinning Programme (EMA) (Grant ID: 04-05-07/NO.BT/PR8933/GBD/27/41/2006). Financial support to Soma Ghosh (IIT/ACAD/PGSandR/F.112//11/BT/92P03) by IIT Kharagpur, India is duly acknowledged. The authors acknowledge Dr. Abhijit Mukherjee, Department of Geology and Geophysics, IIT Kharagpur for providing sediment samples used in the present study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghosh, S., Sar, P. Microcosm based analysis of arsenic release potential of Bacillus sp. strain IIIJ3-1 under varying redox conditions. World J Microbiol Biotechnol 36, 87 (2020). https://doi.org/10.1007/s11274-020-02860-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02860-z