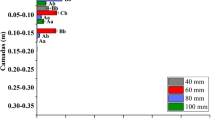
Abstract
Herbicides with long residual period may increase the risk of environmental contamination. Adequate management of forage can reduce the half-life of the picloram, one of the most herbicides used in weed control. This study aims to determine the half-life of picloram, using high-performance liquid chromatography in a cultivated soil with Brachiaria brizantha trimmed or not. Brachiaria brizantha was cultivated in 60 pots filled with samples of oxisol, and 30 others were kept uncultivated with this forage. This plant was cut off close to the ground, after 60 days of emergency on 30 vessels. Picloram was applied in all of the plots. Soil samples were collected at 2, 16, 30, 44, 58, 72, 86, 120, 150, and 180 days after the application of this herbicide. These samples were air-dried and stored at − 20 °C. Picloram was extracted by HPLC/UV-Vis detector. Half-life of this herbicide was calculated using kinetics models. The mere presence of roots in treatment with signalgrass cutoff did not reduce the concentrations of this herbicide, except when the emergence of new leaves occurred. The absence of B. brizantha cultivation in areas with application of picloram increases the risk of environmental contamination and successive crops due to the half-life of this herbicide. Brachiaria brizantha reduced half-life picloram and environmental risk in pastures. The validation method is suitable for determining picloram in low concentrations in soil.

Similar content being viewed by others
References
Assis EC, Silva AA, D’antonino L, Queiroz MELR, Barbosa LC (2011) Leaching of picloram in ultisol under different rainfall volumes. Planta Daninha 29:1129–1136
Assouline S, Or D (2014) The concept of field capacity revisited: defining intrinsic static and dynamic criteria for soil internal drainage dynamics. Water Resour Res 50:4787–4802
Bandžuchováa L, Švorcb L, Sochrb J, Svítkováb J, Chýlkováa J (2015) Sensitive voltammetric method for rapid determination of pyridine herbicide triclopyr on bare boron-doped diamond electrode. Electrochim Acta 152:421–429
Belo AF, Coelho ATCP, Tironi SP, Ferreira EA, Ferreira LR, Silva AA (2011) Photosynthetic activity of plants cultivated in soil contaminated with picloram. Planta Daninha 29:885–892
Boddey RM, Macedo R, Tarré RM, Ferreira E, Oliveira OC, Rezende CP, Cantarutti RB, Pereira JM, Alves BJR, Urquiaga S (2004) Nitrogen cycling in Brachiaria pastures: the key to understanding the process of pasture decline. Agric Ecosyst Environ 103:389–403
Cai Z, Shi S, Li S, Yang B, Chen Q, Zhao X (2013) Microbial degradation characteristics and kinetics of novel pyrimidynyloxybenzoic herbicide ZJ0273 by a newly isolated Bacillus sp. CY. Environ Sci Pollut Res 20:8831–8838
Chaparro JM, Badri DV, Bakker MG, Sugiyama A, Manter DK, Vivanco JM (2013) Root exudation of phytochemicals in Arabidopsis follows specific patterns that are developmentally programmed and correlate with soil microbial functions. PLoS One 8:e55731
Chesney P, Vasquez N (2007) Dynamics of non-structural carbohydrate reserves in pruned Erythrina poeppigiana and Gliricidia sepium trees. Agrofor Syst 69:89–105
De Lojo J, Di Benedetto A (2014) Biomass accumulation and leaf shape can be modulated by an exogenous spray of 6-benzylaminopurine in the ornamental foliage plant, Monstera deliciosa (Liebm.). J Hortic Sci Biotechnol 89:136–140
Duan Z, Homma A, Kobayashi M, Nagata N, Kaneko Y, Fujiki Y, Nishida I (2014) Photoassimilation, assimilate translocation and plasmodesmal biogenesis in the source leaves of Arabidopsis thaliana grown under an increased atmospheric CO2 concentration. Plant Cell Physiol 55:358–369
Fairchild JF, Feltz KP, Sappington LC, Allert AL, Nelson KJ, Valle J (2009) An ecological risk assessment of the acute and chronic toxicity of the herbicide picloram to the threatened bull trout (Salvelinus confluentus) and the rainbow trout (Onchorhyncus mykiss). Arch Environ Contam Toxicol 56:761–769
Farenhorst A, Andronak LA, McQueen RDA (2015) Bulk deposition of pesticides in a Canadian city: Part 1. Glyphosate and other agricultural pesticides. Water Air Soil Pollut 226:1–11
Frkova Z, Badawi N, Johansen A, Schultz-Jensen N, Bester K, Sørensen SR, Karlson UG (2014) Degradation of three benzonitrile herbicides by Aminobacter MSH1 versus soil microbial communities: pathways and kinetics. Pest Manag Sci 70:1291–1298
Ghobadi M, Yamini Y, Ebrahimpour B (2015) Extraction and determination of sulfonylurea herbicides in water and soil samples by using ultrasound-assisted surfactant-enhanced emulsification microextraction and analysis by high-performance liquid chromatography. Ecotoxicol Environ Saf 112:68–73
Huang XF, Chaparro JM, Reardon KF, Zhang R, Shen Q, Vivanco JM (2014) Rhizosphere interactions: root exudates, microbes, and microbial communities. Botany 92:267–275
Kemmerich M, Bernardi G, Adaime MB, Zanella R, Prestes OD (2015) A simple and efficient method for imidazolinone herbicides determination in soil by ultra-high performance liquid chromatography–tandem mass spectrometry. J Chromatogr A 1412:82–89
Maciel GM, Inácio FD, de Sá-Nakanishi AB, Haminiuk CWI, Castoldi R, Comar JF, Bracht A, Peralta RM (2013a) Response of Ganoderma lucidum and Trametes sp. to the herbicide picloram: tolerance, antioxidants and production of ligninolytic enzymes. Pestic Biochem Physiol 105:84–92
Maciel GM, Souza CGM, Araújo CAV, Bona E, Haminiuk CWI, Castoldi R, Bracht A, Peralta RM (2013b) Biosorption of herbicide picloram from aqueous solutions by live and heat-treated biomasses of Ganoderma lucidum (Curtis) P. karst and Trametes sp. Chem Eng J 215-216:331–338
Moraes A, de Faccio Carvalho PC, Anghinoni I, Lustosa SBC, de Andrade SEVG, Kunrath TR (2014) Integrated crop–livestock systems in the Brazilian subtropics. Eur J Agron 57:4–9
Moura GM, Costa NL (2001) Efeito da freqüência e altura de poda na produtividade de raízes e parte aérea em mandioca. Pesq Agrop Brasileira 36:1053–1059
Nascimento AF, Pires FR, Chagas K, Procópio SO, Oliveira MA, Cargnelutti Filho A, Belo AF, Egreja Filho FB (2015) Risk of soil recontamination due to using Eleusine coracana and Panicum maximum straw after phytoremediation of picloram. International Journal of Phytoremediation 17:313–321
Remini H, Mertz C, Belbahi A, Achir N, Dornier M, Madani K (2015) Degradation kinetic modelling of ascorbic acid and colour intensity in pasteurised blood orange juice during storage. Food Chem 173:665–673
Ribani M, Bottoli CBG, Collins CH, Jardim ICSF, Melo LFC (2004) Validation for chromatographic and electrophoretic methods. Química Nova 27:771–780
Sadowsky MJ, Koskinen WC, Bischoff M, Barber BL, Becker JM, Turco RF (2009) Rapid and complete degradation of the herbicide picloram by Lipomyces kononenkoae. J Agric Food Chem 57:4878–4882
Santos LBO, Infante CMC, Masini JC (2010) Determination of picloram in waters by sequential injection chromatography with UV detection. J Braz Chem Soc 21:1557–1562
Uscola M, Villar-Salvador P, Oliet J, Warren CR (2017) Root uptake of inorganic and organic N chemical forms in two coexisting Mediterranean forest trees. Plant Soil 415(1–2):387–392
Wheeler JK, Huggett BA, Tofte AN, Rockwell FE, Holbrook NM (2013) Cutting xylem under tension or supersaturated with gas can generate PLC and the appearance of rapid recovery from embolism. Plant Cell Environ 36:1938–1949
Funding
This research received a for financial support from “Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq),” “Coordenação de Aperfeiçoamento e Pessoal de Nível Superior (CAPES),” and “Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG)”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Passos, A.B.R.J., Souza, M.F., Silva, D.V. et al. Persistence of picloram in soil with different vegetation managements. Environ Sci Pollut Res 25, 23986–23991 (2018). https://doi.org/10.1007/s11356-018-2443-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2443-y