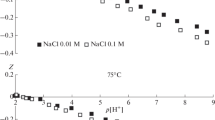
Abstract
Magnetic magnesium-zinc spinel ferrite Mg1 − xZnxFe2O4 (where x = 0.4, 0.6, and 0.8) was investigated as adsorbent for the efficient removal of Sr(II) ions and salicylic acid (SA) contaminants from aqueous medium. The characterization of ferrites was carried out using XRD, VSM, BET, SEM, and EDS. The surface charge of magnetic adsorbents was measured by the drift method. The determination of SA and Sr(II) ion concentrations in the solution phase was carried out by UFLC and complexometry, respectively. It was shown that varying of the Zn(II) content affected the adsorption capacities of magnesium-zinc ferrites. The increasing of zinc content from x(Zn2+) = 0.4 to x(Zn2+) = 0.6 increased the adsorption of Sr(II) ions from 50 to 65 mg/g, and then it was decreased to 36 mg/g for the sample with x(Zn) = 0.8. The Mg0.4Zn0.6Fe2O4 sample demonstrated the maximum adsorption capacity of 74 mg/g. The adsorption isotherm for Sr(II) was fitted by the Dubinin-Radushkevich, Langmuir, Freundlich, and Sips models. The adsorption kinetics of Sr(II) was analyzed by PFO, PSO, and Elovich models. The adsorption kinetics of SA was also investigated. It was demonstrated that the Mg0.2Zn0.8Fe2O4 sample exhibited 90% removal of salicylic acid from the water solutions. The results demonstrated that magnetic Mg-Zn ferrites with spinel structure are good sorbents for the removal of SA and Sr(II) ions from aqueous solution.









Similar content being viewed by others
References
Ainscough TJ, Alagappan P, Oatley-Radcliffe DL, Barron AR (2017) A hybrid super hydrophilic ceramic membrane and carbon nanotube adsorption process for clean water production and heavy metal removal and recovery in remote locations. J Water Process Eng 19:220–230. https://doi.org/10.1016/J.JWPE.2017.08.006
Alby D, Charnay C, Heran M, Prelot B, Zajac J (2018) Recent developments in nanostructured inorganic materials for sorption of cesium and strontium: synthesis and shaping, sorption capacity, mechanisms, and selectivity—a review. J Hazard Mater 344:511–530. https://doi.org/10.1016/J.JHAZMAT.2017.10.047
Alla SK, Kollu P, Meena SS, Poswal HK, Mandal RK, Prasad NK (2018) Mn-substituted cerium oxide nanostructures and their magnetic properties. Mater Res Bull 104:65–71. https://doi.org/10.1016/J.MATERRESBULL.2018.04.008
Arshadi M, Mousavinia F, Abdolmaleki MK, Amiri MJ, Khalafi-Nezhad A (2017) Removal of salicylic acid as an emerging contaminant by a polar nano-dendritic adsorbent from aqueous media. J Colloid Interface Sci 493:138–149. https://doi.org/10.1016/j.jcis.2017.01.017
Choodamani C, Rudraswamy B, Chandrappa GT (2016) Structural, electrical, and magnetic properties of Zn substituted magnesium ferrite. Ceram Int 42:10565–10571. https://doi.org/10.1016/J.CERAMINT.2016.03.120
Deepty M, Srinivas C, Vijaya Babu K, Ranjith Kumar E, Singh Meena S, Prajapat CL, Krisha Mohan N, Sastry DL (2018) Structural and electron spin resonance spectroscopic studies of MnxZn1−xFe2O4 (x = 0.5, 0.6, 0.7) nanoferrites synthesized by sol-gel auto combustion method. J Magn Magn Mater 466:60–68. https://doi.org/10.1016/J.JMMM.2018.06.078
Dotto GL, Cadaval TRS, Pinto LAA (2012) Preparation of bionanoparticles derived from Spirulina platensis and its application for Cr (VI) removal from aqueous solutions. J Ind Eng Chem 18:1925–1930. https://doi.org/10.1016/j.jiec.2012.05.005
Faisal AAH, Al-Wakel SFA, Assi HA et al (2020) Waterworks sludge-filter sand permeable reactive barrier for removal of toxic lead ions from contaminated groundwater. J Water Process Eng 33:101112. https://doi.org/10.1016/J.JWPE.2019.101112
Ghazi N, Mahmoudi Chenari H, Ghodsi FE (2018) Rietveld refinement, morphology analysis, optical and magnetic properties of magnesium-zinc ferrite nanofibers. J Magn Magn Mater 468:132–140. https://doi.org/10.1016/J.JMMM.2018.07.084
Ghezzi L, Spepi A, Agnolucci M, Cristani C, Giovannetti M, Tiné MR, Duce C (2018) Kinetics of release and antibacterial activity of salicylic acid loaded into halloysite nanotubes. Appl Clay Sci 160:88–94. https://doi.org/10.1016/j.clay.2017.11.041
Gore SK, Tumberphale UB, Jadhav SS, Kawale RS, Naushad M, Mane RS (2018) Microwave-assisted synthesis and magneto-electrical properties of Mg-Zn ferrimagnetic oxide nanostructures. Phys B Condens Matter 530:177–182. https://doi.org/10.1016/J.PHYSB.2017.11.044
Harris DC (2007) Quantitative chemical analysis. Seventh, New York
He Z, Meng M, Yan L, Zhu W, Sun F, Yan Y, Liu Y, Liu S (2015) Fabrication of new cellulose acetate blend imprinted membrane assisted with ionic liquid ([BMIM]Cl) for selective adsorption of salicylic acid from industrial wastewater. Sep Purif Technol 145:63–74. https://doi.org/10.1016/j.seppur.2015.03.005
Hu X, Liu B, Liu J, Qin J, Zhao W, Lam KH (2019) Construction of zinc-magnesium-iron multinary spinel core-shell microspheres with enhanced photocatalytic properties of 1, 2-dichlorobenzene toxic species. J Photochem Photobiol A Chem 382:111903. https://doi.org/10.1016/J.JPHOTOCHEM.2019.111903
Hua P, Sellaoui L, Franco D, Netto MS, Luiz Dotto G, Bajahzar A, Belmabrouk H, Bonilla-Petriciolet A, Li Z (2020) Adsorption of acid green and procion red on a magnetic geopolymer based adsorbent: experiments, characterization and theoretical treatment. Chem Eng J 383:123113. https://doi.org/10.1016/J.CEJ.2019.123113
John SP, Mathew J (2019) Determination of ferromagnetic, superparamagnetic and paramagnetic components of magnetization and the effect of magnesium substitution on structural, magnetic and hyperfine properties of zinc ferrite nanoparticles. J Magn Magn Mater 475:160–170. https://doi.org/10.1016/J.JMMM.2018.11.030
Karunanayake AG, Todd OA, Crowley ML, et al (2017) Rapid removal of salicylic acid, 4-nitroaniline, benzoic acid and phthalic acid from wastewater using magnetized fast pyrolysis biochar from waste Douglas fir. Chem Eng J 319:75. https://doi.org/10.1016/j.cej.2017.02.116.
Kefeni KK, Mamba BB, Msagati TAM (2017) Application of spinel ferrite nanoparticles in water and wastewater treatment: a review. Sep Purif Technol 188:399–422. https://doi.org/10.1016/J.SEPPUR.2017.07.015
Kothawale MM, Tangsali RB, Meena SS, Prasad NK, Gangwar A (2019) Mössbauer study and curie temperature configuration on sintering Nano-Ni-Zn ferrite powder. J Supercond Nov Magn 32:2141–2147. https://doi.org/10.1007/s10948-018-4935-x
Kumar TR, Prabukanthan P, Harichandran G, Theerthagiri J, Moydeen AM, Durai G, Kuppusami P, Tatarchuk T (2018) Comparative study of structural, optical and electrical properties of electrochemically deposited Eu, Sm and Gd doped ZnSe thin films. J Mater Sci Mater Electron 29:5638–5648. https://doi.org/10.1007/s10854-018-8533-2
Li WC (2014) Occurrence, sources, and fate of pharmaceuticals in aquatic environment and soil. Environ Pollut 187:193–201. https://doi.org/10.1016/J.ENVPOL.2014.01.015
Liu Y, Wei S, Tian H, Tong H, Xu B (2014) Characterization of soft magnetic spinel ferrite coating prepared by plasma spray. Surf Coat Technol 258:189–199. https://doi.org/10.1016/J.SURFCOAT.2014.09.029
Liu H, Li A, Ding X, Yang F, Sun K (2019) Magnetic induction heating properties of Mg1-xZnxFe2O4 ferrites synthesized by co-precipitation method. Solid State Sci 93:101–108. https://doi.org/10.1016/J.SOLIDSTATESCIENCES.2019.05.005
Marques JL, Lütke SF, Frantz TS et al (2018) Removal of Al (III) and Fe (III) from binary system and industrial effluent using chitosan films. Int J Biol Macromol 120:1667–1673. https://doi.org/10.1016/j.ijbiomac.2018.09.135
Martín J, del MM O, Medina-Carrasco S et al (2018) Removal of priority and emerging pollutants from aqueous media by adsorption onto synthetic organo-funtionalized high-charge swelling micas. Environ Res 164:488–494. https://doi.org/10.1016/j.envres.2018.03.037
Mironyuk I, Tatarchuk T, Naushad M, Vasylyeva H, Mykytyn I (2019a) Highly efficient adsorption of strontium ions by carbonated mesoporous TiO2. J Mol Liq 285:742–753. https://doi.org/10.1016/J.MOLLIQ.2019.04.111
Mironyuk I, Tatarchuk T, Vasylyeva H, Naushad M, Mykytyn I (2019b) Adsorption of Sr(II) cations onto phosphated mesoporous titanium dioxide: mechanism, isotherm and kinetics studies. J Environ Chem Eng 7:103430. https://doi.org/10.1016/j.jece.2019.103430
Mohamed EA, Selim AQ, Ahmed SA, Sellaoui L, Bonilla-Petriciolet A, Erto A, Li Z, Li Y, Seliem MK (2020) H2O2-activated anthracite impregnated with chitosan as a novel composite for Cr(VI) and methyl orange adsorption in single-compound and binary systems: modeling and mechanism interpretation. Chem Eng J 380:122445. https://doi.org/10.1016/J.CEJ.2019.122445
Mohanraj J, Durgalakshmi D, Balakumar S, Aruna P, Ganesan S, Rajendran S, Naushad M (2019) Low cost and quick time absorption of organic dye pollutants under ambient condition using partially exfoliated graphite J Water Process Eng 101078. https://doi.org/10.1016/J.JWPE.2019.101078
Ng LY, Ng CY, Mahmoudi E, Ong CB, Mohammad AW (2018) A review of the management of inflow water, wastewater and water reuse by membrane technology for a sustainable production in shrimp farming. J Water Process Eng 23:27–44. https://doi.org/10.1016/J.JWPE.2018.02.020
Prabukanthan P, Lakshmi R, Harichandran G, Tatarchuk T (2018) Photovoltaic device performance of pure, manganese (Mn2+) doped and irradiated CuInSe2 thin films. New J Chem 42:11642–11652. https://doi.org/10.1039/c8nj01056k
Rajesh Kumar T, Prabukanthan P, Harichandran G, Theerthagiri J, Tatarchuk T, Maiyalagan T, Maia G, Bououdina M (2018) Physicochemical and electrochemical properties of Gd3+−doped ZnSe thin films fabricated by single-step electrochemical deposition process. J Solid State Electrochem 22:1197–1207. https://doi.org/10.1007/s10008-017-3865-z
Reddy DHK, Yun Y-S (2016) Spinel ferrite magnetic adsorbents: alternative future materials for water purification? Coord Chem Rev 315:90–111. https://doi.org/10.1016/J.CCR.2016.01.012
Reyes-Rodríguez PY, Cortés-Hernández DA, Escobedo-Bocardo JC, Almanza-Robles JM, Sánchez-Fuentes HJ, Jasso-Terán A, de León-Prado LE, Méndez-Nonell J, Hurtado-López GF (2017) Structural and magnetic properties of Mg-Zn ferrites (Mg1−xZnxFe2O4) prepared by sol-gel method. J Magn Magn Mater 427:268–271. https://doi.org/10.1016/J.JMMM.2016.10.078
Rodriguez-Narvaez OM, Peralta-Hernandez JM, Goonetilleke A, Bandala ER (2017) Treatment technologies for emerging contaminants in water: a review. Chem Eng J 323:361–380. https://doi.org/10.1016/J.CEJ.2017.04.106
Satheeshkumar MK, Ranjith Kumar E, Srinivas C, Prasad G, Meena SS, Pradeep I, Suriyanarayanan N, Sastry DL (2019) Structural and magnetic properties of CuFe2O4 ferrite nanoparticles synthesized by cow urine assisted combustion method. J Magn Magn Mater 484:120–125. https://doi.org/10.1016/J.JMMM.2019.03.128
Sharma R, Thakur P, Kumar M, Thakur N, Negi NS, Sharma P, Sharma V (2016) Improvement in magnetic behaviour of cobalt doped magnesium zinc nano-ferrites via co-precipitation route. J Alloys Compd 684:569–581. https://doi.org/10.1016/J.JALLCOM.2016.05.200
Sharma R, Thakur P, Sharma P, Sharma V (2017) Ferrimagnetic Ni2+ doped Mg-Zn spinel ferrite nanoparticles for high density information storage. J Alloys Compd 704:7–17. https://doi.org/10.1016/J.JALLCOM.2017.02.021
Sophia AC, Lima EC (2018) Removal of emerging contaminants from the environment by adsorption. Ecotoxicol Environ Saf 150:1–17. https://doi.org/10.1016/J.ECOENV.2017.12.026
Tatarchuk T, Bououdina M, Judith Vijaya J, John Kennedy L (2017) Spinel ferrite nanoparticles: synthesis, crystal structure, properties, and perspective applications. In: Fesenko O, Yatsenko L (eds) Nanophysics, nanomaterials, interface studies, and applications. NANO 2016. Springer Proceedings in Physics. Springer, Cham, pp 305–325
Tatarchuk T, Al-Najar B, Bououdina M, Ahmed MAA (2019a) Catalytic and photocatalytic properties of oxide spinels. In: Martínez L, Kharissova O, Kharisov B (eds) Handbook of ecomaterials. Pp 1701–1750
Tatarchuk T, Bououdina M, Al-Najar B, Bitra RB (2019b) Green and ecofriendly materials for the remediation of inorganic and organic pollutants in water. In: Naushad M (ed) A new generation material graphene: applications in water technology. Springer, Cham, pp 69–110
Tatarchuk T, Myslin M, Mironyuk I, et al (2019c) Synthesis, morphology, crystallite size and adsorption properties of nanostructured Mg–Zn ferrites with enhanced porous structure. J Alloys Compd 152945. https://doi.org/10.1016/J.JALLCOM.2019.152945
Tatarchuk T, Paliychuk N, Bitra RB et al (2019d) Adsorptive removal of toxic Methylene blue and Acid Orange 7 dyes from aqueous medium using cobalt-zinc ferrite nanoadsorbents. Desalin Water Treat 150:374–385. https://doi.org/10.5004/dwt.2019.23751
Tatarchuk T, Shyichuk A, Mironyuk I, Naushad M (2019e) A review on removal of uranium(VI) ions using titanium dioxide based sorbents. J Mol Liq 293:111563. https://doi.org/10.1016/j.molliq.2019.111563
Theerthagiri J, Durai G, Tatarchuk T, Sumathi M, Kuppusami P, Qin J, Choi MY (2019) Synthesis of hierarchical structured rare earth metal–doped Co3O4 by polymer combustion method for high performance electrochemical supercapacitor electrode materials. Ionics (Kiel) 26:2051–2061. https://doi.org/10.1007/s11581-019-03330-9
Tran HN, You S-J, Hosseini-Bandegharaei A, Chao H-P (2017) Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: a critical review. Water Res 120:88–116. https://doi.org/10.1016/J.WATRES.2017.04.014
Tsay C-Y, Chiu Y-C, Tseng Y-K (2019) Investigation on structural, magnetic, and FMR properties for hydrothermally-synthesized magnesium-zinc ferrite nanoparticles. Phys B Condens Matter 570:29–34. https://doi.org/10.1016/J.PHYSB.2019.05.037
Xiao G, Wen R, Liu A, He G, Wu D (2017) Adsorption performance of salicylic acid on a novel resin with distinctive double pore structure. J Hazard Mater 329:77–83. https://doi.org/10.1016/j.jhazmat.2017.01.030
Zaki HM, Al-Heniti SH, Elmosalami TA (2015) Structural, magnetic and dielectric studies of copper substituted nano-crystalline spinel magnesium zinc ferrite. J Alloys Compd 633:104–114. https://doi.org/10.1016/J.JALLCOM.2015.01.304
Zhang L, Sellaoui L, Franco D, Dotto GL, Bajahzar A, Belmabrouk H, Bonilla-Petriciolet A, Oliveira MLS, Li Z (2020) Adsorption of dyes brilliant blue, sunset yellow and tartrazine from aqueous solution on chitosan: analytical interpretation via multilayer statistical physics model. Chem Eng J 382:122952. https://doi.org/10.1016/J.CEJ.2019.122952
Funding
The Ministry of Education and Science of Ukraine (project number 0118U000258) financially supported this research work. One of the authors (Mu. Naushad) is grateful to the Researchers Supporting Project number RSP-2019/8, King Saud University, Riyadh, Saudi Arabia for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tatarchuk, T., Naushad, M., Tomaszewska, J. et al. Adsorption of Sr(II) ions and salicylic acid onto magnetic magnesium-zinc ferrites: isotherms and kinetic studies. Environ Sci Pollut Res 27, 26681–26693 (2020). https://doi.org/10.1007/s11356-020-09043-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09043-1