Abstract
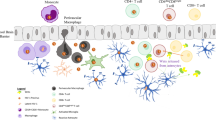
Human immunodeficiency virus (HIV)-associated neurocognitive disorder (HAND) is one of the common causes of cognitive dysfunction and morbidity among infected patients. However, to date, it remains unknown if a transmitted/founder (T/F) HIV-1 leads to neurological disorders during acute phase of infection. Since it is impossible to answer this question in humans, we studied NOD.Cg-Prkdc scid Il2rgtm1Wjl/SzJ mice (NSG) reconstituted with human PBMC (NSG-HuPBL), followed by the peritoneal challenge with the chronic HIV-1JR-FL and the T/F HIV-1BJZS7, respectively. By measuring viral load, P24 antigenemia and P24+ cells in peripheral blood and various tissue compartments, we found that systemic infections were rapidly established in NSG-HuPBL mice by both HIV-1 strains. Although comparable peripheral viral loads were detected during acute infection, the T/F virus appeared to cause less CD4+ T cell loss and less numbers of infected cells in different organs and tissue compartments. Both viruses, however, invaded brains with P24+/CD3+ T cells detected primarily in meninges, cerebral cortex and perivascular areas. Critically, brain infections with HIV-1JR-FL but not with HIV-1BJZS7 resulted in damaged neurons together with activated microgliosis and astrocytosis as determined by significantly increased numbers of Iba1+ microglial cells and GFAP+ astrocytes, respectively. The increased Iba1+ microglia was correlated positively with levels of P24 antigenemia and negatively with numbers of NeuN+ neurons in brains of infected animals. Our findings, therefore, indicate the establishment of two useful NSG-HuPBL models, which may facilitate future investigation of mechanisms underlying HIV-1-induced microgliosis and astrocytosis.






Similar content being viewed by others
References
Baenziger S et al. (2006) Disseminated and sustained HIV infection in CD34+ cord blood cell-transplanted Rag2−/−gamma c−/− mice. Proc Natl Acad Sci U S A 103:15951–15956. doi:10.1073/pnas.0604493103
Balazs AB, Chen J, Hong CM, Rao DS, Yang L, Baltimore D (2012) Antibody-based protection against hiv infection by vectored immunoprophylaxis. Nature 481:81–84. doi:10.1038/nature10660
Balazs AB et al. (2014) Vectored immunoprophylaxis protects humanized mice from mucosal HIV transmission. Nat Med 20:296–300. doi:10.1038/nm.3471
Banks WA, Kumar VB, Franko MW, Bess JW Jr, Arthur LO (2005) Evidence that the species barrier of human immunodeficiency virus-1 does not extend to uptake by the blood–brain barrier: comparison of mouse and human brain microvessels. Life Sci 77:2361–2368. doi:10.1016/j.lfs.2004.11.041
Bobardt MD et al. (2004) Contribution of proteoglycans to human immunodeficiency virus type 1 brain invasion. J Virol 78:6567–6584. doi:10.1128/JVI.78.12.6567-6584.2004
Budka H et al. (1991) HIV-associated disease of the nervous system: review of nomenclature and proposal for neuropathology-based terminology. Brain Pathol 1:143–152. doi:10.1111/j.1750-3639.1991.tb00653.x
Chen Z, Zhou P, Ho DD, Landau NR, Marx PA (1997) Genetically divergent strains of simian immunodeficiency virus use CCR5 as a coreceptor for entry. J Virol 71:2705–2714
Chen Y et al. (2015) Comprehensive characterization of the transmitted/founder env genes from a single MSM cohort in China. J Acquir Immune Defic Syndr 69:403–412 doi:10.1097/QAI.0000000000000649
Dash PK et al. (2011) Loss of neuronal integrity during progressive HIV-1 infection of humanized mice. J Neurosci 31:3148–3157. doi:10.1523/JNEUROSCI.5473-10.2011
Dash PK et al. (2012) Long-acting nanoformulated antiretroviral therapy elicits potent antiretroviral and neuroprotective responses in HIV-1-infected humanized mice. AIDS 26:2135–2144. doi:10.1097/QAD.0b013e328357f5ad
Fiala M et al. (1997) TNF-alpha opens a paracellular route for HIV-1 invasion across the blood-brain barrier. Mol Med 3:553–564
Fischer-Smith T et al. (2001) CNS invasion by CD14+/CD16+ peripheral blood-derived monocytes in HIV dementia: perivascular accumulation and reservoir of HIV infection. J Neurovirol 7:528–541. doi:10.1080/135502801753248114
Gorantla S et al. (2007) Copolymer-1 induces adaptive immune anti-inflammatory glial and neuroprotective responses in a murine model of HIV-1 encephalitis. J Immunol 179:4345–4356
Gorantla S et al. (2010) Links between progressive hiv-1 infection of humanized mice and viral neuropathogenesis. Am J Pathol 177:2938–2949. doi:10.2353/ajpath.2010.100536
Gorantla S, Poluektova L, Gendelman HE (2012) Rodent models for hiv-associated neurocognitive disorders. Trends Neurosci 35:197–208. doi:10.1016/j.tins.2011.12.006
Hadas E, Borjabad A, Chao W, Saini M, Ichiyama K, Potash MJ, Volsky DJ (2007) Testing antiretroviral drug efficacy in conventional mice infected with chimeric HIV-1. AIDS 21:905–909. doi:10.1097/Qad.0b013e3281574549
Harbison C et al. (2014) Giant cell encephalitis and microglial infection with mucosally transmitted simian-human immunodeficiency virus SHIVSF162P3N in rhesus macaques. J Neurovirol 20:62–72. doi:10.1007/s13365-013-0229-z
Huang J, Li X, Coelho-dos-Reis JG, Wilson JM, Tsuji M (2014) An AAV vector-mediated gene delivery approach facilitates reconstitution of functional human CD8+ T cells in mice. PLoS One 9:e88205. doi:10.1371/journal.pone.0088205
Joseph SB, Swanstrom R, Kashuba AD, Cohen MS (2015) Bottlenecks in HIV-1 transmission: insights from the study of founder viruses. Nat Rev Microbiol 13:414–425. doi:10.1038/nrmicro3471
Kang Y et al. (2012) CCR5 antagonist TD-0680 uses a novel mechanism for enhanced potency against HIV-1 entry, cell-mediated infection, and a resistant variant. Journal Biol Chem 287:16499–16509. doi:10.1074/jbc.M112.354084
Keele BF et al. (2008) Identification and characterisation of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc Natl Acad Sci U S A 105:7552–7557. doi:10.1073/pnas.0802203105
Kumar P et al. (2008) T cell-specific siRNA delivery suppresses HIV-1 infection in humanized mice. Cell 134:577–586. doi:10.1016/j.cell.2008.06.034
Liu J, Gong N, Huang X, Reynolds AD, Mosley RL, Gendelman HE (2009) Neuromodulatory activities of CD4 + CD25+ regulatory T cells in a murine model of HIV-1-associated neurodegeneration. J Immunol 182:3855–3865. doi:10.4049/jimmunol.0803330
Long BR, Stoddart CA (2012) Alpha interferon and HIV infection cause activation of human T cells in NSG-BLT mice. J Virol 86:3327–3336. doi:10.1128/JVI.06676-11
Maartens G, Celum C, Lewin SR (2014a) HIV infection: epidemiology, pathogenesis, treatment, and prevention. Lancet 384:258–271. doi:10.1016/S0140-6736(14)60164-1
Matinella A et al. (2015) Neurological complications of HIV infection in pre-HAART and HAART era: a retrospective study. J Neurol 262:1317–1327. doi:10.1007/s00415-015-7713-8
McMichael AJ, Borrow P, Tomaras GD, Goonetilleke N, Haynes BF (2010) The immune response during acute HIV-1 infection: clues for vaccine development. Nat Rev Immunol 10:11–23. doi:10.1038/nri2674
Minagar A, Shapshak P, Fujimura R, Ownby R, Heyes M, Eisdorfer C (2002) The role of macrophage/microglia and astrocytes in the pathogenesis of three neurologic disorders: HIV-associated dementia, Alzheimer disease, and multiple sclerosis. J Neurol Sci 202:13–23
Nakata H et al. (2005) Potent anti-R5 human immunodeficiency virus type 1 effects of a CCR5 antagonist, AK602/ONO4128/GW873140, in a novel human peripheral blood mononuclear cell nonobese diabetic-SCID, interleukin-2 receptor gamma-chain-knocked-out AIDS mouse model J Virol 79:2087–2096 doi:10.1128/JVI.79.4.2087-2096.2005
Paris JJ, Singh HD, Carey AN, McLaughlin JP (2015) Exposure to HIV-1 Tat in brain impairs sensorimotor gating and activates microglia in limbic and extralimbic brain regions of male mice. Behav Brain Res 291:209–218. doi:10.1016/j.bbr.2015.05.021
Peluso MJ et al. (2013) Cerebrospinal fluid and neuroimaging biomarker abnormalities suggest early neurological injury in a subset of individuals during primary HIV infection. J Infect Dis 207:1703–1712. doi:10.1093/infdis/jit088
Persidsky Y et al. (1996) Human immunodeficiency virus encephalitis in SCID mice. Am J Pathol 149:1027–1053
Podhaizer EM, Zou S, Fitting S, Samano KL, El-Hage N, Knapp PE, Hauser KF (2012) Morphine and gp120 toxic interactions in striatal neurons are dependent on HIV-1 strain. J NeuroImmune Pharmacol 7:877–891. doi:10.1007/s11481-011-9326-z
Potash MJ et al. (2005) A mouse model for study of systemic HIV-1 infection, antiviral immune responses, and neuroinvasiveness. Proc Natl Acad Sci U S A 102:3760–3765. doi:10.1073/pnas.0500649102
Ragin AB et al. (2015) Brain alterations within the first 100 days of HIV infection. Ann Clin Transl Neurol 2:12–21. doi:10.1002/acn3.136
Roshorm Y et al. (2009) Novel HIV-1 clade B candidate vaccines designed for HLA-B*5101(+) patients protected mice against chimaeric ecotropic HIV-1 challenge. Eur J Immunol 39:1831–1840. doi:10.1002/eji.200939309
Rumbaugh JA, Tyor W (2015) HIV-associated neurocognitive disorders: five new things. Neurol Clin Pract 5:224–231. doi:10.1212/CPJ.0000000000000117
Sailasuta N et al. (2012) Change in brain magnetic resonance spectroscopy after treatment during acute HIV infection. PLoS One 7:e49272. doi:10.1371/journal.pone.0049272
Sas AR, Bimonte-Nelson H, Smothers CT, Woodward J, Tyor WR (2009) Interferon-alpha causes neuronal dysfunction in encephalitis. J Neurosci 29:3948–3955. doi:10.1523/JNEUROSCI.5595-08.2009
Singh M et al. (2012) An improved protocol for efficient engraftment in NOD/LTSZ-SCIDIL-2Rgammanull mice allows HIV replication and development of anti-HIV immune responses. PLoS One 7:e38491. doi:10.1371/journal.pone.0038491
Stoddart CA et al. (2011) Superior human leukocyte reconstitution and susceptibility to vaginal HIV transmission in humanized NOD-scid IL-2Rgamma(−/−) (NSG) BLT mice. Virology 417:154–160. doi:10.1016/j.virol.2011.05.013
Tyor WR, Power C, Gendelman HE, Markham RB (1993) A model of human-immunodeficiency-virus encephalitis in scid mice. Proc Natl Acad Sci U S A 90:8658–8662. doi:10.1073/pnas.90.18.8658
Valcour V et al. (2012) Central nervous system viral invasion and inflammation during acute HIV infection. J Infect Dis 206:275–282. doi:10.1093/infdis/jis326
Watanabe S et al. (2007) Hematopoietic stem cell-engrafted NOD/SCID/IL2Rgamma null mice develop human lymphoid systems and induce long-lasting HIV-1 infection with specific humoral immune responses. Blood 109:212–218. doi:10.1182/blood-2006-04-017681
Watkins CC, Treisman GJ (2015) Cognitive impairment in patients with AIDS - prevalence and severity. Hiv/Aids 7:35–47. doi:10.2147/HIV.S39665
Zhang L, Meissner E, Chen J, Su L (2010) Current humanized mouse models for studying human immunology and HIV-1 immuno-pathogenesis. Sci China Life Sci 53:195–203. doi:10.1007/s11427-010-0059-7
Zhuang K et al. (2014) Emergence of CD4 independence envelopes and astrocyte infection in R5 simian-human immunodeficiency virus model of encephalitis. J Virol 88:8407–8420. doi:10.1128/Jvi.01237-14
Zou W, Kim BO, Zhou BY, Liu Y, Messing A, He JJ (2007) Protection against human immunodeficiency virus type 1 Tat neurotoxicity by Ginkgo biloba extract EGb 761 involving glial fibrillary acidic protein. Am J Pathol 171:1923–1935. doi:10.2353/ajpath.2007.070333
Acknowledgments
The authors would like to thank Hong Kong Council for the AIDS Trust Fund (MSS 227R) for financial support on neuroAIDS research. We also thank Hong Kong Research Grant Council HKU5/CRF/13G to study T cell mechanism and China’s 12th 5-year National Science and Technology Mega Project 2013ZX10001005002001 as well as the University Development Fund of the University of Hong Kong and Li Ka Shing Faculty of Medicine Matching Fund to HKU AIDS Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors of this manuscript do not report conflict of interest.
Rights and permissions
About this article
Cite this article
Wu, X., Liu, L., Cheung, Kw. et al. Brain Invasion by CD4+ T Cells Infected with a Transmitted/Founder HIV-1BJZS7 During Acute Stage in Humanized Mice. J Neuroimmune Pharmacol 11, 572–583 (2016). https://doi.org/10.1007/s11481-016-9654-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-016-9654-0