Summary
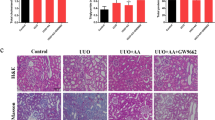
Renal tubulointerstitial fibrosis is the common ending of progreβsive renal disease. It is worth developing new ways to stop the progreβs of renal fibrosis. Peroxisome proliferator-activated receptor-γ (PPARγ) agonists have been studied to treat diabetic nephropathy, cisplatin-induced acute renal injury, ischemia reperfusion injury and adriamycin nephropathy. In this study, unilateral ureteral obstruction (UUO) was used to establish a different renal fibrosis model. PPAR? agonist pioglitazone was administrated by oral gavage and saline was used as control. At 7th and 14th day after the operation, mice were sacrificed for fibrosis test and T lymphocytes subsets test. Unexpectedly, through MASSON staining, immunohistochemistry for α-SMA, and Western blotting for a-SMA and PDGFR-β, we found that pioglitazone failed to attenuate renal fibrosis in UUO mice. However, flow cytometry showed that pioglitazone down-regulated Th1 cells, and up-regulated Th2 cells, Th17 cells and Treg cells. But the Th17/Treg ratio had no significant change by pioglitazone. Real-time PCR results showed that TGF-β and MCP-1 had no significant changes, at the same time, CD4+ T cells associated cytokines were partially regulated by pioglitazone pretreatment. Taken together, pioglitazone failed to suppress renal fibrosis progression caused by UUO.
Similar content being viewed by others
References
Zeisberg M, Neilson EG. Mechanisms of tubulointerstitial fibrosis. J Am Soc Nephrol, 2010,21(11):1819–1834
Nangaku M. Chronic hypoxia and tubulointerstitial injury: a final common pathway to end-stage renal failure. J Am Soc Nephrol, 2006,17(1):17–25
Chung AC, Lan HY. Chemokines in renal injury. J Am Soc Nephrol, 2011,22(5):802–809
Piccotti JR, Li K, Chan SY, et al. Cytokine regulation of chronic cardiac allograft rejection: evidence against a role for Th1 in the disease process. Transplantation, 1999,67(12):1548–1555
Kim SM, Lee SH, Lee A, et al. Targeting T helper 17 by mycophenolate mofetil attenuates diabetic nephropathy progression. Transpl Res, 2015
Sakai N, Wada T. T Helper 2 cytokine signaling in bone marrow-derived fibroblasts: A target for renal fibrosis. J Am Soc Nephrol, 2015,26(12):2896–2898
Jun C, Ke W, Qingshu L, et al. Protective effect of CD4(+)CD25(high)CD127(low) regulatory T cells in renal ischemia-reperfusion injury. Cell Immunol, 2014,289(1–2):106–111
Tapmeier TT, Fearn A, Brown K, et al. Pivotal role of CD4+ T cells in renal fibrosis following ureteric obstruction. Kidney Int, 2010,78(4):351–362
Liu L, Kou P, Zeng Q, et al. CD4+ T lymphocytes, especially Th2 cells, contribute to the progress of renal fibrosis. Am J Nephrol, 2012,36(4):386–396
Lin FJ, Jiang GR, Shan JP, et al. Imbalance of regulatory T cells to Th17 cells in IgA nephropathy. Scand J Clin Lab Invest, 2012,72(3):221–229
Wenbin Z, Guojun G. Resveratrol ameliorates diabetes-induced renal damage through regulating the expression of TGF-beta1, collagen IV and Th17/Treg-related cytokines in rats. West Indian Med J, 2014,63(1):20–25
Li Y, Shi Y, Huang Z, et al. CNI induced Th17/Treg imbalance and susceptibility to renal dysfunction in renal transplantation. Int Immunopharmacol, 2011,11 (12): 2033–2038
Cariou B, Charbonnel B, Staels B. Thiazolidinediones and PPARgamma agonists: time for a reassessment. Trends Endocrinol Metab, 2012,23(5):205–215
Lee YJ, Han HJ. Troglitazone ameliorates high glucose-induced EMT and dysfunction of SGLTs through PI3K/Akt, GSK-3beta, Snail1, and beta-catenin in renal proximal tubule cells. Am J Physiol Renal Physiol, 2010,298(5):F1263–F1275
Pistrosch F, Passauer J, Herbrig K, et al. Effect of thiazolidinedione treatment on proteinuria and renal hemodynamic in type 2 diabetic patients with overt nephropathy. Horm Metab Res, 2012,44(12):914–918
Huang KC, Cherng YG, Chen LJ, et al. Rosiglitazone is effective to improve renal damage in type-1-like diabetic rats. Horm Metab Res, 2014,46(4):240–244
Agarwal R, Saha C, Battiwala M, et al. A pilot randomized controlled trial of renal protection with pioglitazone in diabetic nephropathy. Kidey Int, 2005,68(1):285–292
Schneider CA, Ferrannini E, Defronzo R, et al. Effect of pioglitazone on cardiovascular outcome in diabetes and chronic kidney disease. J Am Soc Nephrol, 2008,19(1): 182–187
Yang HC, Deleuze S, Zuo Y, et al. The PPARgamma agonist pioglitazone ameliorates aging-related progressive renal injury. J Am Soc Nephrol, 2009,20(11): 2380–2388
Jesse CR, Bortolatto CF, Wilhelm EA, et al. The peroxisome proliferator-activated receptor-gamma agonist pioglitazone protects against cisplatin-induced renal damage in mice. J Appl Toxicol, 2014,34(1):25–32
Hu H, Zou C, Xi X, et al. Protective effects of pioglitazone on renal ischemia-reperfusion injury in mice. J Surg Res, 2012,178(1):460–465
Higashi K, Oda T, Kushiyama T, et al. Additive antifibrotic effects of pioglitazone and candesartan on experimental renal fibrosis in mice. Nephrology (Carlton), 2010,15(3):327–335
Han JY, Kim YJ, Kim L, et al. PPARgamma agonist and angiotensin II receptor antagonist ameliorate renal tubulointerstitial fibrosis. J Korean Med Sci, 2010,25(1):35–41
Orasanu G, Ziouzenkova O, Devchand PR, et al. The peroxisome proliferator-activated receptor-gamma agonist pioglitazone represses inflammation in a peroxisome proliferator-activated receptor-alpha-dependent manner in vitro and in vivo in mice. J Am Coll Cardiol, 2008,52(10):869–881
Martens FM, Rabelink TJ, Op TRJ, et al. TNF-alpha induces endothelial dysfunction in diabetic adults, an effect reversible by the PPAR-gamma agonist pioglitazone. Eur Heart J, 2006,27(13):1605–1609
Lea S, Plumb J, Metcalfe H, et al. The effect of peroxisome proliferator-activated receptor-gamma ligands on in vitro and in vivo models of COPD. Eur Respir J, 2014,43(2):409–420
Cipolletta D, Feuerer M, Li A, et al. PPAR-gamma is a major driver of the accumulation and phenotype of adipose tissue Treg cells. Nature, 2012,486(7404):549–553
Bunt SK, Mohr AM, Bailey JM, et al. Rosiglitazone and Gemcitabine in combination reduces immune suppression and modulates T cell populations in pancreatic cancer. Cancer Immunol Immunother, 2013,62(2):225–236
Schaefer KL, Denevich S, Ma C, et al. Intestinal antiinflammatory effects of thiazolidenedione peroxisome proliferator-activated receptor-gamma ligands on T helper type 1 chemokine regulation include nontranscriptional control mechanisms. Inflamm Bowel Dis, 2005,11(3):244–252
Ochodnicky P, Mesarosova L, Cernecka H, et al. Pioglitazone, a PPARgamma agonist, provides comparable protection to angiotensin converting enzyme inhibitor ramipril against adriamycin nephropathy in rat. Eur J Pharmacol, 2014,730:51–60
Shappell S, Gurpinar T, Lechago J, et al. Chronic obstructive uropathy in severe combined immunodeficient (SCID) mice: lymphocyte infiltration is not required for progressive tubulointerstitial injury. J Am Soc Nephrol, 1998,9(6):1008–1017
Cheng P, Zhang Y, Huang H, et al. Association between CCR6 and rheumatoid arthritis: a meta-analysis. Int J Clin Exp Med, 2015,8(4):5388–5396
Cheng P, Sun XY, Yin DL, et al. Nanog down-regulates the Wnt signaling pathway via β-catenin phosphorylation during epidermal stem cell proliferation and differentiation. Cell Biosci, 2015,5:5
Henri S, Chevillard C, Mergani A, et al. Cytokine regulation of periportal fibrosis in humans infected with Schistosoma mansoni: IFN-gamma is associated with protection against fibrosis and TNF-alpha with aggravation of disease. J Immunol, 2002,169(2):929–936
Saito A, Okazaki H, Sugawara I, et al. Potential action of IL-4 and IL-13 as fibrogenic factors on lung fibroblasts in vitro. Int Arch Allergy Immunol, 2003,132(2): 168–176
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was financially supported by grants from the National Natural Science Foundation of China (No. 81470948, No. 81270770, and No. 81300575), and Hubei Provincial Health and Family Planning Youth Project of China (No. WJ2015Q007).
Rights and permissions
About this article
Cite this article
Zhang, Y., Wang, J., Zhou, Qd. et al. Peroxisome proliferator-activated receptor-γ agonist pioglitazone fails to attenuate renal fibrosis caused by unilateral ureteral obstruction in mice. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 41–47 (2016). https://doi.org/10.1007/s11596-016-1539-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1539-1