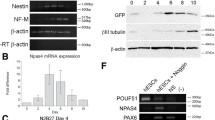
Abstract
Neuropathy target esterase (NTE) is involved in several disorders in adult organisms and embryos. A relationship between NTE and nervous system integrity and maintenance in adult systems has been suggested. NTE-related motor neuron disease is associated with the expression of a mutant form of NTE and the inhibition and further modification of NTE by organophosphorus compounds is the trigger of a delayed neurodegenerative neuropathy. Homozygotic NTE knockout mice embryos are not viable, while heterozygotic NTE knockout mice embryos yields mice with neurological disorders, which suggest that this protein plays a critical role in embryonic development. The present study used D3 mouse embryonic stem cells with the aim of gaining mechanistic insights on the role of Pnpla6 (NTE gene encoding) in the developmental process. D3 cells were silenced by lipofectamine transfection with a specific interference RNA for Pnpla6. Silencing Pnpla6 in D3 monolayer cultures reduced NTE enzymatic activity to 50% 20 h post-treatment, while the maximum loss of Pnpla6 expression reached 80% 48 h postsilencing. Pnpla6 was silenced in embryoid bodies and 545 genes were differentially expressed regarding the control 96 h after silencing, which revealed alterations in multiple genetic pathways, such as cell motion and cell migration, vesicle regulation, and cell adhesion. These findings also allow considering that these altered pathways would impair the formation of respiratory, neural, and vascular tubes causing the deficiencies observed in the in vivo development of nervous and vascular systems. Our findings, therefore, support the previous observations made in vivo concerning lack of viability of mice embryos not expressing NTE and help to understand the biology of several neurological and developmental disorders in which NTE is involved.







Similar content being viewed by others
Notes
Throughout the manuscript, the term NTE is always used for calling the protein called neuropathy target esterase; the term NTE enzymatic activity is used for calling the phenyl valerate esterase activity associated to NTE; while the term Pnpla6 is used for calling the NTE-codifying gene.
References
Akassoglou K.; Malester B.; Xu J. L.; Tessarollo L.; Rosenbluth J.; Chao M. V. Brain-specific deletion of neuropathy target esterase/Swiss cheese results in neurodegeneration. Proc. Natl. Acad. Sci. U. S. A. 101: 5075–5080; 2004.
Bautch V. L.; James J. M. Neurovascular development: the beginning of a beautiful friendship. Cell Adhes. Migr. 3: 199–204; 2009.
Birgbauer E.; Chun J. New developments in the biological functions of lysophospholipids. Cell. Mol. Life Sci. 63: 2695–2701; 2006.
Burnstock G. Purinergic signaling and vascular cell proliferation and death. Arterioscler. Thromb. Vasc. Biol. 22: 364–373; 2002.
Camon E.; Barrell D.; Lee V.; Dimmer E.; Apweiler R. The Gene Ontology Annotation (GOA) database—an integrated resource of GO annotations to the UniProt Knowledgebase. In Silico Biol. 4: 5–6; 2004.
Chang P. A.; Chen R.; Wu Y. J. Reduction of neuropathy target esterase does not affect neuronal differentiation, but moderate expression induces neuronal differentiation in human neuroblastoma (SK-N-SH) cell line. Brain Res. Mol. Brain Res. 141: 30–38; 2005.
Chang P. A.; Sun Q.; Ni X. M.; Qv F. Q.; Wu Y. J.; Song F. Z. Molecular cloning and expression analysis of cDNA ends of chicken neuropathy target esterase. Chem. Biol. Interact. 172: 54–62; 2008.
Chomczynski P.; Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162: 156–159; 1987.
Dechant G. A.; Rodríguez-Tébar A.; Barde Y. A. Neurotrophin receptors. Prog. Neurobiol. 42: 249–254; 1994.
Ferrri G.; Cook B. D.; Terushkin V.; Pintucci G.; Mignatti P. Transforming growth factor-beta 1 (TGF-beta 1) induces angiogenesis through vascular endothelial growth factor (VEGF)-mediated apoptosis. J. Cell. Physiol. 219: 449–458; 2009.
Glynn P. NTE: one target protein for different toxic syndromes with distinct mechanisms. Bioessays 25: 742–745; 2003.
Greiner A. J.; Richardson R. J.; Worden R. M.; Ofoli R. Y. Influence of lysophospholipid hydrolysis by the catalytic domain of neuropathy target esterase on the fluidity of bilayer lipid membranes. Biochim. Biophys. Acta 1798: 1533–1539; 2010.
Hein N. D.; Stuckey J. A.; Rainier S. R.; Fink J. K.; Richardson R. J. Constructs of human neuropathy target esterase catalytic domain containing mutations related to motor neuron disease have altered enzymatic properties. Toxicol. Lett. 196: 67–73; 2010.
Hong M. S.; Hong S. J.; Barhoumi R.; Burghardt R. C.; Donnelly K. C.; Wild J. R.; Venkatraj V.; Tiffany-Castiglioni E. Neurotoxicity induced in differentiated SK-N-SH-SY5Y human neuroblastoma cells by organophosphorus compounds. Toxicol. Appl. Pharmacol. 186: 110–118; 2003.
Howard A. S.; Bucelli R.; Jett D. A.; Bruun D.; Yang D.; Lein P. J. Chlorpyrifos exerts opposing effects on axonal and dendritic growth in primary neuronal cultures. Toxicol. Appl. Pharmacol. 207: 112–124; 2005.
Ip N. Y.; Yancopoulos G. D. Neurotrophic factors and their receptors. Ann. Neurol. 35(Suppl): S13–S16; 1994.
Johnson M. K. The delayed neurotoxic effect of some organophosphorus compounds: identification of the phosphorylation site as an esterase. Biochem. J. 114: 711–717; 1969.
Jones F. S.; Jones P. L. The tenascin family of ECM glycoproteins: structure, function, and regulation during embryonic development and tissue remodeling. Dev. Dyn. 218: 235–259; 2000.
Kretzschmar D.; Hasan G.; Sharma S.; Heisenberg M.; Benzer S. The swiss cheese mutant causes glial hyperwrapping and brain degeneration in Drosophila. J. Neurosci. 17: 7425–7432; 1997.
Li W.; Casida J. E. Organophosphorus neuropathy target esterase inhibitors selectively block outgrowth of neurite-like and cell processes in cultured cells. Toxicol. Lett. 98: 139–146; 1998.
Li Z.; Szurek P. F.; Jiang C.; Pao A.; Bundy B.; Le W. D.; Bradley A.; Yu Y. E. Neuronal differentiation of NTE-deficient embryonic stem cells. Biochem. Biophys. Res. Commun. 30: 1103–1109; 2005.
Lim Y.; Matsui W. Hedgehog signaling in hematopoiesis. Crit. Rev. Eukaryot. Gene Expr. 20: 129–139; 2010.
Livak K. J.; Schmittgen T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25: 402–408; 2001.
Lubarsky B.; Krasnow M. A. Tube morphogenesis: making and shaping biological tubes. Cell 112: 19–28; 2003.
Lush M. J.; Li Y.; Read D. J.; Willis A. C.; Glynn P. Neuropathy target esterase and a homologous Drosophila neurodegeneration-associated mutant protein contain a novel domain conserved from bacteria to man. Biochem. J. 332: 1–4; 1998.
Massicotte C.; Jortner B. S.; Ehrich M. Morphological effects of neuropathy-inducing organophosphorus compounds in primary dorsal root ganglia cell cultures. Neurotoxicology 24: 787–796; 2003.
Moser M.; Li Y.; Vaupel K.; Kretzschmar D.; Kluge R.; Glynn P.; Buettner R. Placental failure and impaired vasculogenesis result in embryonic lethality for neuropathy target esterase-deficient mice. Mol. Cell. Biol. 24: 1667–1679; 2004.
Mühlig-Versen M.; da Cruz A. B.; Tschäpe J. A.; Moser M.; Büttner R.; Athenstaedt K.; Glynn P.; Kretzschmar D. Loss of Swiss cheese/neuropathy target esterase activity causes disruption of phosphatidylcholine homeostasis and neuronal and glial death in adult Drosophila. J. Neurosci. 25: 2865–2873; 2005.
Nakao T.; Ishizawa A.; Ogawa R. Observations of vascularization in the spinal cord of mouse embryos, with special reference to development of boundary membranes and perivascular spaces. Anat. Rec. 221: 663–677; 1988.
Pamies D.; Reig J. A.; Vilanova E.; Sogorb M. A. Expression of neuropathy target esterase in mouse embryonic stem cells during differentiation. Arch. Toxicol. 84: 481–491; 2010.
Quistad G. B.; Barlow C.; Winrow C. J.; Sparks S. E.; Casida J. E. Evidence that mouse brain neuropathy target esterase is a lysophospholipase. Proc. Natl. Acad. Sci. U. S. A. 100: 7983–7987; 2003.
Rainier S.; Albers J. W.; Dyck P. J.; Eldevik O. P.; Wilcock S.; Richardson R. J.; Fink J. K. Motor neuron disease due to neuropathy target esterase gene mutation: clinical features of the index families. Muscle Nerve 43: 19–25; 2011.
Read D. J.; Li Y.; Chao M. V.; Cavanagh J. B.; Glynn P. Neuropathy target esterase is required for adult vertebrate axon maintenance. J. Neurosci. 29: 11594–11600; 2009.
Read D. J.; Li Y.; Chao M. V.; Cavanagh J. B.; Glynn P. Organophosphates induce distal axonal damage, but not brain oedema, by inactivating neuropathy target esterase. Toxicol. Appl. Pharmacol. 245: 108–115; 2010.
Ross B. M.; Kish S. J. Characterization of lysophospholipid metabolizing enzymes in human brain. J. Neurochem. 63: 1839–1848; 1994.
Sachana M.; Flaskos J.; Alexaki E.; Hargreaves A. J. Inhibition of neurite outgrowth in N2a cells by leptophos and carbaryl: effects on neurofilament heavy chain, GAP-43 and HSP-70. Toxicol. In Vitro 17: 115–120; 2003.
Sanford L. P.; Ormsby I.; Gittenberger-de Groot A. C.; Sariola H.; Friedman R.; Boivin G. P.; Cardell E. L.; Doetschman T. TGFbeta2 knockout mice have multiple developmental defects that are non-overlapping with other TGFbeta knockout phenotypes. Development 124: 2659–2670; 1997.
Schmidt A.; Wolde M.; Thiele C.; Fest W.; Kratzin H.; Podtelejnikov A. V.; Witke W.; Huttner W. B.; Söling H. D. Endophilin I mediates synaptic vesicle formation by transfer of arachidonate to lysophosphatidic acid. Nature 401: 133–141; 1999.
Simon M. C.; Keith B. The role of oxygen availability in embryonic development and stem cell function. Nat. Rev. Mol. Cell Biol. 9: 285–296; 2008.
Slotkin T.; Seidler F. Transcriptional profiles reveal similarities and differences in the effects of developmental neurotoxicants on differentiation into neurotransmitter phenotypes in PC12 cells. Brain Res. Bull. 78: 211–225; 2009.
Smyth G.K.; Ritchie M.; Thorne N.; Wettenhall J. Limma: Linear models for microarray data user’s guide. Available: http://www.lcg.unam.mx/~lcollado/R/resources/limma-usersguide.pdf. 2007. Accessed February 2013.
Song F.; Zou C.; Han X.; Zeng T.; Zhang C.; Xie K. Reduction of retrograde axonal transport associated-proteins motor proteins, dynein and dynactin in the spinal cord and cerebral cortex of hens by tri-ortho-cresyl phosphate (TOCP). Neurochem. Int. 60: 99–104; 2012.
Song Y.; Wang M.; Mao F.; Shao M.; Zhao B.; Song Z.; Shao C.; Gong Y. Knockdown of Pnpla6 protein results in motor neuron defects in zebrafish. Dis. Model Mech. 6: 404–413; 2013.
Thorsteinsdóttir S.; Deries M.; Cachaço A. S.; Bajanca F. The extracellular matrix dimension of skeletal muscle development. Dev. Biol. 354: 191–207; 2011.
Vose S. C.; Fujioka K.; Gulevich A. G.; Lin A. Y.; Holland N. T.; Casida J. E. Cellular function of neuropathy target esterase in lysophosphatidylcholine action. Toxicol. Appl. Pharmacol. 232: 376–383; 2008.
Wang A.; Dennis E. A. Mammalian lysophospholipases. Biochim. Biophys. Acta 1439: 1–16; 1999.
Williams D. G. Intramolecular group transfer is a characteristic of neurotoxic esterase and is independent of the tissue source of the enzyme—a comparison of the aging behavior of diisopropyl phosphorofluoridate-labeled proteins in brain, spinal-cord, liver, kidney and spleen from hen and in human-placenta. Biochem. J. 209: 817–829; 1983.
Wilson P. A.; Gardner S. D.; Lambie N. M.; Commans S. A.; Crowther D. J. Characterization of the human patatin-like phospholipase family. J. Lipid Res. 47: 1940–1949; 2006.
Winrow C. J.; Hemming M. L.; Allen D. M.; Quistad G. B.; Casida J. E.; Barlow C. Loss of neuropathy target esterase in mice links organophosphate exposure to hyperactivity. Nat. Genet. 33: 477–485; 2003.
Acknowledgments
The authors would like to express gratitude to Drs. Luis Alcaraz and Maria Isabel Carreres (Bioarrays SL; www.bioarray.es) for their help and support in the analysis the data obtained from the microarrays.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
PDF 35.5 kb
ESM 2
Gene Set Analysis Heat Map. Heat map represents the overexpressed genes (red) and the underexpressed genes (green) in the D3 EBs cultured after Pnpla6 silencing (JPEG 33 kb)
ESM 3
List of the 545 genes with an altered expression at 96 h after Pnpla6 silencing in the D3 embryoid bodies (PDF 77.6 kb)
ESM 4
(PDF 23.6 kb)
ESM 5
(PDF 24.2 kb)
ESM 6
(JPEG 655 kb)
ESM 7
Relationships among genes involved in nervous system development and vasculogenesis. The picture shows the relationships among the 96 genes with altered expression 96 h after transfection of D3 embryoid bodies with iRNA for silencing Pnpla6. The picture was obtained using Ingenuity Pathways Analysis database (PDF 14.7 kb)
Rights and permissions
About this article
Cite this article
Pamies, D., Vilanova, E. & Sogorb, M.A. Functional pathways altered after silencing Pnpla6 (the codifying gene of neuropathy target esterase) in mouse embryonic stem cells under differentiation. In Vitro Cell.Dev.Biol.-Animal 50, 261–273 (2014). https://doi.org/10.1007/s11626-013-9691-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-013-9691-4