Abstract
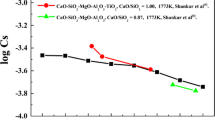
The molar sulphide capacities \( C_{\text{S}}^{'} \) = (mol pct S) (\( P_{{{\text{O}}_{2} }} /P_{{{\text{S}}_{2} }} \))1/2 on four binary systems, MgO-SiO2, CaO-SiO2, MnO-SiO2 and FeO-SiO2 are elucidated so as to compare the magnitudes of the basicities of four metallic oxides and to estimate the temperature dependencies of the basicities of metallic oxides. The enthalpy changes of the reaction 2O− = O + O2−, viz. the silicate polymerization reaction (denoted as \( \Updelta H_{(8)}^{^\circ } \)) have been calculated from the slopes of the log \( C_{\text{S}}^{'} \) vs 1/T curves for four binary silicates. The \( \Updelta H_{(8)}^{^\circ } \) value is considered in the present work to be an index of the basicity of silicate melts. The basicities obtained on the basis of the \( \Updelta H_{(8)}^{^\circ } \) values are in the order MgO < CaO < MnO < FeO, which are determined by two effects; (i) ionicity of chemical bonds between metallic and oxygen ions and (ii) clustering of metallic oxides in silicates. It is also found that the basicity of the FeO-SiO2 system is larger at higher temperatures.


Similar content being viewed by others
References
C.J.B. Fincham and F.D. Richardson: Proc. Roy. Soc. (Lond.), 1954, vol. A223, pp. 40–62.
C.J.B. Fincham and F.D. Richardson: J. Iron Steel Inst., 1954, vol. 178, pp. 4–14.
P.T. Carter and T.G. Macfarlane: J. Iron Steel Inst., 1957, vol. 185, pp. 54–62.
K.P. Abraham and F.D. Richardson: J. Iron. Steel Inst., 1960, vol. 196, pp. 313–17.
M. Görnerup and O. Wijk: Scand. J. Metall., 1996, vol. 25, pp. 103–07.
M. Hino, S. Kitagawa, and S. Ban-ya: ISIJ Int., 1993, vol. 33, pp. 36–42.
R.A. Sharma and F.D. Richardson: J. Iron Steel Inst., 1961, vol. 198, pp. 386–90.
G.J.W. Kor and F.D. Richardson: J. Iron Steel Inst., 1968, vol. 206, pp. 700–04.
M.M. Nzotta, S. Du, and S. Seetharaman: ISIJ Int., 1998, vol. 38, no. 11, pp. 1170–79.
M.M. Nzotta, S. Du, and S. Seetharaman: ISIJ Int., 1999, vol. 39, no. 7, pp. 657–63.
M.M. Nzotta, S. Du, and S. Seetharaman: Metall. Mater. Trans. B, 1999, vol. 30B, pp. 909–20.
A. Shankar, M. Gornerup, A.K. Lahiri, and S. Seetharaman: Metall. Mater. Trans. A, 2006, vol. 37B, pp. 941–47.
R. Nilsson and S. Seetharaman: Scand. J. Metall., 1994, vol. 23, pp. 81–86.
S.R. Simeonov, R. Sridhar, and J.M. Toguri: Metall. Trans. B., 1995, vol. 26B, pp. 325–34.
M.M. Nzotta, S. Du, and S. Seetharaman: ISIJ Int., 1999, vol. 39, pp. 657–63.
J.A. Duffy and M.D. Ingram: J. Non-Cryst. Solids, 1976, vol. 21, pp. 373–410.
I.D. Sommerville and D.J. Sosinsky: Proc. 2nd International Symposium on Metallurgical Slags and Fluxes, 1984, p. 1015.
L.S. Darken and R.W. Gurry: J. Am. chem. Soc., 1946, vol. 67, pp. 1398–1412.
Slag Atlas, 2nd ed., Verein Deutscher Eisenhuttenleute, Verlag Stahleisen GmbH, Düsseldorf, 1995, p. 11.
F.D. Richardson: Physical Chemistry of Melts in Metallurgy, Academic Press, London, 1974, pp. 85–88.
G.W. Toop and C.S. Samis: Canad. Met. Quart., 1962, vol. 1, pp. 129–52.
G.W. Toop and C.S. Samis: Trans. Metall. Soc. AIME, 1962, vol. 224, pp. 878–87.
C.R. Masson, J.R. Smith, and S.G. Whiteway: Can. J. Chem., 1970, vol. 48, pp. 1456–64.
C.R. Masson: Glass 1977, Proceedings ofthe 11th International Congress on Glass, J. Gotz, ed., Prague 1977, vol. 1, p. 3.
Acknowledgments
The authors are thankful to the Swedish Steel Producers Association and National Natural Science Foundation of China (No. 51104013), China Postdoctoral Science special Foundation (No. 201004056) for the financial support in carrying out this work. The authors are also thankful to Dr. Lidong Teng for valuable discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted October 20, 2011.
Rights and permissions
About this article
Cite this article
Wang, Lj., Hayashi, M., Chou, Kc. et al. An Insight into Slag Structure from Sulphide Capacities. Metall Mater Trans B 43, 1338–1343 (2012). https://doi.org/10.1007/s11663-012-9713-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-012-9713-8