Abstract
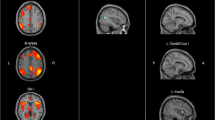
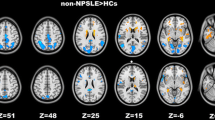
Using resting-state functional magnetic resonance imaging and graph theory approaches to investigate the topological characteristics of functional networks and their potential correlations with clinical information in patients with systemic lupus erythematosus (SLE). A total of 41 patients and 35 volunteers were consecutively recruited. Detailed clinical data of all participants were recorded. All participants underwent a resting-state functional magnetic resonance imaging examination. Functional networks were constructed by a Pearson correlation matrix of 116 brain regions. The topological properties were analyzed by graph theory. Parametric tests were used to compare the topological properties between the groups. Partial correlation analysis was used to identify relationships between the abnormal topological properties and the clinical data. The nodal network metrics were abnormal in the SLE patients compared to the controls. Decreased nodal efficiency was identified in the right insula, bilateral putamen, and bilateral Heschl’s gyrus in the SLE patients. Decreased degree centrality was also found in the right amygdala and bilateral Heschl’s gyrus. In addition, the SLE patients showed decreased network functional connectivity (FC) between several regions, particularly between the basal ganglia and the cerebellum. Moreover, FC values between the right putamen and vermis 6 were positively correlated with Mini-Mental State Examination scores. The nodal efficiency and the degree centrality values in the left Heschl’s gyrus were both positively correlated with the course of the disease. The topological structure of the functional network was apparently abnormal in SLE patients. FC values between the right putamen and vermis 6 may serve as a neuroimaging marker for evaluating the progressive cognitive decline in SLE patients. Decreased synergy between the basal ganglia region and the cerebellum in the extrapyramidal system may be one cause of cognitive dysfunction in SLE patients.




Similar content being viewed by others
Abbreviations
- FC:
-
functional connectivity
- FDR:
-
False Discovery Rate
- fMRI:
-
functional magnetic resonance imaging
- HC:
-
healthy control
- MMSE:
-
Mini-Mental State Examination
- NAA:
-
N-acetylaspartate
- Gamma:
-
normalized clustering coefficient
- Lambda:
-
normalized characteristic path length
- NPSLE:
-
neuropsychiatric systemic lupus erythematosus
- SLE:
-
Systemic lupus erythematosus
- Sigma:
-
small-worldness
References
O'Neill, S., & Cervera, R. (2010). Systemic lupus erythematosus[J]. Best Practice & Research. Clinical Rheumatology, 24(6), 841–855. https://doi.org/10.1016/j.berh.2010.10.006.
Wigren, M., Svenungsson, E., Mattisson, I. Y., Gustafsson, J. T., Gunnarsson, I., Zickert, A., Elvin, K., Jensen-Urstad, K., Bengtsson, A., Gullstrand, B., Fredrikson, G. N., & Nilsson, J. (2018). Cardiovascular disease in systemic lupus erythematosus is associated with increased levels of biomarkers reflecting receptor-activated apoptosis[J]. Atherosclerosis, 270, 1–7. https://doi.org/10.1016/j.atherosclerosis.2018.01.022.
Tselios, K., Gladman, D. D., Su, J., Ace, O., & Urowitz, M. B. (2017). Evolution of risk factors for atherosclerotic cardiovascular events in systemic lupus Erythematosus: A Longterm prospective study[J]. The Journal of Rheumatology, 44(12), 1841–1849. https://doi.org/10.3899/jrheum.161121.
Piga, M., Chessa, E., Peltz, M. T., Floris, A., Mathieu, A., & Cauli, A. (2017). Demyelinating syndrome in SLE encompasses different subtypes: Do we need new classification criteria? Pooled results from systematic literature review and monocentric cohort analysis[J]. Autoimmunity Reviews, 16(3), 244–252. https://doi.org/10.1016/j.autrev.2017.01.011.
Meszaros, Z. S., Perl, A., & Faraone, S. V. (2012). Psychiatric symptoms in systemic lupus erythematosus: A systematic review[J]. The Journal of Clinical Psychiatry, 73(7), 993–1001. https://doi.org/10.4088/JCP.11r07425.
Filley, C. M., Kozora, E., Brown, M. S., Miller, D. E., West, S. G., Arciniegas, D. B., Grimm, A., & Zhang, L. (2009). White matter microstructure and cognition in non-neuropsychiatric systemic lupus erythematosus[J]. Cognitive and Behavioral Neurology, 22(1), 38–44. https://doi.org/10.1097/WNN.0b013e318190d174.
Popescu, A., & Kao, A. H. (2011). Neuropsychiatric systemic lupus erythematosus[J]. Current Neuropharmacology, 9(3), 449–457. https://doi.org/10.2174/157015911796557984.
Niu, C., Tan, X., Liu, X., Han, K., Niu, M., Xu, J., Zhao, L., Wu, Y., Deng, F., Huang, Q., Wu, X., Huang, H., Xu, Y., & Huang, R. (2018). Cortical thickness reductions associate with abnormal resting-state functional connectivity in non-neuropsychiatric systemic lupus erythematosus[J]. Brain Imaging and Behavior, 12(3), 674–684. https://doi.org/10.1007/s11682-017-9729-4.
Hou, J., Lin, Y., Zhang, W., Song, L., Wu, W., Wang, J., Zhou, D., Zou, Q., Fang, Y., He, M., & Li, H. (2013). Abnormalities of frontal-parietal resting-state functional connectivity are related to disease activity in patients with systemic lupus erythematosus[J]. PLoS One, 8(9), e74530. https://doi.org/10.1371/journal.pone.0074530.
Shapira-Lichter, I., Weinstein, M., Lustgarten, N., Ash, E., Litinsky, I., Aloush, V., Anouk, M., Caspi, D., Hendler, T., & Paran, D. (2016). Impaired diffusion tensor imaging findings in the corpus callosum and cingulum may underlie impaired learning and memory abilities in systemic lupus erythematosus[J]. Lupus, 25(11), 1200–1208. https://doi.org/10.1177/0961203316636471.
Sporns, O., Tononi, G., & Kotter, R. (2005). The human connectome: A structural description of the human brain[J]. PLoS Computational Biology, 1(4), e42. https://doi.org/10.1371/journal.pcbi.0010042.
Liao, X., Vasilakos, A. V., & He, Y. (2017). Small-world human brain networks: Perspectives and challenges[J]. Neuroscience and Biobehavioral Reviews, 77, 286–300. https://doi.org/10.1016/j.neubiorev.2017.03.018.
Xu, M., Tan, X., Zhang, X., Guo, Y., Mei, Y., Feng, Q., Xu, Y., & Feng, Y. (2017). Alterations of white matter structural networks in patients with non-neuropsychiatric systemic lupus erythematosus identified by probabilistic tractography and connectivity-based analyses[J]. Neuroimage Clin, 13, 349–360. https://doi.org/10.1016/j.nicl.2016.12.021.
Wiseman, S. J., Bastin, M. E., Amft, E. N., Belch, J. F. F., Ralston, S. H., & Wardlaw, J. M. (2018). Cognitive function, disease burden and the structural connectome in systemic lupus erythematosus[J]. Lupus, 27(8), 1329–1337. https://doi.org/10.1177/0961203318772666.
Hochberg, M. C. (1997). Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus[J]. Arthritis and Rheumatism, 40(9), 1725. https://doi.org/10.1002/1529-0131(199709)40:9<1725::AID-ART29>3.0.CO;2-Y.
Calhoun, V. D., Wager, T. D., Krishnan, A., Rosch, K. S., Seymour, K. E., Nebel, M. B., Mostofsky, S. H., Nyalakanai, P., & Kiehl, K. (2017). The impact of T1 versus EPI spatial normalization templates for fMRI data analyses[J]. Human Brain Mapping, 38(11), 5331–5342. https://doi.org/10.1002/hbm.23737.
Yang, G. J., Murray, J. D., Repovs, G., Cole, M. W., Savic, A., Glasser, M. F., Pittenger, C., Krystal, J. H., Wang, X. J., Pearlson, G. D., Glahn, D. C., & Anticevic, A. (2014). Altered global brain signal in schizophrenia[J]. PNAS, 111(20), 7438–7443. https://doi.org/10.1073/pnas.1405289111.
Liao, W., Zhang, Z., Mantini, D., Xu, Q., Wang, Z., Chen, G., Jiao, Q., Zang, Y. F., & Lu, G. (2013). Relationship between large-scale functional and structural covariance networks in idiopathic generalized epilepsy[J]. Brain Connectivity, 3(3), 240–254. https://doi.org/10.1089/brain.2012.0132.
Zhang, J., Wang, J., Wu, Q., Kuang, W., Huang, X., He, Y., & Gong, Q. (2011a). Disrupted brain connectivity networks in drug-naive, first-episode major depressive disorder[J]. Biological Psychiatry, 70(4), 334–342. https://doi.org/10.1016/j.biopsych.2011.05.018.
Zhang, Z., Liao, W., Chen, H., Mantini, D., Ding, J. R., Xu, Q., Wang, Z., Yuan, C., Chen, G., Jiao, Q., & Lu, G. (2011b). Altered functional-structural coupling of large-scale brain networks in idiopathic generalized epilepsy[J]. Brain, 134(Pt 10), 2912–2928. https://doi.org/10.1093/brain/awr223.
Harris, R. E., Sundgren, P. C., Pang, Y., Hsu, M., Petrou, M., Kim, S. H., McLean, S., Gracely, R. H., & Clauw, D. J. (2008). Dynamic levels of glutamate within the insula are associated with improvements in multiple pain domains in fibromyalgia[J]. Arthritis and Rheumatism, 58(3), 903–907. https://doi.org/10.1002/art.23223.
Cagnoli, P., Harris, R. E., Frechtling, D., Berkis, G., Gracley, R. H., Graft, C. C., Lowe, S. E., Chenevert, T. L., McCune, W., Gebarski, S., & Sundgren, P. C. (2013). Reduced insular glutamine and N-acetylaspartate in systemic lupus erythematosus: A single-voxel (1)H-MR spectroscopy study[J]. Academic Radiology, 20(10), 1286–1296. https://doi.org/10.1016/j.acra.2013.07.011.
Sundgren, P. C., Jennings, J., Attwood, J. T., Nan, B., Gebarski, S., McCune, W., Pang, Y., & Maly, P. (2005). MRI and 2D-CSI MR spectroscopy of the brain in the evaluation of patients with acute onset of neuropsychiatric systemic lupus erythematosus[J]. Neuroradiology, 47(8), 576–585. https://doi.org/10.1007/s00234-005-1371-y.
Watson, P., Storbeck, J., Mattis, P., & Mackay, M. (2012). Cognitive and emotional abnormalities in systemic lupus erythematosus: Evidence for amygdala dysfunction[J]. Neuropsychology Review, 22(3), 252–270. https://doi.org/10.1007/s11065-012-9213-2.
Zimmermann, N., Correa, D. G., Kubo, T. A., et al. (2017). Global cognitive impairment in systemic lupus Erythematosus patients: A structural MRI study[J]. Clinical Neuroradiology, 27(1), 23–29. https://doi.org/10.1007/s00062-015-0397-8.
Zhang, H. Y., Tang, H., Chen, W. X., Ji, G. J., Ye, J., Wang, N., Wu, J. T., & Guan, B. (2015). Mapping the functional connectivity of the substantia nigra, red nucleus and dentate nucleus: A network analysis hypothesis associated with the extrapyramidal system[J]. Neuroscience Letters, 606, 36–41. https://doi.org/10.1016/j.neulet.2015.08.029.
de Oliveira-Souza, R., & Tovar-Moll, F. (2012). The unbearable lightness of the extrapyramidal system[J]. Journal of the History of the Neurosciences, 21(3), 280–292. https://doi.org/10.1080/0964704X.2011.595652.
Vinas-Guasch, N., & Wu, Y. J. (2017). The role of the putamen in language: A meta-analytic connectivity modeling study[J]. Brain Structure & Function, 222(9), 3991–4004. https://doi.org/10.1007/s00429-017-1450-y.
Kozora, E., Filley, C. M., Erkan, D., Uluğ, A. M., Vo, A., Ramon, G., Burleson, A., Zimmerman, R. D., & Lockshin, M. D. (2018). Longitudinal evaluation of diffusion tensor imaging and cognition in systemic lupus erythematosus[J]. Lupus, 27(11), 1810–1818. https://doi.org/10.1177/0961203318793215.
Jung, R. E., Segall, J. M., Grazioplene, R. G., Qualls, C., Sibbitt, W. L., & Roldan, C. A. (2010). Cortical thickness and subcortical gray matter reductions in neuropsychiatric systemic lupus erythematosus[J]. PLoS One, 5(3), e9302. https://doi.org/10.1371/journal.pone.0009302.
Gottwald, B., Wilde, B., Mihajlovic, Z., & Mehdorn, H. M. (2004). Evidence for distinct cognitive deficits after focal cerebellar lesions[J]. Journal of Neurology, Neurosurgery, and Psychiatry, 75(11), 1524–1531. https://doi.org/10.1136/jnnp.2003.018093.
Habas, C. (2010). Functional imaging of the deep cerebellar nuclei: A review[J]. Cerebellum, 9(1), 22–28. https://doi.org/10.1007/s12311-009-0119-3.
Bengtsson, S. L., Ehrsson, H. H., Forssberg, H., & Ullén, F. (2004). Dissociating brain regions controlling the temporal and ordinal structure of learned movement sequences[J]. The European Journal of Neuroscience, 19(9), 2591–2602. https://doi.org/10.1111/j.0953-816X.2004.03269.x.
Kirschen, M. P., Davis-Ratner, M. S., Milner, M. W., Chen, S. H., Schraedley-Desmond, P., Fisher, P. G., & Desmond, J. E. (2008). Verbal memory impairments in children after cerebellar tumor resection[J]. Behavioural Neurology, 20(1–2), 39–53. https://doi.org/10.3233/BEN-2008-0216.
Laywer, G., Nyman, H., Agartz, I., Arnborg, S., Jönsson, E. G., Sedvall, G. C., & Hall, H. (2006). Morphological correlates to cognitive dysfunction in schizophrenia as studied with Bayesian regression[J]. BMC Psychiatry, 6, 31. https://doi.org/10.1186/1471-244X-6-31.
Duan, D., Shen, L., Li, C., Cui, C., Shu, T., & Zheng, J. (2017). Memory impairment in occipital periventricular hyperintensity patients is associated with reduced functional responses in the insula and Heschl's gyrus[J]. The International Journal of Neuroscience, 127(6), 493–500. https://doi.org/10.1080/00207454.2016.1198345.
Funding
This study has received funding by National Natural Science Foundation of China (NSFC81571652); Science and technology project of Yangzhou (YZ2018059); “333 Project” of Jiangsu Province (BRA2017154). Yangzhou City Science and Education Strengthening Key Talents Project (ZDRC201873).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All the procedures involving human subjects were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
We confirm that informed consent was obtained from all the participants included in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 140 kb)
Rights and permissions
About this article
Cite this article
Cao, ZY., Wang, N., Jia, JT. et al. Abnormal topological organization in systemic lupus erythematosus: a resting-state functional magnetic resonance imaging analysis. Brain Imaging and Behavior 15, 14–24 (2021). https://doi.org/10.1007/s11682-019-00228-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00228-y