Abstract
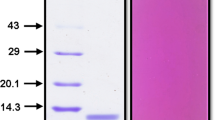
A trypsin inhibitor from seeds of faba bean (Vicia faba L.) was purified to near homogeneity as judged by native-PAGE with about 11 % recovery using ammonium sulphate fractionation, ion-exchange chromatography on DEAE-cellulose and gel filtration through Sephadex G-100. The inhibitor had a molecular weight of 18 kD as determined by SDS-PAGE and Sephadex G-100. The inhibitor inhibited trypsin and chymotrypsin to the extent of 48 and 12 %, respectively. The inhibtion was of non-competitive type with dissociation constant for the enzyme inhibitor complex in the region of 0.07 mg·ml−1. The inhibtor was stable between pH 4 and 5. It completely lost its activity when heated at 125 °C for 1 h or at 100 °C for 2 h. The inhibitor also lost its activity on exposure to 2-mercaptoethanol. Based on these properties, it could be concluded that Vicia faba trypsin inhibitor belongs to Bowman-Birk type of inhibitors, as it has molecular weight lower than generally observed for Kunitz type inhibitors.
Similar content being viewed by others
Abbreviations
- PAGE:
-
polyacrylamide gel electrophoresis
- DEAE:
-
diethyl amino ethyl
- SDS:
-
sodium dodecyl sulphate
- kD:
-
kilodalton
- CM:
-
carboxy methyl
References
Annapurna S.S, C.S. Ramadoss and D.S. Prasad, 1991. Characterization of trypsin/chymotrypsin inhibitor from Jack fruit (Artocarpus integrifolia) seeds. J. Sci. Food Agric. 54: 605–618.
Asao T., F. Imai, I. Tsuji, M. Tashiro, K. Iwani and F. Ibuki, 1991b. Purification and characterization of a Bowman-Birk type proteinase inhibitor from faba bean (Vicia faba L.). Agric. Biol. Chem. 55(3): 707–713.
Bergeron D. and S.S. Nielsen, 1993. Partial characterization of trypsin inhibitors and N-terminal sequences of five trypsin isoinhibitors of Great Northern beans (Phaseolus vulgaris). J. Agric. Food Chem. 41: 1544–1549.
Davis B.J., 1964. Disc electrophoresis. II. method and application to human serum proteins. Ann. N. Y. Acad. Sci. 121: 404–427.
Ferrasson E., L. Quillien and J. Gueguen, 1997. Proteinase inhibitors from pea seeds: Purification and characterization. J. Agric Food Chem. 45: 127–131.
Godbole S.A, T.G. Krishna and C.R. Bhatia, 1994a. Purification and characterization of protease inhibitors from pigeonpea (Cajanus cajan L.) seeds. J. Sci. Food Agric. 64: 87–93.
Godbole S.A, T.G. Krishna and C.R. Bhatia, 1994b. Further characterization of protease inhibitors from pigeonpea (Cajanus cajan L.) seeds. J. Sci. Food Agric. 64: 331–335.
Gumbmann M.R., W.L. Spangler, G.M. Dugan and J.J. Rackis, 1986. Safety of trypsin inhibitors in the diet: effect on the rat pancreas of long term feeding on soy flour and soy protein isolate. In: Nutritional and Toxicological Significance of Enzyme Inhibitors in Foods (M. Friedman, ed.) pp. 33–79 Plenum Press. New York.
Hilder V.A., A.M.R. Gatehouse, S.E. Sheerman, R.F. Barker and D. Boulter, 1987. A novel mechanism of insect resistance engineered into tobacco. Nature, 330: 160–163.
Jouanin L., M.B. Bottino, C. Girard, G. Morrot and M. Giband, 1998. Transgenic plants for insect resistance. Plant Sci. 131: 1–11.
Kakade M.L., N. Simons and I.E. Liener, 1969. An evaluation of natural vs synthetic substrates for measuring the antitryptic activity of soybean samples. Cereal Chem. 46: 518–526.
Laemmli U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Liener I.E. and M.L. Kakade, 1980. Protease inhibitors. In: The Toxic Constituents of Plant Food Stuffs. (2nd ed) pp 7–71. Academic Press, New York.
Lowry O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with Folin phenol reagent. J. Biol. Chem. 193: 265–275.
Norimura H., I. Hirashiki, F. Ogata, N. Yoshida and A. Ito, 1990. Purification of Bowman-Birk type inhibitor of high molecular weight from Wisteria seeds. Agric. Biol. Chem. 54: 3029–3030.
Noroika N., S. Hara, T. Ikenaka and J. Abe, 1988. Distribution of the Kunitz and the Bowman-Birk family proteinase inhibitors in leguminous seeds. Agric. Biol. Chem. 52: 1245–1252.
Pichare M.M., and M.S. Kachole, 1996. Protease inhibitors of pigeonpea (Cajanus cajan) and its wide relatives. Physiol. Plant. 98: 845–851.
Prabhu K.S., K. Saldanha and N. Pattabiraman, 1984. Natural plant enzyme inhibitors: A comparative study of the action of legume inhibitors on human and bovine pancreatic proteases. J. Sci. Food Agric. 35: 314–321.
Pusztai A., W.B. Watt and J.C. Stewart, 1991. A comprehensive scheme for the isolation of trypsin inhibitors and the agglutinin from soybean seeds. J. Agric. Food Chem. 39: 862–866.
Ryan C.A., 1990. Protease inhibitors in plants: Genes for improving defences against insects and pathogens. Ann. Rev. Phytopathology 28: 425–449.
Whiteley E.J. and D.E. Bowman, 1975. Isolation and properties of navy bean proteinase inhibitor component I. Arch. Biochem. Biophys. 169: 42–50.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gupta, P., Dhawan, K., Malhotra, S.P. et al. Purification and characterization of trypsin inhibitor from seeds of faba bean (Vicia faba L.). Acta Physiol Plant 22, 433–438 (2000). https://doi.org/10.1007/s11738-000-0085-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-000-0085-3