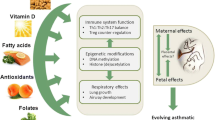
Abstract
Asthma occurs as a result of complex interactions of environmental and genetic factors. Clinical studies and animal models of asthma indicate offspring of allergic mothers have increased risk of development of allergies. Environmental factors including stress-induced corticosterone and vitamin E isoforms during pregnancy regulate the risk for offspring development of allergy. In this review, we discuss mechanisms for the development of allergic disease early in life, environmental factors that may impact the development of risk for allergic disease early in life, and how the variation in global prevalence of asthma may be explained, at least in part, by some environmental components.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Martinez FD. Genes, environments, development and asthma: a reappraisal. Eur Respir J. 2007;29:179–84.
Martinez FD, Vercelli D. Asthma. Lancet. 2013;382:1360–72.
Bousquet J, Bousquet PJ, Godard P, Daures JP. The public health implications of asthma. Bull World Health Organ. 2005;83:548–54.
Vollmer WM, Osborne ML, Buist AS. 20-year trends in the prevalence of asthma and chronic airflow obstruction in an HMO. Am J Respir Crit Care Med. 1998;157:1079–84.
Friebele E. The attack of asthma. Environ Health Perspect. 1996;104:22–5.
van Schayck CP, Smit HA. The prevalence of asthma in children: a reversing trend. Eur Respir J. 2005;26:647–50.
Lim RH, Kobzik L. Maternal transmission of asthma risk. Am J Reprod Immunol. 2009;61:1–10.
Hamada K et al. Allergen-independent maternal transmission of asthma susceptibility. J Immunol. 2003;170:1683–9.
Fedulov AV, Leme AS, Kobzik L. Duration of allergic susceptibility in maternal transmission of asthma risk. Am J Reprod Immunol. 2007;58:120–8.
Hubeau C, Apostolou I, Kobzik L. Targeting of CD25 and glucocorticoid-induced TNF receptor family-related gene-expressing T cells differentially modulates asthma risk in offspring of asthmatic and normal mother mice. J Immunol. 2007;178:1477–87.
Hubeau C, Apostolou I, Kobzik L. Adoptively transferred allergen-specific T cells cause maternal transmission of asthma risk. Am J Pathol. 2006;168:1931–9.
Herz U et al. Allergic sensitization and allergen exposure during pregnancy favor the development of atopy in the neonate. Int Arch Allergy Immunol. 2001;124:193–6.
Herz U et al. Prenatal sensitization in a mouse model. Am J Respir Crit Care Med. 2000;162:S62–5.
Jarrett E, Hall E. Selective suppression of IgE antibody responsiveness by maternal influence. Nature. 1979;280:145–7.
Wark PA, Murphy V, Mattes J. The interaction between mother and fetus and the development of allergic asthma. Expert Rev Respir Med. 2014;8:57–66.
Blumer N, Herz U, Wegmann M, Renz H. Prenatal lipopolysaccharide-exposure prevents allergic sensitization and airway inflammation, but not airway responsiveness in a murine model of experimental asthma. Clin Exp Allergy. 2005;35:397–402.
Devereux G, Barker RN, Seaton A. Antenatal determinants of neonatal immune responses to allergens. Clin Exp Allergy. 2002;32:43–50.
Uthoff H et al. Critical role of preconceptional immunization for protective and nonpathological specific immunity in murine neonates. J Immunol. 2003;171:3485–92.
Kurukulaaratchy RJ, Waterhouse L, Matthews SM, Arshad SH. Are influences during pregnancy associated with wheezing phenotypes during the first decade of life? Acta Paediatr. 2005;94:553–8.
Celedon JC et al. Exposure to cat allergen, maternal history of asthma, and wheezing in first 5 years of life. Lancet. 2002;360:781–2.
Kurukulaaratchy RJ, Matthews S, Waterhouse L, Arshad SH. Factors influencing symptom expression in children with bronchial hyperresponsiveness at 10 years of age. J Allergy Clin Immunol. 2003;112:311–6.
Latzin P et al. Prospectively assessed incidence, severity, and determinants of respiratory symptoms in the first year of life. Pediatr Pulmonol. 2007;42:41–50.
Martinez FD et al. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N Engl J Med. 1995;332:133–8.
Litonjua AA, Carey VJ, Burge HA, Weiss ST, Gold DR. Parental history and the risk for childhood asthma. Does mother confer more risk than father? Am J Respir Crit Care Med. 1998;158:176–81.
Lim RH, Kobzik L, Dahl M. Risk for asthma in offspring of asthmatic mothers versus fathers: a meta-analysis. PLoS One. 2010;5:e10134.
Folsgaard NV et al. Neonatal cytokine profile in the airway mucosal lining fluid is skewed by maternal atopy. Am J Respir Crit Care Med. 2012;185:275–80.
Bousquet J et al. MeDALL (Mechanisms of the Development of ALLergy): an integrated approach from phenotypes to systems medicine. Allergy. 2011;66:596–604.
Sonnenschein-van der Voort, A. M. et al. Influence of maternal and cord blood C-reactive protein on childhood respiratory symptoms and eczema. Pediatr Allergy Immunol. 2013;24:469–475.
Giwercman C et al. Increased risk of eczema but reduced risk of early wheezy disorder from exclusive breast-feeding in high-risk infants. J Allergy Clin Immunol. 2010;125:866–71.
Hamada K et al. Allergen-independent maternal transmission of asthma susceptibility. J Immunol. 2003;170:1683–9.
Lim RH, Kobzik L. Transplacental passage of interleukins 4 and 13? PLoS One. 2009;4:e4660.
Leme AS et al. Role of breast milk in a mouse model of maternal transmission of asthma susceptibility. J Immunol. 2006;176:762–9.
Hsu P, Nanan R. Foetal immune programming: hormones, cytokines, microbes and regulatory T cells. J Reprod Immunol. 2014;104–105:2–7.
Barrett EG, Rudolph K, Bowen LE, Bice DE. Parental allergic status influences the risk of developing allergic sensitization and an asthmatic-like phenotype in canine offspring. Immunology. 2003;110:493–500.
Abdala-Valencia H, Berdnikovs S, Soveg F, Cook-Mills JM. Alpha-tocopherol supplementation of allergic female mice inhibits development of CD11c+CD11b+ dendritic cells in utero and allergic inflammation in neonates. Am J Physiol Lung Cell Mol Physiol. 2014;307:L482–496. In this report using a mouse model, it was demonstrated that offspring of allergic mothers have an increase in numbers of distinct subsets of dendritic cells. The increase in numbers of these dendritic cell subsets in the fetal liver and pup lung of offspring of allergic mothers was blocked by supplementation of the allergic mother at the time of pregnancy with the vitamin E isoform α-tocopherol. In addition, α-tocopherol directly regulated bone marrow differentiation of these dendritic cell subsets in vitro.
Lim RH, Arredouani MS, Fedulov A, Kobzik L, Hubeau C. Maternal allergic contact dermatitis causes increased asthma risk in offspring. Respir Res. 2007;8:56.
Fedulov AV, Kobzik L. Allergy risk is mediated by dendritic cells with congenital epigenetic changes. Am J Respir Cell Mol Biol. 2011;44:285–92.
Williams JW. Transcription factor IRF4 drives dendritic cells to promote Th2 differentiation. Nat Commun. 2013;4:2990. There are many subsets of dendritic cells in the lung. In this report, they demonstrated that lung IRF4+ dendritic cell subsets drive the Th2 response in mice.
Mikhaylova L, Zhang Y, Kobzik L, Fedulov AV. Link between epigenomic alterations and genome-wide aberrant transcriptional response to allergen in dendritic cells conveying maternal asthma risk. PLoS One. 2013;8:e70387. The offspring of allergic mothers have an elevated risk of developing allergic responses in humans and animal models. This increased risk of developing allergic responses in offspring of allergic mothers is mediated by changes in offspring dendritic cells. In this report, they identified epigenomic changes in dendritic cells of offspring of allergic mice.
Stoney RM et al. Maternal breast milk long-chain n-3 fatty acids are associated with increased risk of atopy in breastfed infants. Clin Exp Allergy. 2004;34:194–200.
Reichardt P et al. Fatty acids in colostrum from mothers of children at high risk of atopy in relation to clinical and laboratory signs of allergy in the first year of life. Allergy. 2004;59:394–400.
Miyake Y, Okubo H, Sasaki S, Tanaka K, Hirota Y. Maternal dietary patterns during pregnancy and risk of wheeze and eczema in Japanese infants aged 16–24 months: the Osaka Maternal and Child Health Study. Pediatr Allergy Immunol. 2011;22:734–41.
Verhasselt V et al. Breast milk-mediated transfer of an antigen induces tolerance and protection from allergic asthma. Nat Med. 2008;14:170–5.
Mosconi E et al. Breast milk immune complexes are potent inducers of oral tolerance in neonates and prevent asthma development. Mucosal Immunol. 2010;3:461–74.
Muniz BP et al. Tolerogenic microenvironment in neonatal period induced by maternal immunization with ovalbumin. Immunobiology. 2014;219:377–84.
Fedulov AV et al. Pulmonary exposure to particles during pregnancy causes increased neonatal asthma susceptibility. Am J Respir Cell Mol Biol. 2008;38:57–67.
Gerhold K et al. Prenatal initiation of endotoxin airway exposure prevents subsequent allergen-induced sensitization and airway inflammation in mice. J Allergy Clin Immunol. 2006;118:666–73.
Gerhold K, Bluemchen K, Franke A, Stock P, Hamelmann E. Exposure to endotoxin and allergen in early life and its effect on allergen sensitization in mice. J Allergy Clin Immunol. 2003;112:389–96.
Tulic MK, Knight DA, Holt PG, Sly PD. Lipopolysaccharide inhibits the late-phase response to allergen by altering nitric oxide synthase activity and interleukin-10. Am J Respir Cell Mol Biol. 2001;24:640–6.
Gerhold K et al. Endotoxins prevent murine IgE production, T(H)2 immune responses, and development of airway eosinophilia but not airway hyperreactivity. J Allergy Clin Immunol. 2002;110:110–6.
Lima C et al. Modulation of the induction of lung and airway allergy in the offspring of IFN-gamma-treated mother mice. J Immunol. 2005;175:3554–9.
Fedulov A, Silverman E, Xiang Y, Leme A, Kobzik L. Immunostimulatory CpG oligonucleotides abrogate allergic susceptibility in a murine model of maternal asthma transmission. J Immunol. 2005;175:4292–300.
de Brito CA et al. CpG-induced Th1-type response in the downmodulation of early development of allergy and inhibition of B7 expression on T cells of newborn mice. J Clin Immunol. 2010;30:280–91.
Netting MJ, Middleton PF, Makrides M. Does maternal diet during pregnancy and lactation affect outcomes in offspring? A systematic review of food-based approaches Nutrition. 2014;30:1225–41.
Allan, K. M. et al. Maternal vitamin D and E intakes during pregnancy are associated with asthma in children. Eur Respir J 30, erj01022-02014 (2014).
Junge KM, Lehmann I, Borte M. Can vitamin D intake during pregnancy affect the risk of allergy in children? Expert Rev Clin Immunol. 2013;9:699–701.
Cook-Mills JM, Avila PC. Vitamin E and D regulation of allergic asthma immunopathogenesis. Int Immunopharmacol. 2014;29:007.
Camargo Jr CA et al. Maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. Am J Clin Nutr. 2007;85:788–95.
Beigelman A et al. The association between vitamin D status and the rate of exacerbations requiring oral corticosteroids in preschool children with recurrent wheezing. J Allergy Clin Immunol. 2014;133:1489–92.
Chawes BL et al. Cord blood 25(OH)-vitamin D deficiency and childhood asthma, allergy and eczema: the COPSAC2000 birth cohort study. PLoS One. 2014;9:e99856.
Uauy R et al. Safety and efficacy of omega-3 fatty acids in the nutrition of very low birth weight infants: soy oil and marine oil supplementation of formula. J Pediatr. 1994;124:612–20.
Boyle FG, Yuhas RJ, Lien EL. Red blood cell and tissue phospholipid fatty acid profiles of weanling rats fed infant formula fat blends containing soy and/or corn oil. Ann Nutr Metab. 1996;40:234–42.
Nelson SE, Rogers RR, Frantz JA, Ziegler EE. Palm olein in infant formula: absorption of fat and minerals by normal infants. Am J Clin Nutr. 1996;64:291–6.
Cook-Mills JM, McCary CA. Isoforms of vitamin E differentially regulate inflammation. Endocr Metab Immune Disord Drug Targets. 2010;10:348–66.
Berdnikovs S et al. Isoforms of vitamin E have opposing immunoregulatory functions during inflammation by regulating leukocyte recruitment. J Immunol. 2009;182:4395–405.
Hunter SC, Cahoon EB. Enhancing vitamin E in oilseeds: unraveling tocopherol and tocotrienol biosynthesis. Lipids. 2007;42:97–108.
Wolf G. How an increased intake of alpha-tocopherol can suppress the bioavailability of gamma-tocopherol. Nutr Rev. 2006;64:295–9.
Leonard SW, Terasawa Y, Farese Jr RV, Traber MG. Incorporation of deuterated RRR- or all-rac-alpha-tocopherol in plasma and tissues of alpha-tocopherol transfer protein–null mice. Am J Clin Nutr. 2002;75:555–60.
Brigelius-Flohe R, Traber MG. Vitamin E: function and metabolism. FASEB J. 1999;13:1145–55.
Podda M, Weber C, Traber MG, Packer L. Simultaneous determination of tissue tocopherols, tocotrienols, ubiquinols, and ubiquinones. J Lipid Res. 1996;37:893–901.
Traber MG, Kayden HJ. Preferential incorporation of alpha-tocopherol vs gamma-tocopherol in human lipoproteins. Am J Clin Nutr. 1989;49:517–26.
McCary CA et al. Vitamin E isoforms directly bind PKCalpha and differentially regulate activation of PKCalpha. Biochem J. 2012;441:189–98.
McCary CA, Abdala-Valencia H, Berdnikovs S, Cook-Mills JM. Supplemental and highly elevated tocopherol doses differentially regulate allergic inflammation: reversibility of alpha-tocopherol and gamma-tocopherol’s effects. J Immunol. 2011;186:3674–85.
Cook-Mills JM, Marchese M, Abdala-Valencia H. Vascular cell adhesion molecule-1 expression and signaling during disease: regulation by reactive oxygen species and antioxidants. Antioxid Redox Signal. 2011;15:1607–38.
Cook-Mills JM, Marchese ME, Abdala-Valencia H. Vascular cell adhesion molecule-1 expression and signaling during disease: regulation by reactive oxygen species and antioxidants. Antioxid Redox Signal. 2011;15:1607–38.
Cook-Mills, J. M. in Eosinophils in health and disease. (ed J.J. and Rosenberg Lee, H.F) 139–153 (Elsevier, 2012)
Marchese ME. The vitamin E isoforms alpha-tocopherol and gamma-tocopherol have opposite associations with spirometric parameters: the CARDIA study. Respir Res. 2014;15:31. The prevalence of allergies has increased in just a few decades suggesting that environmental factors likely impact allergies and asthma. In this report, it is demonstrated in a 20-year prospective study with 4500 individuals in the USA that, by age 21, human plasma α-tocopherol associates with better lung spirometry and human plasma γ-tocopherol associates with worse lung spirometry. Thus, the balance of α-tocopherol and γ-tocopherol regulates adult allergic responses. Regulation of allergies and asthma in women has the potential to influence the development of risk of allergies in her children.
Tabak C et al. Dietary factors and pulmonary function: a cross sectional study in middle aged men from three European countries. Thorax. 1999;54:1021–6.
Smit HA, Grievink L, Tabak C. Dietary influences on chronic obstructive lung disease and asthma: a review of the epidemiological evidence. Proc Nutr Soc. 1999;58:309–19.
Weiss ST. Diet as a risk factor for asthma. Ciba Fndn Symp. 1997;206:244–57.
Troisi RJ et al. A prospective study of diet and adult-onset asthma. Am J Respir Crit Care Med. 1995;151:1401–8.
Dow L et al. Does dietary intake of vitamins C and E influence lung function in older people? Am J Respir Crit Care Med. 1996;154:1401–4.
Wagner KH, Kamal-Eldin A, Elmadfa I. Gamma-tocopherol—an underestimated vitamin? Ann Nutr Metab. 2004;48:169–88.
Jiang Q, Christen S, Shigenaga MK, Ames BN. Gamma-tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am J Clin Nutr. 2001;74:714–22.
Muller-Schmehl K et al. Localization of alpha-tocopherol transfer protein in trophoblast, fetal capillaries’ endothelium and amnion epithelium of human term placenta. Free Radic Res. 2004;38:413–20.
Jishage K et al. Vitamin E is essential for mouse placentation but not for embryonic development itself. Biol Reprod. 2005;73:983–7.
Kalayci O, Besler T, Kilinc K, Sekerel BE, Saraclar Y. Serum levels of antioxidant vitamins (alpha tocopherol, beta carotene, and ascorbic acid) in children with bronchial asthma. Turk J Peds. 2000;42:17–21.
Kelly FJ, Mudway I, Blomberg A, Frew A, Sandstrom T. Altered lung antioxidant status in patients with mild asthma. Lancet. 1999;354:482–3.
Shvedova AA, Kisin ER, Kagan VE, Karol MH. Increased lipid peroxidation and decreased antioxidants in lungs of guinea pigs following an allergic pulmonary response. Tox Appl Pharm. 1995;132:72–81.
Schunemann HJ et al. The relation of serum levels of antioxidant vitamins C and E, retinol and carotenoids with pulmonary function in the general population. Am J Respir Crit Care Med. 2001;163:1246–55.
Zourbas S, Dubanchet S, Martal J, Chaouat G. Localization of pro-inflammatory (IL-12, IL-15) and anti-inflammatory (IL-11, IL-13) cytokines at the foetomaternal interface during murine pregnancy. Clin Exp Immunol. 2001;126:519–28.
Ostojic S et al. Demonstration of the presence of IL-16, IL-17 and IL-18 at the murine fetomaternal interface during murine pregnancy. Am J Reprod Immunol. 2003;49:101–12.
Bowen JM, Chamley L, Mitchell MD, Keelan JA. Cytokines of the placenta and extra-placental membranes: biosynthesis, secretion and roles in establishment of pregnancy in women. Placenta. 2002;23:239–56.
Gregor H et al. The passage of granulocyte-macrophage colony-stimulating factor across the human placenta perfused in vitro. J Soc Gynecol Investig. 1999;6:307–10.
Lim R, Fedulov AV, Kobzik L. Maternal stress during pregnancy increases neonatal allergy susceptibility: role of glucocorticoids. Am J Physiol Lung Cell Mol Physiol. 2014;307:L141–148. Maternal factors that are transferred from allergic mothers to offspring for the increased risk of allergy in the offspring were not known. In this report, they demonstrated in mice that maternal corticosterone is sufficient to increase the risk of allergic responses in offspring.
Costa-Pinto FA, Basso AS, Britto LR, Malucelli BE, Russo M. Avoidance behavior and neural correlates of allergen exposure in a murine model of asthma. Brain Behav Immun. 2005;19:52–60.
Costa-Pinto FA et al. Neural correlates of IgE-mediated allergy. Ann N Y Acad Sci. 2006;1088:116–31.
Portela Cde P, Massoco Cde O, de Lima WT, Palermo-Neto J. Stress-induced increment on total bronchoalveolar cell count in OVA-sensitized rats. Physiol Behav. 2001;72:415–20.
Portela CP et al. Effects of stress and neuropeptides on airway responses in ovalbumin-sensitized rats. Neuroimmunomodulation. 2007;14:105–11.
Portela Cde P, Tiberio Ide F, Leick-Maldonado EA, Martins MA, Palermo-Neto J. Effects of diazepam and stress on lung inflammatory response in OVA-sensitized rats. Am J Physiol Lung Cell Mol Physiol. 2002;282:L1289–95.
Tonelli LH et al. Allergic rhinitis induces anxiety-like behavior and altered social interaction in rodents. Brain Behav Immun. 2009;23:784–93.
Lu Y et al. Effects of stress in early life on immune functions in rats with asthma and the effects of music therapy Early-life psychological stress exacerbates adult mouse asthma via the hypothalamus-pituitary-adrenal axis. J Asthma. 2010;47:526–31.
Chida Y, Sudo N, Sonoda J, Hiramoto T, Kubo C. Early-life psychological stress exacerbates adult mouse asthma via the hypothalamus-pituitary-adrenal axis. Am J Respir Crit Care Med. 2007;175:316–22.
Strine TW et al. Depression and anxiety in the United States: findings from the 2006 Behavioral Risk Factor Surveillance System. Psychiatr Serv. 2008;59:1383–90.
Cheung TK et al. Gastroesophageal reflux disease is associated with poor asthma control, quality of life, and psychological status in Chinese asthma patients. Chest. 2009;135:1181–5.
Sansone RA, Sansone LA. Asthma: wheezing, woes, and worries. Psychiatry. 2008;5:48–52.
Di Marco F et al. Close correlation between anxiety, depression, and asthma control. Respir Med. 2010;104:22–8.
Cordina M, Fenech AG, Vassallo J, Cacciottolo JM. Anxiety and the management of asthma in an adult outpatient population. Ther Adv Respir Dis. 2009;3:227–33.
von Hertzen LC. Maternal stress and T-cell differentiation of the developing immune system: possible implications for the development of asthma and atopy. J Allergy Clin Immunol. 2002;109:923–8.
Huang CC, Shih MC, Hsu NC, Chien Y, Chung BC. Fetal glucocorticoid synthesis is required for development of fetal adrenal medulla and hypothalamus feedback suppression. Endocrinology. 2012;153:4749–56.
Norbiato G, Bevilacqua M, Vago T, Clerici M. Glucocorticoids and Th-1, Th-2 type cytokines in rheumatoid arthritis, osteoarthritis, asthma, atopic dermatitis and AIDS. Clin Exp Rheumatol. 1997;15:315–23.
Ramirez F, Fowell DJ, Puklavec M, Simmonds S, Mason D. Glucocorticoids promote a TH2 cytokine response by CD4+ T cells in vitro. J Immunol. 1996;156:2406–12.
Groer MW, Humenick S, Hill PD. Characterizations and psychoneuroimmunologic implications of secretory immunoglobulin A and cortisol in preterm and term breast milk. J Perinat Neonatal Nurs. 1994;7:42–51.
Murphy VE et al. Reduced 11beta-hydroxysteroid dehydrogenase type 2 activity is associated with decreased birth weight centile in pregnancies complicated by asthma. J Clin Endocrinol Metab. 2002;87:1660–8.
Murphy VE et al. Maternal asthma is associated with reduced female fetal growth. Am J Respir Crit Care Med. 2003;168:1317–23.
Compliance with Ethics Guidelines
Conflict of Interest
Dr. Cook-Mills has a patent pre-application, Compositions and Methods for the Treatment of Natal and Prenatal Conditions with Alpha-Tocopherol Pending.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human subjects performed by any of the authors.
Sources of Support
This study was supported by National Institutes of Health Grant R01 AT004837 (J.M.C-M).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Basic and Applied Science
Rights and permissions
About this article
Cite this article
Cook-Mills, J.M. Maternal Influences over Offspring Allergic Responses. Curr Allergy Asthma Rep 15, 1 (2015). https://doi.org/10.1007/s11882-014-0501-1
Published:
DOI: https://doi.org/10.1007/s11882-014-0501-1