Abstract
Purpose of Review
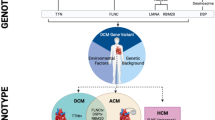
Genetic studies have identified an increasing number of culprit disease genes in patients with dilated cardiomyopathy (DCM). While these studies were originally conducted in patients with primary DCM, recent research has identified culprit mutations among patients who develop DCM in response to an environmental exposure. This review will summarize the genetic architecture of DCM secondary to alcohol, anthracyclines, and pregnancy, as well as the potential modifying role of exercise.
Recent Findings
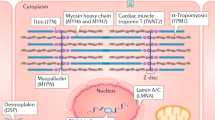
Mutations in DCM-associated genes are identified in 10–20% of patients categorized as having a secondary cardiomyopathy, a similar frequency to patients with primary DCM. The most commonly mutated gene is TTN, which encodes the sarcomere protein titin. Clinical outcomes differ among patients with or without an identifiable mutation.
Summary
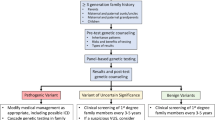
Genetic contributors should be sought in patients with presumed secondary cardiomyopathy. Future studies are necessary to determine the prospective management of previously unaffected patients known to have a DCM-associated mutation at the time of pregnancy or exposure to cardiotoxins.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Pinto YM, Elliott PM, Arbustini E, et al. Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur Heart J. 2016. https://doi.org/10.1093/eurheartj/ehv727.
Bozkurt B, Colvin M, Cook J, et al. Current diagnostic and treatment strategies for specific dilated Cardiomyopathies: A Scientific Statement from the American Heart Association. Circulation. 2016. https://doi.org/10.1161/CIR.0000000000000455.
Codd MB, Sugrue DD, Gersh BJ, Melton LJ. Epidemiology of idiopathic dilated and hypertrophic cardiomyopathy: a population-based study in Olmsted County, Minnesota, 1975-1984. Circulation. 1989. https://doi.org/10.1161/01.CIR.80.3.564.
Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019. https://doi.org/10.1161/CIR.0000000000000659.
Stolfo D, Uijl A, Vedin O, Strömberg A, Faxén UL, Rosano GMC, et al. Sex-based differences in heart failure across the ejection fraction spectrum: phenotyping, and prognostic and therapeutic implications. JACC Hear Fail. 2019. https://doi.org/10.1016/j.jchf.2019.03.011.
McMurray J, Packer M, Desai A. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014. https://doi.org/10.1056/NEJMoa1409077.
Swedberg K, Komajda M, Böhm M, Borer JS, Ford I, Dubost-Brama A, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet. 2010. https://doi.org/10.1016/S0140-6736(10)61198-1.
Colucci WS, Kolias TJ, Adams KF, Armstrong WF, Ghali JK, Gottlieb SS, et al. Metoprolol reverses left ventricular remodeling in patients with asymptomatic systolic dysfunction: the REversal of VEntricular remodeling with Toprol-XL (REVERT) trial. Circulation. 2007;116:49–56.
Chambers DC, Cherikh WS, Goldfarb SB, et al. The international thoracic organ transplant registry of the International Society for Heart and Lung Transplantation: thirty-fifth adult lung and heart-lung transplant report—2018; Focus theme: Multiorgan Transplantation. J Hear Lung Transplant. 2018. https://doi.org/10.1016/j.healun.2018.07.020.
Mahon NG, Murphy RT, MacRae CA, Caforio ALP, Elliott PM, McKenna WJ. Echocardiographic evaluation in asymptomatic relatives of patients with dilated cardiomyopathy reveals preclinical disease. Ann Intern Med. 2005.
Hershberger RE, Morales A, Siegfried JD. Clinical and genetic issues in dilated cardiomyopathy: a review for genetics professionals. Genet Med. 2010. https://doi.org/10.1097/GIM.0b013e3181f2481f.
Truncations of Titin Causing Dilated Cardiomyopathy - NEJMoa1110186. http://www.nejm.org/doi/pdf/10.1056/NEJMoa1110186. Accessed 24 Apr 2013.
Zimmerman RS, Cox S, Lakdawala NK, Cirino A, Mancini-DiNardo D, Clark E, et al. A novel custom resequencing array for dilated cardiomyopathy. Genet Med. 2010;12:268–78.
Jansweijer JA, Nieuwhof K, Russo F, Van Den Berg MP, Wilde AAM, De Boer RA, et al. Truncating titin mutations cause a mild and treatable form of dilated cardiomyopathy. Eur Hear J. 2015.
Schafer S, De Marvao A, Adami E, et al. Titin-truncating variants affect heart function in disease cohorts and the general population. Nat GenetNat Genet. 2017. https://doi.org/10.1038/ng.3719.
World Health Organization (2018) Global status report on alcohol and health 2018. Glob status Rep alcohol. doi: /entity/substance_abuse/publications/global_alcohol_report/en/index.html.
Sacks JJ, Gonzales KR, Bouchery EE, Tomedi LE, Brewer RD. 2010 national and state costs of excessive alcohol consumption. Am J Prev Med. 2015. https://doi.org/10.1016/j.amepre.2015.05.031.
Laonigro I, Correale M, Di Biase M, Altomare E. Alcohol abuse and heart failure. Eur J Heart Fail. 2009. https://doi.org/10.1093/eurjhf/hfp037.
Fernández-Solà J. Cardiovascular risks and benefits of moderate and heavy alcohol consumption. Nat Rev Cardiol. 2015. https://doi.org/10.1038/nrcardio.2015.91.
Djoussé L, Gaziano JM. Alcohol consumption and risk of heart failure in the physicians’ health study I. Circulation. 2007. https://doi.org/10.1161/CIRCULATIONAHA.106.661868.
Abramson JL, Williams SA, Krumholz HM, Vaccarino V. Moderate alcohol consumption and risk of heart failure among older persons. J Am Med Assoc. 2001. https://doi.org/10.1001/jama.285.15.1971.
Wood AM, Kaptoge S, Butterworth AS, et al. Risk thresholds for alcohol consumption: combined analysis of individual-participant data for 599 912 current drinkers in 83 prospective studies. Lancet. 2018. https://doi.org/10.1016/S0140-6736(18)30134-X.
Costanzo S, Di Castelnuovo A, Donati MB, Iacoviello L, de Gaetano G. Alcohol consumption and mortality in patients with cardiovascular disease. A Meta-Analysis. J Am Coll Cardiol. 2010. https://doi.org/10.1016/j.jacc.2010.01.006.
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:2129–200.
Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American heart association nutrition committee. Circulation. 2006. https://doi.org/10.1161/CIRCULATIONAHA.106.176158.
•• Ware JS, Amor-Salamanca A, Tayal U, et al. Genetic etiology for alcohol-induced cardiac toxicity. J Am Coll Cardiol. 2018. https://doi.org/10.1016/j.jacc.2018.03.462 Very important study describing the frequency of DCM associated mutations amongst patients with alcoholic cardiomyopathy.
Cardinale D, Colombo A, Bacchiani G, et al. Early detection of anthracycline cardiotoxicity and improvement with heart failure therapy. Circulation. 2015. https://doi.org/10.1161/CIRCULATIONAHA.114.013777.
Zamorano JL, Lancellotti P, Muñoz DR, Aboyans V, Asteggiano R, Galderisi M, et al. ESC position paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for practice guidelines: the task force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:2768–801.
Armenian SH, Xu L, Ky B, Sun C, Farol LT, Pal SK, et al. Cardiovascular disease among survivors of adult-onset cancer: a community-based retrospective cohort study. J Clin Oncol. 2016. https://doi.org/10.1200/JCO.2015.64.0409.
Swain SM, Whaley FS, Ewer MS. Congestive heart failure in patients treated with doxorubicin: a retrospective analysis of three trials. Cancer. 2003. https://doi.org/10.1002/cncr.11407.
Wojnowski L, Kulle B, Schirmer M, et al. NAD(P)H oxidase and multidrug resistance protein genetic polymorphisms are associated with doxorubicin-induced cardiotoxicity. Circulation. 2005. https://doi.org/10.1161/CIRCULATIONAHA.105.576850.
Wells QS, Veatch OJ, Fessel JP, et al. Genome-wide association and pathway analysis of left ventricular function after anthracycline exposure in adults. Pharmacogenet Genomics. 2017. https://doi.org/10.1097/FPC.0000000000000284.
Blanco JG, Leisenring WM, Gonzalez-Covarrubias VM, Kawashima TI, Davies SM, Relling MV, et al. Genetic polymorphisms in the carbonyl reductase 3 gene CBR3 and the NAD(P)H:quinone oxidoreductase 1 gene NQ01 in patients who developed anthracycline-related congestive heart failure after childhood cancer. Cancer. 2008. https://doi.org/10.1002/cncr.23534.
•• Garcia-Pavia P, Kim Y, Alejandra Restrepo-Cordoba M, et al. Genetic variants associated with Cancer therapy-induced cardiomyopathy. Circulation. 2019. https://doi.org/10.1161/CIRCULATIONAHA.118.037934 Very important study describing the frequency of truncating variants in TTN amongst patients with anthracycline cardiomyopathy.
Hilfiker-Kleiner D, Sliwa K, Mebazaa A, van der Meer P, Bauersachs J, Mbakwem A, et al. Long-term prognosis, subsequent pregnancy, contraception and overall management of peripartum cardiomyopathy: practical guidance paper from the heart failure Association of the European Society of cardiology study group on Peripartum cardiomyopathy. Eur J Heart Fail. 2018;20:951–62.
Koenig T, Hilfiker-Kleiner D, Bauersachs J. Peripartum cardiomyopathy. Herz. 2018;43:431–7.
Elkayam U. Clinical characteristics of peripartum cardiomyopathy in the United States: diagnosis, prognosis, and management. J Am Coll Cardiol. 2011;58:659–70.
Van Spaendonck-Zwarts KY, Posafalvi A, Van Den Berg MP, et al. Titin genemutations are common in families with both peripartum cardiomyopathy and dilated cardiomyopathy. Eur Heart J. 2014;35:2165–73.
Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C, et al. ESC guidelines on the management of cardiovascular diseases during pregnancy. Eur Heart J. 2011;32:3147–97.
•• Ware JS, Li J, Mazaika E, et al. shared genetic predisposition in Peripartum and dilated cardiomyopathies. Obstet Anesth Dig. 2016;36:144–5 Very important study of the genetic basis of peripartum cardiomyopathy.
Haghikia A, Podewski E, Libhaber E, et al. Phenotyping and outcome on contemporary management in a German cohort of patients with peripartum cardiomyopathy. Basic Res Cardiol. 2013. https://doi.org/10.1007/s00395-013-0366-9.
McNamara DM, Elkayam U, Alharethi R, Damp J, Hsich E, Ewald G, et al. Clinical outcomes for Peripartum cardiomyopathy in North America. J Am Coll Cardiol. 2015;66:905–14.
Dickstein K, Cohen-Solal A, Filippatos G, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2008. Eur Heart J. 2008. https://doi.org/10.1093/eurheartj/ehn309.
O’Connor CM, Whellan DJ, Lee KL, et al. Efficacy and safety of exercise training in patients with chronic heart failure HF-ACTION randomized controlled trial. JAMA. 2009. https://doi.org/10.1001/jama.2009.454.
Ho CY, Day SM, Ashley EA, et al. Genotype and lifetime burden of disease in hypertrophic cardiomyopathy: insights from the Sarcomeric human cardiomyopathy registry (SHaRe). Circulation. 2018. https://doi.org/10.1161/CIRCULATIONAHA.117.033200.
Towbin JA, McKenna WJ, Abrams DJ, et al. 2019 HRS expert consensus statement on evaluation, risk stratification, and Management of Arrhythmogenic Cardiomyopathy. Heart Rhythm. 2019. https://doi.org/10.1016/j.hrthm.2019.05.007.
Ruwald AC, Marcus F, Estes NAM, Link M, McNitt S, Polonsky B, et al. Association of competitive and recreational sport participation with cardiac events in patients with arrhythmogenic right ventricular cardiomyopathy: results from the north American multidisciplinary study of arrhythmogenic right ventricular cardiomyopath. Eur Heart J. 2015. https://doi.org/10.1093/eurheartj/ehv110.
Pasotti M, Klersy C, Pilotto A, et al. Long-term outcome and risk stratification in dilated Cardiolaminopathies. J Am Coll Cardiol. 2008. https://doi.org/10.1016/j.jacc.2008.06.044.
Cattin ME, Ferry A, Vignaud A, Mougenot N, Jacquet A, Wahbi K, et al. Mutation in Lamin a/C sensitizes the myocardium to exercise-induced mechanical stress but has no effect on skeletal muscles in mouse. Neuromuscul Disord. 2016. https://doi.org/10.1016/j.nmd.2016.05.010.
Chandar S, Yeo LS, Leimena C, et al. Effects of mechanical stress and carvedilol in Lamin a/C-deficient dilated cardiomyopathy. Circ Res. 2010. https://doi.org/10.1161/CIRCRESAHA.109.204388.
Maron BJ, Chaitman BR, Ackerman MJ, et al. Recommendations for physical activity and recreational sports participation for young patients with genetic cardiovascular diseases. Circulation. 2004. https://doi.org/10.1161/01.CIR.0000128363.85581.E1.
Maron BJ, Udelson JE, Bonow RO, Nishimura RA, Ackerman MJ, Estes NAM, et al. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: task force 3: hypertrophic cardiomyopathy, Arrhythmogenic right ventricular cardiomyopathy and other cardiomyopathies, and myocarditis: a Scientif. J Am Coll Cardiol. 2015. https://doi.org/10.1016/j.jacc.2015.09.035.
Pelliccia A, Fagard R, Bjørnstad HH, et al. Recommendations for competitive sports participation in athletes with cardiovascular disease. Med Dello Sport. 2005.
Pelliccia A, Solberg EE, Papadakis M, et al. Recommendations for participation in competitive and leisure time sport in athletes with cardiomyopathies, myocarditis, and pericarditis: position statement of the sport cardiology section of the European Association of Preventive Cardiology (EAPC). Eur Heart J. 2019. https://doi.org/10.1093/eurheartj/ehy730.
Maron BJ, Zipes DP, Kovacs RJ. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: preamble, principles, and general considerations. J Am Coll Cardiol. 2015. https://doi.org/10.1016/j.jacc.2015.09.032.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Peter Marstrand receives research support from The Danish Heart Foundation (Grant 17-R115-A7532-22065), The Elite Research Travel Grant from the Danish Ministry of Higher Education and Science, The FUKAP Foundation, Torben og Alice Frimodts Fond, Augustinus fonden and Knud Højgaards fond.
Neal K. Lakdawala serves as a consultant for Myokardia Inc. and Array BioPharma. He also receives unrestricted research support from the O’Hare Family Foundation for investigation of LMNA cardiomyopathy.
Kermshlise Picard declares no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Myocardial Disease
Rights and permissions
About this article
Cite this article
Marstrand, P., Picard, K. & Lakdawala, N.K. Second Hits in Dilated Cardiomyopathy. Curr Cardiol Rep 22, 8 (2020). https://doi.org/10.1007/s11886-020-1260-3
Published:
DOI: https://doi.org/10.1007/s11886-020-1260-3