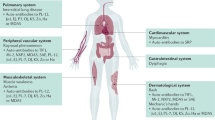
Abstract
The idiopathic inflammatory myopathies (IIMs) comprise a group of autoimmune disorders that target skeletal muscle. They are characterized by typical laboratory and clinical features including muscle weakness, elevated muscle enzymes, characteristic histopathology of muscle biopsies, as well as electromyography abnormalities. The IIMs are divided into polymyositis, dermatomyositis, inclusion body myositis, nonspecific myositis, and immune-mediated necrotizing myopathy (IMNM). IMNM is distinguished by the absence of primary inflammation on muscle biopsy. IMNM may be associated with myositis-specific autoantibodies (i.e., anti-SRP and anti-HMGCR) and malignancy, in association with viral infections (HIV or hepatitis C), or in relation to other connective tissue diseases (i.e., scleroderma). Typical clinical findings such as severe muscle weakness, highly elevated creatine kinase (CK) levels, as well as resistance to conventional immunosuppressive therapy are associated with this subtype of IIM. This review provides an overview of this disease entity and focuses on its diagnosis and treatment.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Cox S, Limaye V, Hill C, Blumbergs P, Roberts-Thomson P. Idiopathic inflammatory myopathies: diagnostic criteria, classification and epidemiological features. Int. J. Rheum. Dis. 2010:117–24.
Iago PF, Albert SO, Andreu FC, Xavier MG, Jose RP, Jordi PL, et al. Pregnancy in adult-onset idiopathic inflammatory myopathy: report from a cohort of myositis patients from a single center. Semin. Arthritis Rheum. [Internet]. 2014; Available from: http://www.ncbi.nlm.nih.gov/pubmed/24906908.
Gazeley DJ, Cronin ME. Diagnosis and treatment of the idiopathic inflammatory myopathies. Ther. Adv. Musculoskelet. Dis. [Internet]. 2011;3:315–24. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3383495&tool=pmcentrez&rendertype=abstract.
Hoogendijk JE, Amato AA, Lecky BR, Choy EH, Lundberg IE, Rose MR, et al. 119th ENMC international workshop: trial design in adult idiopathic inflammatory myopathies, with the exception of inclusion body myositis, 10-12 October 2003, Naarden, The Netherlands. Neuromuscul Disord England. 2004:337–45.
Amato A a, Greenberg S a. Inflammatory myopathies. Continuum (Minneap. Minn). [Internet]. 2013;19:1615–33. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24305450. An excellent overview of inflammatory myopathies with helpful author commentary.
Bohan A, Peter JB. Polymyositis and dermatomyositis. N Engl J Med. 1975;292:344–7.
Targoff IN, Miller FW, Medsger Jr TA, Oddis CV. Classification criteria for the idiopathic inflammatory myopathies. Curr Opin Rheumatol. 1997;9:527–35.
Tanimoto K, Nakano K, Kano S, Mori S, Ueki H, Nishitani H, et al. Classification criteria for polymyositis and dermatomyositis. J Rheumatol. 1995;22:668–74.
Troyanov Y, Targoff IN, Tremblay J-L, Goulet J-R, Raymond Y, Senécal J-L. Novel classification of idiopathic inflammatory myopathies based on overlap syndrome features and autoantibodies: analysis of 100 French Canadian patients. Medicine (Baltimore). 2005;84:231–49.
Lazarou IN, Guerne P-A. Classification, diagnosis, and management of idiopathic inflammatory myopathies. J. Rheumatol. [Internet]. 2013;40:550–64. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23504386. A comprehensive review of IIMs, including detailed analysis of current advances in classification, diagnosis and management.
Quinn C, Salameh JS, Smith T, Souayah N. Necrotizing myopathies: an update. J Clin Neuromuscul Dis USA. 2015;16:131–40.
Castro C, Gourley M. Diagnosis and treatment of inflammatory myopathy: issues and management. Ther. Adv. Musculoskelet. Dis [Internet]. 2012;4:111–20. Available from: http://tab.sagepub.com/content/early/2011/12/19/1759720X11425092.abstract.
Mammen AL. Necrotizing myopathies: beyond statins. Curr Opin Rheumatol. 2014;26:679–83.
Bronner IM, Hoogendijk JE, Wintzen AR, Van Der Meulen MFG, Linssen WHJP, Wokke JHJ, et al. Necrotising myopathy, an unusual presentation of a steroid-responsive myopathy. J Neurol. 2003;250:480–5.
Smith B. Skeletal muscle necrosis associated with cainoma. J Pathol England. 1969;97:207–10.
Wegener S, Bremer J, Komminoth P, Jung HH, Weller M. Paraneoplastic necrotizing myopathy with a mild inflammatory component: a case report and review of the literature. Case Rep. Oncol. 2010:88–92.
Levin MI, Mozaffar T, Al-Lozi MT, Pestronk A. Paraneoplastic necrotizing myopathy: clinical and pathological features. Neurology. 1998:764–7.
Sampson JB, Smith SM, Smith AG, Singleton JR, Chin S, Pestronk A, et al. Paraneoplastic myopathy: response to intravenous immunoglobulin. Neuromuscul Disord. 2007;17:404–8.
Galani E, Bonakis A, Christodoulou C, Klouvas G, Angeliki D, Skarlos D. Can cetuximab affect paraneoplastic myopathy? J Neurooncol. 2009;93:437–8.
Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve. 2010;41:185–90.
Allenbach Y, Drouot L, Rigolet A, Charuel JL, Jouen F, Romero NB, et al. Anti-HMGCR autoantibodies in European patients with autoimmune necrotizing myopathies: inconstant exposure to statin. Medicine (Baltimore). [Internet]. 2014;93:150–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24797170. A comprehensive analysis of anti-HMGCR positive patients in a European cohort.
Dalakas MC. Review: an update on inflammatory and autoimmune myopathies. Neuropathol. Appl. Neurobiol. 2011:226–42.
Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum. 2010;62:2757–66.
Satoh J, Eguchi Y, Narukiyo T, Mizuta T, Kobayashi O, Kawai M, et al. Necrotizing myopathy in a patient with chronic hepatitis C virus infection: a case report and a review of the literature. Intern Med [Internet]. 2000;39:176–81. Available from: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10732841.
Wrzolek MA, Sher JH, Kozlowski PB, Rao C. Skeletal muscle pathology in AIDS: an autopsy study. Muscle Nerve USA. 1990;13:508–15.
Snider WD, Simpson DM, Nielsen S, Gold JW, Metroka CE, Posner JB. Neurological complications of acquired immune deficiency syndrome: analysis of 50 patients. Ann Neurol. 1983;14:403–18.
Liang C, Needham M. Necrotizing autoimmune myopathy. Curr Opin Rheumatol. 2011;23:612–9.
Stern R, Gold J, DiCarlo EF. Myopathy complicating the acquired immune deficiency syndrome. Muscle Nerve. 1987:318–22.
Simpson DM. Myopathy associated with human immunodeficiency virus (HIV) but not with zidovudine. Ann Intern Med USA. 1988:842.
Simpson DM, Bender AN. Human immunodeficiency virus-associated myopathy: analysis of 11 patients. Ann Neurol. 1988;24:79–84.
Lange DJ, Britton CB, Younger DS, Hays AP. The neuromuscular manifestations of human immunodeficiency virus infections. Arch Neurol. 1988;45:1084–8.
Mammen AL, Chung T, Christopher-Stine L, Rosen P, Rosen A, Doering KR, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme a reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum. 2011;63:713–21. Landmark paper regarding initial identification of the HMGCR autoantibody in a cohort of patients with IMNM.
Ellis E, Tan JA, Lester S, Tucker G, Blumbergs P, Roberts-Thomson P, et al. Necrotizing myopathy: clinicoserologic associations. Muscle Nerve. 2012;45:189–94.
Bhansing KJ, Lammens M, Knaapen HK, van Riel PL, van Engelen BG, Vonk MC. Scleroderma-polymyositis overlap syndrome versus idiopathic polymyositis and systemic sclerosis: a descriptive study on clinical features and myopathology. Arthritis Res. Ther. [Internet]. 2014;16:R111. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4060195&tool=pmcentrez&rendertype=abstract
Ranque B, Authier F-J, Le-Guern V, Pagnoux C, Berezne A, Allanore Y, et al. A descriptive and prognostic study of systemic sclerosis-associated myopathies. Ann Rheum Dis. 2009;68:1474–7.
Reeves WH, Nigam SK, Blobel G. Human autoantibodies reactive with the signal-recognition particle. Proc Natl Acad Sci USA. 1986;83:9507–11.
Miller T, Al-Lozi MT, Lopate G, Pestronk A. Myopathy with antibodies to the signal recognition particle: clinical and pathological features. J. Neurol. Neurosurg. Psychiatry. 2002:420–8.
Kao AH, Lacomis D, Lucas M, Fertig N, Oddis CV. Anti-signal recognition particle autoantibody in patients with and patients without idiopathic inflammatory myopathy. Arthritis Rheum. 2004;50:209–15.
Hengstman GJD, ter Laak HJ, Vree Egberts WTM, Lundberg IE, Moutsopoulos HM, Vencovsky J, et al. Anti-signal recognition particle autoantibodies: marker of a necrotising myopathy. Ann Rheum Dis England. 2006;65:1635–8.
Love LA, Leff RL, Fraser DD, Targoff IN, Dalakas M, Plotz PH, et al. A new approach to the classification of idiopathic inflammatory myopathy: myositis-specific autoantibodies define useful homogeneous patient groups. Medicine (Baltimore). 1991;70:360–74.
Fernandez C, Bardin N, De Paula AM, Salort-Campana E, Benyamine A, Franques J, et al. Correlation of clinicoserologic and pathologic classifications of inflammatory myopathies: study of 178 cases and guidelines for diagnosis. Medicine (Baltimore). [Internet]. 2013;92:15–24. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23269233
Mammen AL, Pak K, Williams EK, Brisson D, Coresh J, Selvin E, et al. Rarity of anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase antibodies in statin users, including those with self-limited musculoskeletal side effects. Arthritis Care Res. 2012;64:269–72.
Albayda J, Mammen AL. Is statin-induced myositis part of the polymyositis disease spectrum? Curr. Rheumatol. Rep USA. 2014;16:433.
Amato AA, Barohn RJ. Evaluation and treatment of inflammatory myopathies. J Neurol Neurosurg Psychiatr England. 2009;80:1060–8.
Suzuki S, Nishikawa A, Kuwana M, Nishimura H, Watanabe Y, Nakahara J, et al. Inflammatory myopathy with anti-signal recognition particle antibodies: case series of 100 patients. Orphanet J Rare Dis England. 2015;10:61. Comprehensive case series analayzing a large number of patients with anti-SRP associated myopathy.
Mohassel P, Mammen AL. Statin-associated autoimmune myopathy and anti-HMGCR autoantibodies. Muscle Nerve. 2013:477–83.
Takada T, Hirakata M, Suwa A, Kaneko Y, Kuwana M, Ishihara T, et al. Clinical and histopathological features of myopathies in Japanese patients with anti-SRP autoantibodies. Mod Rheumatol Japan. 2009;19:156–64.
Sugie K, Eura N, Kobayashi Y, Sawa N, Ueno S. Clinicopathological and neuroradiological features of myopathy associated with antibodies to signal recognition particle (SRP). Eur. J. Intern. Med. [Internet]. Elsevier; 2015;24:e120. Available from: doi:10.1016/j.ejim.2013.08.306
Wang L, Liu L, Hao H, Gao F, Liu X, Wang Z, et al. Myopathy with anti-signal recognition particle antibodies: clinical and histopathological features in Chinese patients. Neuromuscul Disord. 2014;24:335–41.
Zheng Y, Liu L, Wang L, Xiao J, Wang Z, Lv H, et al. Magnetic resonance imaging changes of thigh muscles in myopathy with antibodies to signal recognition particle. Rheumatology (Oxford)England. 2015;54:1017–24.
Basnayake SK, Blumbergs P, Tan JA, Roberts-Thompson PJ, Limaye V. Inflammatory myopathy with anti-SRP antibodies: case series of a South Australian cohort. Clin Rheumatol Germany. 2015;34:603–8.
Watanabe Y, Suzuki S, Nishimura H, Murata K, Kurashige T, Ikawa M, et al. Statins and myotoxic effects associated with anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase autoantibodies: an observational study in Japan. Medicine (Baltimore) USA. 2015;94:e416. Helpful paper analyzing anti-HMGCR associated IMNM in a Japanese cohort.
Allenbach Y, Benveniste O. Acquired necrotizing myopathies. Curr. Opin. Neurol. [Internet]. 2013;26:554–60. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23995277
Ramanathan S, Langguth D, Hardy TA, Garg N, Bundell C, Rojana-Udomsart A, et al. Clinical course and treatment of anti-HMGCR antibody-associated necrotizing autoimmune myopathy. Neurol Neuroimmunol neuroinflammation USA. 2015;2:e96.
Werner JL, Christopher-Stine L, Ghazarian SR, Pak KS, Kus JE, Daya NR, et al. Antibody levels correlate with creatine kinase levels and strength in anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase-associated autoimmune myopathy. Arthritis Rheum USA. 2012;64:4087–93.
Hanisch F, Müller T, Stoltenburg G, Zierz S. Unusual manifestations in two cases of necrotizing myopathy associated with SRP-antibodies. Clin Neurol Neurosurg. 2012;114:1104–6.
Tansley S, Gunawardena H. The evolving spectrum of polymyositis and dermatomyositis-moving towards clinicoserological syndromes: a critical review. Clin Rev Allergy Immunol. 2013;1–10.
Preuße C, Goebel HH, Held J, Wengert O, Scheibe F, Irlbacher K, et al. Immune-mediated necrotizing myopathy is characterized by a specific Th1-M1 polarized immune profile. Am J Pathol. 2012;181:2161–71.
Kawabata T, Komaki H, Saito T, Saito Y, Nakagawa E, Sugai K, et al. A pediatric patient with myopathy associated with antibodies to a signal recognition particle. Brain Dev. 2012;34:877–80.
Maeshima K, Kiyonaga Y, Imada C, Iwakura M, Hamasaki H, Haranaka M, et al. Successful treatment of refractory anti-signal recognition particle myopathy using abatacept. Rheumatol. (United Kingdom). 2014:379–80.
Naert E, De Bleecker JL, Lumen N, Rottey S. Necrotizing myopathy as a paraneoplastic syndrome associated with renal cell carcinoma. Acta Clin Belg England. 2015;70:61–4.
Levine TD. Rituximab in the treatment of dermatomyositis: an open-label pilot study. Arthritis Rheum. 2005;52:601–7.
Sultan SM, Ng KP, Edwards JC, Isenberg DA, Cambridge G, Edwards JCW. Clinical outcome following B cell depletion therapy in eight patients with refractory idiopathic inflammatory myopathy. Clin Exp Rheumatol. 2008;26:887–93.
Lambotte O, Kotb R, Maigne G, Blanc FX, Goujard C, Delfraissy JF. Efficacy of rituximab in refractory polymyositis. J Rheumatol. 2005;32:1369–70.
Valiyil R, Casciola-Rosen L, Hong G, Mammen A, Christopher-Stine L. Rituximab therapy for myopathy associated with anti-signal recognition particle antibodies: a case series. Arthritis Care Res. 2010;62:1328–34.
Whelan BR, Isenberg DA. Poor response of anti-SRP-positive idiopathic immune myositis to B-cell depletion. Rheumatology (Oxford) England. 2009:594–5.
Arlet J-B, Dimitri D, Pagnoux C, Boyer O, Maisonobe T, Authier F-J, et al. Marked efficacy of a therapeutic strategy associating prednisone and plasma exchange followed by rituximab in two patients with refractory myopathy associated with antibodies to the signal recognition particle (SRP). Neuromuscul Disord England. 2006;16:334–6.
Needham M, Fabian V, Knezevic W, Panegyres P, Zilko P, Mastaglia FL. Progressive myopathy with up-regulation of MHC-I associated with statin therapy. Neuromuscul Disord. 2007;17:194–200.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Christopher-Stine reports personal fees from Walgreens, Novartis, Idera Pharmaceuticals, Ono Pharma UK, Marathon Pharmaceuticals, MedImmune/Astra Zeneca, and Mallinckrodt. In addition, Dr. Christopher-Stine has a patent HMGCR assay with royalties paid to Inova Diagnostics.
Dr. Basharat reports personal fees as a round-table participant for Novartis and Amgen.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Inflammatory Muscle Disease
Rights and permissions
About this article
Cite this article
Basharat, P., Christopher-Stine, L. Immune-Mediated Necrotizing Myopathy: Update on Diagnosis and Management. Curr Rheumatol Rep 17, 72 (2015). https://doi.org/10.1007/s11926-015-0548-6
Published:
DOI: https://doi.org/10.1007/s11926-015-0548-6