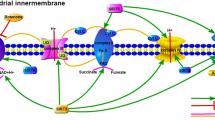
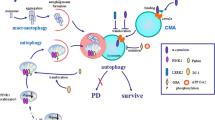
Abstract
Parkinson’s disease (PD) is one of the most common degenerative disorders of the central nervous system that produces motor and non-motor symptoms. The majority of cases are idiopathic and characterized by the presence of Lewy bodies containing fibrillar α-synuclein. Small ubiquitin-related modifier (SUMO) immunoreactivity was observed among others in cases with PD. Key disease-associated proteins are SUMO-modified, linking this posttranslational modification to neurodegeneration. SUMOylation and SUMO-mediated mechanisms have been intensively studied in recent years, revealing nuclear and extranuclear functions for SUMO in a variety of cellular processes, including the regulation of transcriptional activity, modulation of signal transduction pathways, and response to cellular stress. This points to a role for SUMO more than just an antagonist to ubiquitin and proteasomal degradation. The identification of risk and age-at-onset gene loci was a breakthrough in PD and promoted the understanding of molecular mechanisms in the pathology. PD has been increasingly linked with mitochondrial dysfunction and impaired mitochondrial quality control. Interestingly, SUMO is involved in many of these processes and up-regulated in response to cellular stress, further emphasizing the importance of SUMOylation in physiology and disease.


Similar content being viewed by others
References
Abbas, N., Lucking, C. B., Ricard, S., Durr, A., Bonifati, V., De Michele, G., et al. (1999). A wide variety of mutations in the parkin gene are responsible for autosomal recessive parkinsonism in Europe. French Parkinson’s Disease Genetics Study Group and the European Consortium on Genetic Susceptibility in Parkinson’s Disease. Human Molecular Genetics, 8(4), 567–574.
Akhtar, M. W., Sunico, C. R., Nakamura, T., & Lipton, S. A. (2012). Redox regulation of protein function via cysteine S-Nitrosylation and its relevance to neurodegenerative diseases. International Journal of Cell Biology, 2012, 463756.
Alves da Costa, C., & Checler, F. (2011). Apoptosis in Parkinson’s disease: Is p53 the missing link between genetic and sporadic Parkinsonism? Cellular Signalling, 23(6), 963–968.
Anderson, D. D., Eom, J. Y., & Stover, P. J. (2012). Competition between sumoylation and ubiquitination of serine hydroxymethyltransferase 1 determines its nuclear localization and its accumulation in the nucleus. The Journal of Biological Chemistry, 287(7), 4790–4799.
Andres-Mateos, E., Perier, C., Zhang, L., Blanchard-Fillion, B., Greco, T. M., Thomas, B., et al. (2007). DJ-1 gene deletion reveals that DJ-1 is an atypical peroxiredoxin-like peroxidase. Proceedings of the National Academy of Sciences of the United States of America, 104(37), 14807–14812.
Bandopadhyay, R., Kingsbury, A. E., Cookson, M. R., Reid, A. R., Evans, I. M., Hope, A. D., et al. (2004). The expression of DJ-1 (PARK7) in normal human CNS and idiopathic Parkinson’s disease. Brain: A Journal of Neurology, 127(Pt 2), 420–430.
Bandopadhyay, R., Kingsbury, A. E., Muqit, M. M., Harvey, K., Reid, A. R., Kilford, L., et al. (2005). Synphilin-1 and parkin show overlapping expression patterns in human brain and form aggresomes in response to proteasomal inhibition. Neurobiology of Disease, 20(2), 401–411.
Becker, J., Barysch, S. V., Karaca, S., Dittner, C., Hsiao, H. H., Berriel Diaz, M., et al. (2013). Detecting endogenous SUMO targets in mammalian cells and tissues. Nature Structural & Molecular Biology, 20(4), 525–531.
Becker, D., Richter, J., Tocilescu, M. A., Przedborski, S., & Voos, W. (2012). Pink1 kinase and its membrane potential (Deltapsi)-dependent cleavage product both localize to outer mitochondrial membrane by unique targeting mode. The Journal of Biological Chemistry, 287(27), 22969–22987.
Beilina, A., Van Der Brug, M., Ahmad, R., Kesavapany, S., Miller, D. W., Petsko, G. A., et al. (2005). Mutations in PTEN-induced putative kinase 1 associated with recessive parkinsonism have differential effects on protein stability. Proceedings of the National Academy of Sciences of the United States of America, 102(16), 5703–5708.
Betarbet, R., Sherer, T. B., MacKenzie, G., Garcia-Osuna, M., Panov, A. V., & Greenamyre, J. T. (2000). Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nature Neuroscience, 3(12), 1301–1306.
Beyer, K. (2006). Alpha-synuclein structure, posttranslational modification and alternative splicing as aggregation enhancers. Acta Neuropathologica, 112(3), 237–251.
Beyer, K., Domingo-Sabat, M., Humbert, J., Carrato, C., Ferrer, I., & Ariza, A. (2008). Differential expression of alpha-synuclein, parkin, and synphilin-1 isoforms in Lewy body disease. Neurogenetics, 9(3), 163–172.
Blackinton, J. G., Anvret, A., Beilina, A., Olson, L., Cookson, M. R., & Galter, D. (2007). Expression of PINK1 mRNA in human and rodent brain and in Parkinson’s disease. Brain Research, 1184, 10–16.
Bonifati, V., Rizzu, P., van Baren, M. J., Schaap, O., Breedveld, G. J., Krieger, E., et al. (2003). Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science, 299(5604), 256–259.
Bossis, G., & Melchior, F. (2006). Regulation of SUMOylation by reversible oxidation of SUMO conjugating enzymes. Molecular Cell, 21(3), 349–357.
Botella, J. A., Bayersdorfer, F., Gmeiner, F., & Schneuwly, S. (2009). Modelling Parkinson’s disease in Drosophila. NeuroMolecular Medicine, 11(4), 268–280.
Braak, H., Del Tredici, K., Rub, U., de Vos, R. A., Jansen Steur, E. N., & Braak, E. (2003). Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiology of Aging, 24(2), 197–211.
Bretaud, S., Allen, C., Ingham, P. W., & Bandmann, O. (2007). p53-dependent neuronal cell death in a DJ-1-deficient zebrafish model of Parkinson’s disease. Journal of Neurochemistry, 100(6), 1626–1635.
Bus, J. S., & Gibson, J. E. (1984). Paraquat: Model for oxidant-initiated toxicity. Environmental Health Perspectives, 55, 37–46.
Buschmann, T., Lerner, D., Lee, C. G., & Ronai, Z. (2001). The Mdm-2 amino terminus is required for Mdm2 binding and SUMO-1 conjugation by the E2 SUMO-1 conjugating enzyme Ubc9. The Journal of Biological Chemistry, 276(44), 40389–40395.
Canet-Aviles, R. M., Wilson, M. A., Miller, D. W., Ahmad, R., McLendon, C., Bandyopadhyay, S., et al. (2004). The Parkinson’s disease protein DJ-1 is neuroprotective due to cysteine-sulfinic acid-driven mitochondrial localization. Proceedings of the National Academy of Sciences of the United States of America, 101(24), 9103–9108.
Chandra, S., Fornai, F., Kwon, H. B., Yazdani, U., Atasoy, D., Liu, X., et al. (2004). Double-knockout mice for alpha- and beta-synucleins: Effect on synaptic functions. Proceedings of the National Academy of Sciences of the United States of America, 101(41), 14966–14971.
Chandra, S., Gallardo, G., Fernandez-Chacon, R., Schluter, O. M., & Sudhof, T. C. (2005). Alpha-synuclein cooperates with CSPalpha in preventing neurodegeneration. Cell, 123(3), 383–396.
Chang, C. C., Lin, D. Y., Fang, H. I., Chen, R. H., & Shih, H. M. (2005). Daxx mediates the small ubiquitin-like modifier-dependent transcriptional repression of Smad4. The Journal of Biological Chemistry, 280(11), 10164–10173.
Chang, C. C., Naik, M. T., Huang, Y. S., Jeng, J. C., Liao, P. H., Kuo, H. Y., et al. (2011). Structural and functional roles of Daxx SIM phosphorylation in SUMO paralog-selective binding and apoptosis modulation. Molecular Cell, 42(1), 62–74.
Chang, H. Y., Nishitoh, H., Yang, X., Ichijo, H., & Baltimore, D. (1998). Activation of apoptosis signal-regulating kinase 1 (ASK1) by the adapter protein Daxx. Science, 281(5384), 1860–1863.
Chao, H. W., Hong, C. J., Huang, T. N., Lin, Y. L., & Hsueh, Y. P. (2008). SUMOylation of the MAGUK protein CASK regulates dendritic spinogenesis. The Journal of Cell Biology, 182(1), 141–155.
Chartier-Harlin, M. C., Kachergus, J., Roumier, C., Mouroux, V., Douay, X., Lincoln, S., et al. (2004). Alpha-synuclein locus duplication as a cause of familial Parkinson’s disease. Lancet, 364(9440), 1167–1169.
Chen, H., & Chan, D. C. (2009). Mitochondrial dynamics–fusion, fission, movement, and mitophagy–in neurodegenerative diseases. Human molecular genetics, 18(R2), R169–176.
Chen, L., & Chen, J. (2003). MDM2-ARF complex regulates p53 sumoylation. Oncogene, 22(34), 5348–5357.
Chen, D., Gao, F., Li, B., Wang, H., Xu, Y., Zhu, C., et al. (2010a). Parkin mono-ubiquitinates Bcl-2 and regulates autophagy. The Journal of Biological Chemistry, 285(49), 38214–38223.
Chen, J., Li, L., & Chin, L. S. (2010b). Parkinson disease protein DJ-1 converts from a zymogen to a protease by carboxyl-terminal cleavage. Human Molecular Genetics, 19(12), 2395–2408.
Chen, J., Lin, J., & Levine, A. J. (1995). Regulation of transcription functions of the p53 tumor suppressor by the mdm-2 oncogene. Molecular Medicine, 1(2), 142–152.
Chen, A., Wang, P. Y., Yang, Y. C., Huang, Y. H., Yeh, J. J., Chou, Y. H., et al. (2006). SUMO regulates the cytoplasmonuclear transport of its target protein Daxx. Journal of Cellular Biochemistry, 98(4), 895–911.
Cheng, J., Kang, X., Zhang, S., & Yeh, E. T. (2007). SUMO-specific protease 1 is essential for stabilization of HIF1alpha during hypoxia. Cell, 131(3), 584–595.
Cheng, J., Wang, D., Wang, Z., & Yeh, E. T. (2004). SENP1 enhances androgen receptor-dependent transcription through desumoylation of histone deacetylase 1. Molecular and Cellular Biology, 24(13), 6021–6028.
Choi, S. J., Chung, S. S., Rho, E. J., Lee, H. W., Lee, M. H., Choi, H. S., et al. (2006). Negative modulation of RXRalpha transcriptional activity by small ubiquitin-related modifier (SUMO) modification and its reversal by SUMO-specific protease SUSP1. The Journal of Biological Chemistry, 281(41), 30669–30677.
Choi, P., Ostrerova-Golts, N., Sparkman, D., Cochran, E., Lee, J. M., & Wolozin, B. (2000). Parkin is metabolized by the ubiquitin/proteosome system. NeuroReport, 11(12), 2635–2638.
Chu, Y., & Yang, X. (2011). SUMO E3 ligase activity of TRIM proteins. Oncogene, 30(9), 1108–1116.
Chung, S. S., Ahn, B. Y., Kim, M., Kho, J. H., Jung, H. S., & Park, K. S. (2011). SUMO modification selectively regulates transcriptional activity of peroxisome-proliferator-activated receptor gamma in C2C12 myotubes. The Biochemical Journal, 433(1), 155–161.
Chung, K. K., Zhang, Y., Lim, K. L., Tanaka, Y., Huang, H., Gao, J., et al. (2001). Parkin ubiquitinates the alpha-synuclein-interacting protein, synphilin-1: Implications for Lewy-body formation in Parkinson disease. Nature Medicine, 7(10), 1144–1150.
Cleeter, M. W., Cooper, J. M., & Schapira, A. H. (1992). Irreversible inhibition of mitochondrial complex I by 1-methyl-4-phenylpyridinium: Evidence for free radical involvement. Journal of Neurochemistry, 58(2), 786–789.
Cookson, M. R., Lockhart, P. J., McLendon, C., O’Farrell, C., Schlossmacher, M., & Farrer, M. J. (2003). RING finger 1 mutations in Parkin produce altered localization of the protein. Human Molecular Genetics, 12(22), 2957–2965.
Croxton, R., Puto, L. A., de Belle, I., Thomas, M., Torii, S., Hanaii, F., et al. (2006). Daxx represses expression of a subset of antiapoptotic genes regulated by nuclear factor-kappaB. Cancer Research, 66(18), 9026–9035.
Cuervo, A. M., Wong, E. S., & Martinez-Vicente, M. (2010). Protein degradation, aggregation, and misfolding. Movement Disorders, 25(Suppl 1), S49–S54.
Dagata, V., & Cavallaro, S. (2004). Parkin transcript variants in rat and human brain. Neurochemical Research, 29(9), 1715–1724.
David, G., Neptune, M. A., & DePinho, R. A. (2002). SUMO-1 modification of histone deacetylase 1 (HDAC1) modulates its biological activities. The Journal of Biological Chemistry, 277(26), 23658–23663.
Davidson, B., Hadar, R., Schlossberg, A., Sternlicht, T., Slipicevic, A., Skrede, M., et al. (2008). Expression and clinical role of DJ-1, a negative regulator of PTEN, in ovarian carcinoma. Human Pathology, 39(1), 87–95.
Davis, G. C., Williams, A. C., Markey, S. P., Ebert, M. H., Caine, E. D., Reichert, C. M., et al. (1979). Chronic Parkinsonism secondary to intravenous injection of meperidine analogues. Psychiatry Research, 1(3), 249–254.
de la Vega, L., Grishina, I., Moreno, R., Kruger, M., Braun, T., & Schmitz, M. L. (2012). A redox-regulated SUMO/acetylation switch of HIPK2 controls the survival threshold to oxidative stress. Molecular Cell, 46(4), 472–483.
Dedmon, M. M., Christodoulou, J., Wilson, M. R., & Dobson, C. M. (2005). Heat shock protein 70 inhibits alpha-synuclein fibril formation via preferential binding to prefibrillar species. The Journal of Biological Chemistry, 280(15), 14733–14740.
Deeg, S., Gralle, M., Sroka, K., Bahr, M., Wouters, F. S., & Kermer, P. (2010). BAG1 restores formation of functional DJ-1 L166P dimers and DJ-1 chaperone activity. The Journal of Cell Biology, 188(4), 505–513.
Dekker, M. C., Eshuis, S. A., Maguire, R. P., Veenma-van der Duijn, L., Pruim, J., Snijders, P. J., et al. (2004). PET neuroimaging and mutations in the DJ-1 gene. Journal of Neural Transmission, 111(12), 1575–1581.
Desterro, J. M., Rodriguez, M. S., & Hay, R. T. (1998). SUMO-1 modification of IkappaBalpha inhibits NF-kappaB activation. Molecular Cell, 2(2), 233–239.
Dorval, V., & Fraser, P. E. (2006). Small ubiquitin-like modifier (SUMO) modification of natively unfolded proteins tau and alpha-synuclein. The Journal of Biological Chemistry, 281(15), 9919–9924.
Doss-Pepe, E. W., Chen, L., & Madura, K. (2005). Alpha-synuclein and parkin contribute to the assembly of ubiquitin lysine 63-linked multiubiquitin chains. The Journal of Biological Chemistry, 280(17), 16619–16624.
Elfferich, P., Verleun-Mooijman, M. C., Maat-Kievit, J. A., van de Warrenburg, B. P., Abdo, W. F., Eshuis, S. A., et al. (2011). Breakpoint mapping of 13 large parkin deletions/duplications reveals an exon 4 deletion and an exon 7 duplication as founder mutations. Neurogenetics, 12(4), 263–271.
Engelender, S., Kaminsky, Z., Guo, X., Sharp, A. H., Amaravi, R. K., Kleiderlein, J. J., et al. (1999). Synphilin-1 associates with alpha-synuclein and promotes the formation of cytosolic inclusions. Nature Genetics, 22(1), 110–114.
Errico, F., Santini, E., Migliarini, S., Borgkvist, A., Centonze, D., Nasti, V., et al. (2008). The GTP-binding protein Rhes modulates dopamine signalling in striatal medium spiny neurons. Molecular and Cellular Neurosciences, 37(2), 335–345.
Escobar-Cabrera, E., Okon, M., Lau, D. K., Dart, C. F., Bonvin, A. M., & McIntosh, L. P. (2011). Characterizing the N- and C-terminal Small ubiquitin-like modifier (SUMO)-interacting motifs of the scaffold protein DAXX. The Journal of Biological Chemistry, 286(22), 19816–19829.
Fallon, L., Moreau, F., Croft, B. G., Labib, N., Gu, W. J., & Fon, E. A. (2002). Parkin and CASK/LIN-2 associate via a PDZ-mediated interaction and are co-localized in lipid rafts and postsynaptic densities in brain. The Journal of Biological Chemistry, 277(1), 486–491.
Farrer, M., Gwinn-Hardy, K., Muenter, M., DeVrieze, F. W., Crook, R., Perez-Tur, J., et al. (1999). A chromosome 4p haplotype segregating with Parkinson’s disease and postural tremor. Human Molecular Genetics, 8(1), 81–85.
Figueroa-Romero, C., Iniguez-Lluhi, J. A., Stadler, J., Chang, C. R., Arnoult, D., Keller, P. J., et al. (2009). SUMOylation of the mitochondrial fission protein Drp1 occurs at multiple nonconsensus sites within the B domain and is linked to its activity cycle. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, 23(11), 3917–3927.
Foti, R., Zucchelli, S., Biagioli, M., Roncaglia, P., Vilotti, S., Calligaris, R., et al. (2010). Parkinson disease-associated DJ-1 is required for the expression of the glial cell line-derived neurotrophic factor receptor RET in human neuroblastoma cells. The Journal of Biological Chemistry, 285(24), 18565–18574.
Fujiwara, H., Hasegawa, M., Dohmae, N., Kawashima, A., Masliah, E., Goldberg, M. S., et al. (2002). alpha-Synuclein is phosphorylated in synucleinopathy lesions. Nature Cell Biology, 4(2), 160–164.
Galter, D., Westerlund, M., Belin, A. C., & Olson, L. (2007). DJ-1 and UCH-L1 gene activity patterns in the brains of controls, Parkinson and schizophrenia patients and in rodents. Physiology & Behavior, 92(1–2), 46–53.
Geisler, S., Holmstrom, K. M., Skujat, D., Fiesel, F. C., Rothfuss, O. C., Kahle, P. J., et al. (2010). PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nature Cell Biology, 12(2), 119–131.
Geiss-Friedlander, R., & Melchior, F. (2007). Concepts in sumoylation: A decade on. Nature Reviews Molecular Cell Biology, 8(12), 947–956.
Giasson, B. I., Duda, J. E., Murray, I. V., Chen, Q., Souza, J. M., Hurtig, H. I., et al. (2000). Oxidative damage linked to neurodegeneration by selective alpha-synuclein nitration in synucleinopathy lesions. Science, 290(5493), 985–989.
Girdwood, D., Bumpass, D., Vaughan, O. A., Thain, A., Anderson, L. A., Snowden, A. W., et al. (2003). P300 transcriptional repression is mediated by SUMO modification. Molecular Cell, 11(4), 1043–1054.
Goedert, M., Spillantini, M. G., Del Tredici, K., & Braak, H. (2013). 100 years of Lewy pathology. Nature Reviews. Neurology, 9(1), 13–24.
Gorner, K., Holtorf, E., Waak, J., Pham, T. T., Vogt-Weisenhorn, D. M., Wurst, W., et al. (2007). Structural determinants of the C-terminal helix-kink-helix motif essential for protein stability and survival promoting activity of DJ-1. The Journal of Biological Chemistry, 282(18), 13680–13691.
Gosavi, N., Lee, H. J., Lee, J. S., Patel, S., & Lee, S. J. (2002). Golgi fragmentation occurs in the cells with prefibrillar alpha-synuclein aggregates and precedes the formation of fibrillar inclusion. The Journal of Biological Chemistry, 277(50), 48984–48992.
Gostissa, M., Hengstermann, A., Fogal, V., Sandy, P., Schwarz, S. E., Scheffner, M., et al. (1999). Activation of p53 by conjugation to the ubiquitin-like protein SUMO-1. The EMBO Journal, 18(22), 6462–6471.
Greene, A. W., Grenier, K., Aguileta, M. A., Muise, S., Farazifard, R., Haque, M. E., et al. (2012). Mitochondrial processing peptidase regulates PINK1 processing, import and Parkin recruitment. EMBO Reports, 13(4), 378–385.
Harder, Z., Zunino, R., & McBride, H. (2004). Sumo1 conjugates mitochondrial substrates and participates in mitochondrial fission. Current Biology: CB, 14(4), 340–345.
Hata, Y., Butz, S., & Sudhof, T. C. (1996). CASK: A novel dlg/PSD95 homolog with an N-terminal calmodulin-dependent protein kinase domain identified by interaction with neurexins. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 16(8), 2488–2494.
Hattori, N., Kitada, T., Matsumine, H., Asakawa, S., Yamamura, Y., Yoshino, H., et al. (1998). Molecular genetic analysis of a novel Parkin gene in Japanese families with autosomal recessive juvenile parkinsonism: Evidence for variable homozygous deletions in the Parkin gene in affected individuals. Annals of Neurology, 44(6), 935–941.
Haupt, Y., Maya, R., Kazaz, A., & Oren, M. (1997). Mdm2 promotes the rapid degradation of p53. Nature, 387(6630), 296–299.
Hauser, D. N., & Hastings, T. G. (2013). Mitochondrial dysfunction and oxidative stress in Parkinson’s disease and monogenic parkinsonism. Neurobiology of disease, 51, 35–42.
Hecker, C. M., Rabiller, M., Haglund, K., Bayer, P., & Dikic, I. (2006). Specification of SUMO1- and SUMO2-interacting motifs. The Journal of Biological Chemistry, 281(23), 16117–16127.
Heikkila, R. E., Nicklas, W. J., Vyas, I., & Duvoisin, R. C. (1985). Dopaminergic toxicity of rotenone and the 1-methyl-4-phenylpyridinium ion after their stereotaxic administration to rats: Implication for the mechanism of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine toxicity. Neuroscience Letters, 62(3), 389–394.
Henn, I. H., Bouman, L., Schlehe, J. S., Schlierf, A., Schramm, J. E., Wegener, E., et al. (2007). Parkin mediates neuroprotection through activation of IkappaB kinase/nuclear factor-kappaB signaling. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 27(8), 1868–1878.
Hietakangas, V., Anckar, J., Blomster, H. A., Fujimoto, M., Palvimo, J. J., Nakai, A., et al. (2006). PDSM, a motif for phosphorylation-dependent SUMO modification. Proceedings of the National Academy of Sciences of the United States of America, 103(1), 45–50.
Hod, Y., Pentyala, S. N., Whyard, T. C., & El-Maghrabi, M. R. (1999). Identification and characterization of a novel protein that regulates RNA-protein interaction. Journal of Cellular Biochemistry, 72(3), 435–444.
Hoege, C., Pfander, B., Moldovan, G. L., Pyrowolakis, G., & Jentsch, S. (2002). RAD6-dependent DNA repair is linked to modification of PCNA by ubiquitin and SUMO. Nature, 419(6903), 135–141.
Honda, R., Tanaka, H., & Yasuda, H. (1997). Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Letters, 420(1), 25–27.
Huang, J., Yan, J., Zhang, J., Zhu, S., Wang, Y., Shi, T., et al. (2012). SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane. Nature Communications, 3, 911.
Hwang, S., Song, S., Hong, Y. K., Choi, G., Suh, Y. S., Han, S. Y., et al. (2013). Drosophila DJ-1 decreases neural sensitivity to stress by negatively regulating Daxx-like protein through dFOXO. PLoS Genetics, 9(4), e1003412.
Imai, Y., Soda, M., Inoue, H., Hattori, N., Mizuno, Y., & Takahashi, R. (2001). An unfolded putative transmembrane polypeptide, which can lead to endoplasmic reticulum stress, is a substrate of Parkin. Cell, 105(7), 891–902.
Ishov, A. M., Sotnikov, A. G., Negorev, D., Vladimirova, O. V., Neff, N., Kamitani, T., et al. (1999). PML is critical for ND10 formation and recruits the PML-interacting protein daxx to this nuclear structure when modified by SUMO-1. The Journal of Cell Biology, 147(2), 221–234.
Jakes, R., Spillantini, M. G., & Goedert, M. (1994). Identification of two distinct synucleins from human brain. FEBS Letters, 345(1), 27–32.
Jang, M. S., Ryu, S. W., & Kim, E. (2002). Modification of Daxx by small ubiquitin-related modifier-1. Biochemical and Biophysical Research Communications, 295(2), 495–500.
Jang, H. D., Yoon, K., Shin, Y. J., Kim, J., & Lee, S. Y. (2004). PIAS3 suppresses NF-kappaB-mediated transcription by interacting with the p65/RelA subunit. The Journal of Biological Chemistry, 279(23), 24873–24880.
Javitch, J. A., D’Amato, R. J., Strittmatter, S. M., & Snyder, S. H. (1985). Parkinsonism-inducing neurotoxin, N-methyl-4-phenyl-1,2,3,6 -tetrahydropyridine: Uptake of the metabolite N-methyl-4-phenylpyridine by dopamine neurons explains selective toxicity. Proceedings of the National Academy of Sciences of the United States of America, 82(7), 2173–2177.
Jeong, H., Kim, M. S., Kwon, J., Kim, K. S., & Seol, W. (2006). Regulation of the transcriptional activity of the tyrosine hydroxylase gene by androgen receptor. Neuroscience Letters, 396(1), 57–61.
Jiang, Q., Ren, Y., & Feng, J. (2008). Direct binding with histone deacetylase 6 mediates the reversible recruitment of parkin to the centrosome. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 28(48), 12993–13002.
Junn, E., Taniguchi, H., Jeong, B. S., Zhao, X., Ichijo, H., & Mouradian, M. M. (2005). Interaction of DJ-1 with Daxx inhibits apoptosis signal-regulating kinase 1 activity and cell death. Proceedings of the National Academy of Sciences of the United States of America, 102(27), 9691–9696.
Kalia, L. V., Kalia, S. K., McLean, P. J., Lozano, A. M., & Lang, A. E. (2013). alpha-Synuclein oligomers and clinical implications for Parkinson disease. Annals of Neurology, 73(2), 155–169.
Kang, L., Moriarty, G. M., Woods, L. A., Ashcroft, A. E., Radford, S. E., & Baum, J. (2012). N-terminal acetylation of alpha-synuclein induces increased transient helical propensity and decreased aggregation rates in the intrinsically disordered monomer. Protein Science: A Publication of the Protein Society, 21(7), 911–917.
Kawamata, H., McLean, P. J., Sharma, N., & Hyman, B. T. (2001). Interaction of alpha-synuclein and synphilin-1: Effect of Parkinson’s disease-associated mutations. Journal of Neurochemistry, 77(3), 929–934.
Khelifi, A. F., D’Alcontres, M. S., & Salomoni, P. (2005). Daxx is required for stress-induced cell death and JNK activation. Cell Death and Differentiation, 12(7), 724–733.
Kim, Y. M., Jang, W. H., Quezado, M. M., Oh, Y., Chung, K. C., Junn, E., et al. (2011). Proteasome inhibition induces alpha-synuclein SUMOylation and aggregate formation. Journal of the Neurological Sciences, 307(1–2), 157–161.
Kim, R. H., Peters, M., Jang, Y., Shi, W., Pintilie, M., Fletcher, G. C., et al. (2005). DJ-1, a novel regulator of the tumor suppressor PTEN. Cancer Cell, 7(3), 263–273.
Kim, J. M., Shin, H. I., Cha, S. S., Lee, C. S., Hong, B. S., Lim, S., et al. (2012). DJ-1 promotes angiogenesis and osteogenesis by activating FGF receptor-1 signaling. Nature Communications, 3, 1296.
Kinumi, T., Kimata, J., Taira, T., Ariga, H., & Niki, E. (2004). Cysteine-106 of DJ-1 is the most sensitive cysteine residue to hydrogen peroxide-mediated oxidation in vivo in human umbilical vein endothelial cells. Biochemical and Biophysical Research Communications, 317(3), 722–728.
Kirsh, O., Seeler, J. S., Pichler, A., Gast, A., Muller, S., Miska, E., et al. (2002). The SUMO E3 ligase RanBP2 promotes modification of the HDAC4 deacetylase. The EMBO Journal, 21(11), 2682–2691.
Kitada, T., Asakawa, S., Hattori, N., Matsumine, H., Yamamura, Y., Minoshima, S., et al. (1998). Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature, 392(6676), 605–608.
Kitada, T., Asakawa, S., Minoshima, S., Mizuno, Y., & Shimizu, N. (2000). Molecular cloning, gene expression, and identification of a splicing variant of the mouse parkin gene. Mammalian Genome: Official Journal of the International Mammalian Genome Society, 11(6), 417–421.
Kitao, Y., Imai, Y., Ozawa, K., Kataoka, A., Ikeda, T., Soda, M., et al. (2007). Pael receptor induces death of dopaminergic neurons in the substantia nigra via endoplasmic reticulum stress and dopamine toxicity, which is enhanced under condition of parkin inactivation. Human Molecular Genetics, 16(1), 50–60.
Kruger, R., Kuhn, W., Muller, T., Woitalla, D., Graeber, M., Kosel, S., et al. (1998). Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease. Nature Genetics, 18(2), 106–108.
Krumova, P., Meulmeester, E., Garrido, M., Tirard, M., Hsiao, H. H., Bossis, G., et al. (2011). Sumoylation inhibits alpha-synuclein aggregation and toxicity. The Journal of Cell Biology, 194(1), 49–60.
Kubbutat, M. H., Jones, S. N., & Vousden, K. H. (1997). Regulation of p53 stability by Mdm2. Nature, 387(6630), 299–303.
Kubo, S. I., Kitami, T., Noda, S., Shimura, H., Uchiyama, Y., Asakawa, S., et al. (2001). Parkin is associated with cellular vesicles. Journal of Neurochemistry, 78(1), 42–54.
Kumaran, R., Vandrovcova, J., Luk, C., Sharma, S., Renton, A., Wood, N. W., et al. (2009). Differential DJ-1 gene expression in Parkinson’s disease. Neurobiology of Disease, 36(2), 393–400.
Kuo, H. Y., Chang, C. C., Jeng, J. C., Hu, H. M., Lin, D. Y., Maul, G. G., et al. (2005). SUMO modification negatively modulates the transcriptional activity of CREB-binding protein via the recruitment of Daxx. Proceedings of the National Academy of Sciences of the United States of America, 102(47), 16973–16978.
Lamoliatte, F., Bonneil, E., Durette, C., Caron-Lizotte, O., Wildemann, D., Zerweck, J., et al. (2013). Targeted identification of SUMOylation sites in human proteins using affinity enrichment and paralog-specific reporter ions. Molecular & cellular proteomics: MCP
Langston, J. W., Ballard, P., Tetrud, J. W., & Irwin, I. (1983). Chronic Parkinsonism in humans due to a product of meperidine-analog synthesis. Science, 219(4587), 979–980.
Lee, M. H., Lee, S. W., Lee, E. J., Choi, S. J., Chung, S. S., Lee, J. I., et al. (2006). SUMO-specific protease SUSP4 positively regulates p53 by promoting Mdm2 self-ubiquitination. Nature Cell Biology, 8(12), 1424–1431.
Lehembre, F., Muller, S., Pandolfi, P. P., & Dejean, A. (2001). Regulation of Pax3 transcriptional activity by SUMO-1-modified PML. Oncogene, 20(1), 1–9.
Leroy, E., Anastasopoulos, D., Konitsiotis, S., Lavedan, C., & Polymeropoulos, M. H. (1998). Deletions in the Parkin gene and genetic heterogeneity in a Greek family with early onset Parkinson’s disease. Human Genetics, 103(4), 424–427.
Li, H., Leo, C., Zhu, J., Wu, X., O’Neil, J., Park, E. J., et al. (2000). Sequestration and inhibition of Daxx-mediated transcriptional repression by PML. Molecular and Cellular Biology, 20(5), 1784–1796.
Li, H. M., Niki, T., Taira, T., Iguchi-Ariga, S. M., & Ariga, H. (2005). Association of DJ-1 with chaperones and enhanced association and colocalization with mitochondrial Hsp70 by oxidative stress. Free Radical Research, 39(10), 1091–1099.
Li, R., Wei, J., Jiang, C., Liu, D., Deng, L., Zhang, K., et al. (2013). Akt SUMOylation regulates cell proliferation and tumorigenesis. Cancer research.
Lim, K. L., Chew, K. C., Tan, J. M., Wang, C., Chung, K. K., Zhang, Y., et al. (2005). Parkin mediates nonclassical, proteasomal-independent ubiquitination of synphilin-1: implications for Lewy body formation. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 25(8), 2002–2009.
Lin, D. Y., Huang, Y. S., Jeng, J. C., Kuo, H. Y., Chang, C. C., Chao, T. T., et al. (2006). Role of SUMO-interacting motif in Daxx SUMO modification, subnuclear localization, and repression of sumoylated transcription factors. Molecular Cell, 24(3), 341–354.
Lin, W., & Kang, U. J. (2008). Characterization of PINK1 processing, stability, and subcellular localization. Journal of Neurochemistry, 106(1), 464–474.
Mabb, A. M., Wuerzberger-Davis, S. M., & Miyamoto, S. (2006). PIASy mediates NEMO sumoylation and NF-kappaB activation in response to genotoxic stress. Nature Cell Biology, 8(9), 986–993.
Mahajan, R., Delphin, C., Guan, T., Gerace, L., & Melchior, F. (1997). A small ubiquitin-related polypeptide involved in targeting RanGAP1 to nuclear pore complex protein RanBP2. Cell, 88(1), 97–107.
Mai, S., Klinkenberg, M., Auburger, G., Bereiter-Hahn, J., & Jendrach, M. (2010). Decreased expression of Drp1 and Fis1 mediates mitochondrial elongation in senescent cells and enhances resistance to oxidative stress through PINK1. Journal of Cell Science, 123(Pt 6), 917–926.
Malik, B. R., Gillespie, J. M., & Hodge, J. J. (2013). CASK and CaMKII function in the mushroom body alpha’/beta’ neurons during Drosophila memory formation. Frontiers in Neural Circuits, 7, 52.
Maroteaux, L., Campanelli, J. T., & Scheller, R. H. (1988). Synuclein: A neuron-specific protein localized to the nucleus and presynaptic nerve terminal. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 8(8), 2804–2815.
Masliah, E., Rockenstein, E., Veinbergs, I., Mallory, M., Hashimoto, M., Takeda, A., et al. (2000). Dopaminergic loss and inclusion body formation in alpha-synuclein mice: Implications for neurodegenerative disorders. Science, 287(5456), 1265–1269.
Matunis, M. J., Coutavas, E., & Blobel, G. (1996). A novel ubiquitin-like modification modulates the partitioning of the Ran-GTPase-activating protein RanGAP1 between the cytosol and the nuclear pore complex. The Journal of Cell Biology, 135(6 Pt 1), 1457–1470.
Mauri, F., McNamee, L. M., Lunardi, A., Chiacchiera, F., Del Sal, G., Brodsky, M. H., et al. (2008). Modification of Drosophila p53 by SUMO modulates its transactivation and pro-apoptotic functions. The Journal of Biological Chemistry, 283(30), 20848–20856.
McNally, R. S., Davis, B. K., Clements, C. M., Accavitti-Loper, M. A., Mak, T. W., & Ting, J. P. (2011). DJ-1 enhances cell survival through the binding of Cezanne, a negative regulator of NF-kappaB. The Journal of Biological Chemistry, 286(6), 4098–4106.
Meissner, C., Lorenz, H., Weihofen, A., Selkoe, D. J., & Lemberg, M. K. (2011). The mitochondrial intramembrane protease PARL cleaves human Pink1 to regulate Pink1 trafficking. Journal of Neurochemistry, 117(5), 856–867.
Menzies, F. M., Yenisetti, S. C., & Min, K. T. (2005). Roles of Drosophila DJ-1 in survival of dopaminergic neurons and oxidative stress. Current Biology: CB, 15(17), 1578–1582.
Meulener, M., Whitworth, A. J., Armstrong-Gold, C. E., Rizzu, P., Heutink, P., Wes, P. D., et al. (2005). Drosophila DJ-1 mutants are selectively sensitive to environmental toxins associated with Parkinson’s disease. Current Biology: CB, 15(17), 1572–1577.
Meulmeester, E., Kunze, M., Hsiao, H. H., Urlaub, H., & Melchior, F. (2008). Mechanism and consequences for paralog-specific sumoylation of ubiquitin-specific protease 25. Molecular Cell, 30(5), 610–619.
Michel, P. P., Toulorge, D., Guerreiro, S., & Hirsch, E. C. (2013). Specific needs of dopamine neurons for stimulation in order to survive: Implication for Parkinson disease. FASEB Journa: Official Publication of the Federation of American Societies for Experimental Biology.
Mizuta, H., & Kuroda, Y. (2004). Cloning and functional characterization of a rat Daxx that functions as a corepressor for the androgen receptor. Cell Biology International, 28(8–9), 609–614.
Morett, E., & Bork, P. (1999). A novel transactivation domain in parkin. Trends in Biochemical Sciences, 24(6), 229–231.
Mori, F., Tanji, K., Odagiri, S., Hattori, M., Hoshikawa, Y., Kono, C., et al. (2012). Ubiquitin-related proteins in neuronal and glial intranuclear inclusions in intranuclear inclusion body disease. Pathology International, 62(6), 407–411.
Mullett, S. J., Hamilton, R. L., & Hinkle, D. A. (2009). DJ-1 immunoreactivity in human brain astrocytes is dependent on infarct presence and infarct age. Neuropathology: Official Journal of the Japanese Society of Neuropathology, 29(2), 125–131.
Murakami, T., Shoji, M., Imai, Y., Inoue, H., Kawarabayashi, T., Matsubara, E., et al. (2004). Pael-R is accumulated in Lewy bodies of Parkinson’s disease. Annals of Neurology, 55(3), 439–442.
Nagakubo, D., Taira, T., Kitaura, H., Ikeda, M., Tamai, K., Iguchi-Ariga, S. M., et al. (1997). DJ-1, a novel oncogene which transforms mouse NIH3T3 cells in cooperation with ras. Biochemical and Biophysical Research Communications, 231(2), 509–513.
Narendra, D. P., Jin, S. M., Tanaka, A., Suen, D. F., Gautier, C. A., Shen, J., et al. (2010). PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biology, 8(1), e1000298.
Narendra, D., Tanaka, A., Suen, D. F., & Youle, R. J. (2008). Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. The Journal of Cell Biology, 183(5), 795–803.
Neumann, M., Muller, V., Gorner, K., Kretzschmar, H. A., Haass, C., & Kahle, P. J. (2004). Pathological properties of the Parkinson’s disease-associated protein DJ-1 in alpha-synucleinopathies and tauopathies: Relevance for multiple system atrophy and Pick’s disease. Acta Neuropathologica, 107(6), 489–496.
Niki, T., Takahashi-Niki, K., Taira, T., Iguchi-Ariga, S. M., & Ariga, H. (2003). DJBP: A novel DJ-1-binding protein, negatively regulates the androgen receptor by recruiting histone deacetylase complex, and DJ-1 antagonizes this inhibition by abrogation of this complex. Molecular Cancer Research: MCR, 1(4), 247–261.
Oh, Y., & Chung, K. C. (2013). UHRF2, a ubiquitin E3 ligase, acts as a small ubiquitin-like modifier E3 ligase for zinc finger protein 131. The Journal of Biological Chemistry, 288(13), 9102–9111.
Oh, Y., Kim, Y. M., Mouradian, M. M., & Chung, K. C. (2011). Human Polycomb protein 2 promotes alpha-synuclein aggregate formation through covalent SUMOylation. Brain Research, 1381, 78–89.
Ohshima, T., Koga, H., & Shimotohno, K. (2004). Transcriptional activity of peroxisome proliferator-activated receptor gamma is modulated by SUMO-1 modification. The Journal of Biological Chemistry, 279(28), 29551–29557.
Olzmann, J. A., Bordelon, J. R., Muly, E. C., Rees, H. D., Levey, A. I., Li, L., et al. (2007a). Selective enrichment of DJ-1 protein in primate striatal neuronal processes: Implications for Parkinson’s disease. The Journal of Comparative Neurology, 500(3), 585–599.
Olzmann, J. A., Li, L., Chudaev, M. V., Chen, J., Perez, F. A., Palmiter, R. D., et al. (2007b). Parkin-mediated K63-linked polyubiquitination targets misfolded DJ-1 to aggresomes via binding to HDAC6. The Journal of Cell Biology, 178(6), 1025–1038.
Park, J., Lee, J. H., La, M., Jang, M. J., Chae, G. W., Kim, S. B., et al. (2007). Inhibition of NF-kappaB acetylation and its transcriptional activity by Daxx. Journal of Molecular Biology, 368(2), 388–397.
Patel, V. P., & Chu, C. T. (2011). Nuclear transport, oxidative stress, and neurodegeneration. International Journal of Clinical and Experimental Pathology, 4(3), 215–229.
Petrucelli, L., O’Farrell, C., Lockhart, P. J., Baptista, M., Kehoe, K., Vink, L., et al. (2002). Parkin protects against the toxicity associated with mutant alpha-synuclein: Proteasome dysfunction selectively affects catecholaminergic neurons. Neuron, 36(6), 1007–1019.
Pichler, A., Gast, A., Seeler, J. S., Dejean, A., & Melchior, F. (2002). The nucleoporin RanBP2 has SUMO1 E3 ligase activity. Cell, 108(1), 109–120.
Pilsl, A., & Winklhofer, K. F. (2012). Parkin, PINK1 and mitochondrial integrity: Emerging concepts of mitochondrial dysfunction in Parkinson’s disease. Acta Neuropathologica, 123(2), 173–188.
Plun-Favreau, H., Klupsch, K., Moisoi, N., Gandhi, S., Kjaer, S., Frith, D., et al. (2007). The mitochondrial protease HtrA2 is regulated by Parkinson’s disease-associated kinase PINK1. Nature Cell Biology, 9(11), 1243–1252.
Polymeropoulos, M. H., Lavedan, C., Leroy, E., Ide, S. E., Dehejia, A., Dutra, A., et al. (1997). Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science, 276(5321), 2045–2047.
Pountney, D. L., Chegini, F., Shen, X., Blumbergs, P. C., & Gai, W. P. (2005). SUMO-1 marks subdomains within glial cytoplasmic inclusions of multiple system atrophy. Neuroscience Letters, 381(1–2), 74–79.
Pountney, D. L., Huang, Y., Burns, R. J., Haan, E., Thompson, P. D., Blumbergs, P. C., et al. (2003). SUMO-1 marks the nuclear inclusions in familial neuronal intranuclear inclusion disease. Experimental Neurology, 184(1), 436–446.
Pountney, D. L., Raftery, M. J., Chegini, F., Blumbergs, P. C., & Gai, W. P. (2008). NSF, Unc-18-1, dynamin-1 and HSP90 are inclusion body components in neuronal intranuclear inclusion disease identified by anti-SUMO-1-immunocapture. Acta Neuropathologica, 116(6), 603–614.
Pourcet, B., Pineda-Torra, I., Derudas, B., Staels, B., & Glineur, C. (2010). SUMOylation of human peroxisome proliferator-activated receptor alpha inhibits its trans-activity through the recruitment of the nuclear corepressor NCoR. The Journal of Biological Chemistry, 285(9), 5983–5992.
Pridgeon, J. W., Olzmann, J. A., Chin, L. S., & Li, L. (2007). PINK1 protects against oxidative stress by phosphorylating mitochondrial chaperone TRAP1. PLoS Biology, 5(7), e172.
Prudden, J., Pebernard, S., Raffa, G., Slavin, D. A., Perry, J. J., Tainer, J. A., et al. (2007). SUMO-targeted ubiquitin ligases in genome stability. The EMBO Journal, 26(18), 4089–4101.
Pype, S., Declercq, W., Ibrahimi, A., Michiels, C., Van Rietschoten, J. G., Dewulf, N., et al. (2000). TTRAP, a novel protein that associates with CD40, tumor necrosis factor (TNF) receptor-75 and TNF receptor-associated factors (TRAFs), and that inhibits nuclear factor-kappa B activation. The Journal of Biological Chemistry, 275(24), 18586–18593.
Rizzu, P., Hinkle, D. A., Zhukareva, V., Bonifati, V., Severijnen, L. A., Martinez, D., et al. (2004). DJ-1 colocalizes with tau inclusions: A link between parkinsonism and dementia. Annals of Neurology, 55(1), 113–118.
Rodriguez, M. S., Desterro, J. M., Lain, S., Midgley, C. A., Lane, D. P., & Hay, R. T. (1999). SUMO-1 modification activates the transcriptional response of p53. The EMBO Journal, 18(22), 6455–6461.
Rothgiesser, K. M., Fey, M., & Hottiger, M. O. (2010). Acetylation of p65 at lysine 314 is important for late NF-kappaB-dependent gene expression. BMC Genomics, 11, 22.
Saitoh, H., & Hinchey, J. (2000). Functional heterogeneity of small ubiquitin-related protein modifiers SUMO-1 versus SUMO-2/3. The Journal of Biological Chemistry, 275(9), 6252–6258.
Sandebring, A., Thomas, K. J., Beilina, A., van der Brug, M., Cleland, M. M., Ahmad, R., et al. (2009). Mitochondrial alterations in PINK1 deficient cells are influenced by calcineurin-dependent dephosphorylation of dynamin-related protein 1. PLoS One, 4(5), e5701.
Santiago, A., Godsey, A. C., Hossain, J., Zhao, L. Y., & Liao, D. (2009). Identification of two independent SUMO-interacting motifs in Daxx: Evolutionary conservation from Drosophila to humans and their biochemical functions. Cell Cycle, 8(1), 76–87.
Sayre, L. M. (1989). Biochemical mechanism of action of the dopaminergic neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Toxicology Letters, 48(2), 121–149.
Schlossmacher, M. G., Frosch, M. P., Gai, W. P., Medina, M., Sharma, N., Forno, L., et al. (2002). Parkin localizes to the Lewy bodies of Parkinson disease and dementia with Lewy bodies. The American Journal of Pathology, 160(5), 1655–1667.
Schulz-Schaeffer, W. J. (2012). Neurodegeneration in Parkinson disease: Moving Lewy bodies out of focus. Neurology, 79(24), 2298–2299.
Seliger, B., Fedorushchenko, A., Brenner, W., Ackermann, A., Atkins, D., Hanash, S., et al. (2007). Ubiquitin COOH-terminal hydrolase 1: A biomarker of renal cell carcinoma associated with enhanced tumor cell proliferation and migration. Clinical Cancer Research, 13(1), 27–37.
Sharon, R., Bar-Joseph, I., Frosch, M. P., Walsh, D. M., Hamilton, J. A., & Selkoe, D. J. (2003). The formation of highly soluble oligomers of alpha-synuclein is regulated by fatty acids and enhanced in Parkinson’s disease. Neuron, 37(4), 583–595.
Shaw, J. M., & Nunnari, J. (2002). Mitochondrial dynamics and division in budding yeast. Trends in Cell Biology, 12(4), 178–184.
Shendelman, S., Jonason, A., Martinat, C., Leete, T., & Abeliovich, A. (2004). DJ-1 is a redox-dependent molecular chaperone that inhibits alpha-synuclein aggregate formation. PLoS Biology, 2(11), e362.
Sherer, T. B., Betarbet, R., Stout, A. K., Lund, S., Baptista, M., Panov, A. V., et al. (2002). An in vitro model of Parkinson’s disease: Linking mitochondrial impairment to altered alpha-synuclein metabolism and oxidative damage. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 22(16), 7006–7015.
Sherer, T. B., Kim, J. H., Betarbet, R., & Greenamyre, J. T. (2003). Subcutaneous rotenone exposure causes highly selective dopaminergic degeneration and alpha-synuclein aggregation. Experimental Neurology, 179(1), 9–16.
Shiba-Fukushima, K., Imai, Y., Yoshida, S., Ishihama, Y., Kanao, T., Sato, S., et al. (2012). PINK1-mediated phosphorylation of the Parkin ubiquitin-like domain primes mitochondrial translocation of Parkin and regulates mitophagy. Scientific Reports, 2, 1002.
Shiio, Y., & Eisenman, R. N. (2003). Histone sumoylation is associated with transcriptional repression. Proceedings of the National Academy of Sciences of the United States of America, 100(23), 13225–13230.
Shimizu, K., Ohtaki, K., Matsubara, K., Aoyama, K., Uezono, T., Saito, O., et al. (2001). Carrier-mediated processes in blood–brain barrier penetration and neural uptake of paraquat. Brain Research, 906(1–2), 135–142.
Shimshek, D. R., Schweizer, T., Schmid, P., & van der Putten, P. H. (2012). Excess alpha-synuclein worsens disease in mice lacking ubiquitin carboxy-terminal hydrolase L1. Scientific Reports, 2, 262.
Shimura, H., Hattori, N., Kubo, S., Mizuno, Y., Asakawa, S., Minoshima, S., et al. (2000). Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nature Genetics, 25(3), 302–305.
Shimura, H., Hattori, N., Kubo, S., Yoshikawa, M., Kitada, T., Matsumine, H., et al. (1999). Immunohistochemical and subcellular localization of Parkin protein: Absence of protein in autosomal recessive juvenile parkinsonism patients. Annals of Neurology, 45(5), 668–672.
Shinbo, Y., Niki, T., Taira, T., Ooe, H., Takahashi-Niki, K., Maita, C., et al. (2006). Proper SUMO-1 conjugation is essential to DJ-1 to exert its full activities. Cell Death and Differentiation, 13(1), 96–108.
Singleton, A. B., Farrer, M., Johnson, J., Singleton, A., Hague, S., Kachergus, J., et al. (2003). alpha-Synuclein locus triplication causes Parkinson’s disease. Science, 302(5646), 841.
Spillantini, M. G., Schmidt, M. L., Lee, V. M., Trojanowski, J. Q., Jakes, R., & Goedert, M. (1997). Alpha-synuclein in Lewy bodies. Nature, 388(6645), 839–840.
Stambolic, V., Suzuki, A., de la Pompa, J. L., Brothers, G. M., Mirtsos, C., Sasaki, T., et al. (1998). Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell, 95(1), 29–39.
Steffan, J. S., Agrawal, N., Pallos, J., Rockabrand, E., Trotman, L. C., Slepko, N., et al. (2004). SUMO modification of Huntingtin and Huntington’s disease pathology. Science, 304(5667), 100–104.
Stichel, C. C., Augustin, M., Kuhn, K., Zhu, X. R., Engels, P., Ullmer, C., et al. (2000). Parkin expression in the adult mouse brain. The European Journal of Neuroscience, 12(12), 4181–4194.
Stindt, M. H., Carter, S., Vigneron, A. M., Ryan, K. M., & Vousden, K. H. (2011). MDM2 promotes SUMO-2/3 modification of p53 to modulate transcriptional activity. Cell Cycle, 10(18), 3176–3188.
Subramaniam, S., Mealer, R. G., Sixt, K. M., Barrow, R. K., Usiello, A., & Snyder, S. H. (2010). Rhes, a physiologic regulator of sumoylation, enhances cross-sumoylation between the basic sumoylation enzymes E1 and Ubc9. The Journal of Biological Chemistry, 285(27), 20428–20432.
Subramaniam, S., Sixt, K. M., Barrow, R., & Snyder, S. H. (2009). Rhes, a striatal specific protein, mediates mutant-huntingtin cytotoxicity. Science, 324(5932), 1327–1330.
Sudharsan, R., & Azuma, Y. (2012). The SUMO ligase PIAS1 regulates UV-induced apoptosis by recruiting Daxx to SUMOylated foci. Journal of Cell Science, 125(Pt 23), 5819–5829.
Sytnyk, V., Leshchyns’ka, I., Nikonenko, A. G., & Schachner, M. (2006). NCAM promotes assembly and activity-dependent remodeling of the postsynaptic signaling complex. The Journal of Cell Biology, 174(7), 1071–1085.
Taguchi, N., Ishihara, N., Jofuku, A., Oka, T., & Mihara, K. (2007). Mitotic phosphorylation of dynamin-related GTPase Drp1 participates in mitochondrial fission. The Journal of Biological Chemistry, 282(15), 11521–11529.
Taira, T., Saito, Y., Niki, T., Iguchi-Ariga, S. M., Takahashi, K., & Ariga, H. (2004). DJ-1 has a role in antioxidative stress to prevent cell death. EMBO Reports, 5(2), 213–218.
Takahashi, K., Taira, T., Niki, T., Seino, C., Iguchi-Ariga, S. M., & Ariga, H. (2001). DJ-1 positively regulates the androgen receptor by impairing the binding of PIASx alpha to the receptor. The Journal of Biological Chemistry, 276(40), 37556–37563.
Takahashi-Fujigasaki, J., & Fujigasaki, H. (2006). Histone deacetylase (HDAC) 4 involvement in both Lewy and Marinesco bodies. Neuropathology and Applied Neurobiology, 32(5), 562–566.
Takami, Y., Nakagami, H., Morishita, R., Katsuya, T., Cui, T. X., Ichikawa, T., et al. (2007). Ubiquitin carboxyl-terminal hydrolase L1, a novel deubiquitinating enzyme in the vasculature, attenuates NF-kappaB activation. Arteriosclerosis, Thrombosis, and Vascular Biology, 27(10), 2184–2190.
Tao, X., & Tong, L. (2003). Crystal structure of human DJ-1, a protein associated with early onset Parkinson’s disease. The Journal of Biological Chemistry, 278(33), 31372–31379.
Taschenberger, G., Toloe, J., Tereshchenko, J., Akerboom, J., Wales, P., Benz, R., et al. (2013). ss-synuclein aggregates and induces neurodegeneration in dopaminergic neurons. Annals of neurology.
Tatham, M. H., Matic, I., Mann, M., & Hay, R. T. (2011). Comparative proteomic analysis identifies a role for SUMO in protein quality control. Science signaling, 4(178), rs4.
Terashima, T., Kawai, H., Fujitani, M., Maeda, K., & Yasuda, H. (2002). SUMO-1 co-localized with mutant atrophin-1 with expanded polyglutamines accelerates intranuclear aggregation and cell death. NeuroReport, 13(17), 2359–2364.
Tillman, J. E., Yuan, J., Gu, G., Fazli, L., Ghosh, R., Flynt, A. S., et al. (2007). DJ-1 binds androgen receptor directly and mediates its activity in hormonally treated prostate cancer cells. Cancer Research, 67(10), 4630–4637.
Tirard, M., Hsiao, H. H., Nikolov, M., Urlaub, H., Melchior, F., & Brose, N. (2012). In vivo localization and identification of SUMOylated proteins in the brain of His6-HA-SUMO1 knock-in mice. Proceedings of the National Academy of Sciences of the United States of America, 109(51), 21122–21127.
Tofaris, G. K., Razzaq, A., Ghetti, B., Lilley, K. S., & Spillantini, M. G. (2003). Ubiquitination of alpha-synuclein in Lewy bodies is a pathological event not associated with impairment of proteasome function. The Journal of Biological Chemistry, 278(45), 44405–44411.
Torii, S., Egan, D. A., Evans, R. A., & Reed, J. C. (1999). Human Daxx regulates Fas-induced apoptosis from nuclear PML oncogenic domains (PODs). The EMBO Journal, 18(21), 6037–6049.
Ueda, K., Fukushima, H., Masliah, E., Xia, Y., Iwai, A., Yoshimoto, M., et al. (1993). Molecular cloning of cDNA encoding an unrecognized component of amyloid in Alzheimer disease. Proceedings of the National Academy of Sciences of the United States of America, 90(23), 11282–11286.
Ullmann, R., Chien, C. D., Avantaggiati, M. L., & Muller, S. (2012). An acetylation switch regulates SUMO-dependent protein interaction networks. Molecular Cell, 46(6), 759–770.
Um, J. W., & Chung, K. C. (2006). Functional modulation of parkin through physical interaction with SUMO-1. Journal of Neuroscience Research, 84(7), 1543–1554.
Um, J. W., Min, D. S., Rhim, H., Kim, J., Paik, S. R., & Chung, K. C. (2006). Parkin ubiquitinates and promotes the degradation of RanBP2. The Journal of Biological Chemistry, 281(6), 3595–3603.
Uzunova, K., Gottsche, K., Miteva, M., Weisshaar, S. R., Glanemann, C., Schnellhardt, M., et al. (2007). Ubiquitin-dependent proteolytic control of SUMO conjugates. The Journal of Biological Chemistry, 282(47), 34167–34175.
Valente, E. M., Abou-Sleiman, P. M., Caputo, V., Muqit, M. M., Harvey, K., Gispert, S., et al. (2004). Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science, 304(5674), 1158–1160.
van de Warrenburg, B. P., Lammens, M., Lucking, C. B., Denefle, P., Wesseling, P., Booij, J., et al. (2001). Clinical and pathologic abnormalities in a family with parkinsonism and parkin gene mutations. Neurology, 56(4), 555–557.
van Duijn, C. M., Dekker, M. C., Bonifati, V., Galjaard, R. J., Houwing-Duistermaat, J. J., Snijders, P. J., et al. (2001). Park7, a novel locus for autosomal recessive early-onset parkinsonism, on chromosome 1p36. American Journal of Human Genetics, 69(3), 629–634.
Vilotti, S., Biagioli, M., Foti, R., Dal Ferro, M., Lavina, Z. S., Collavin, L., et al. (2012). The PML nuclear bodies-associated protein TTRAP regulates ribosome biogenesis in nucleolar cavities upon proteasome inhibition. Cell Death and Differentiation, 19(3), 488–500.
Wadosky, K. M., & Willis, M. S. (2012). The story so far: Post-translational regulation of peroxisome proliferator-activated receptors by ubiquitination and SUMOylation. American Journal of Physiology. Heart and Circulatory Physiology, 302(3), H515–H526.
Wakabayashi, K., Engelender, S., Yoshimoto, M., Tsuji, S., Ross, C. A., & Takahashi, H. (2000). Synphilin-1 is present in Lewy bodies in Parkinson’s disease. Annals of Neurology, 47(4), 521–523.
Wang, H., Song, P., Du, L., Tian, W., Yue, W., Liu, M., et al. (2011). Parkin ubiquitinates Drp1 for proteasome-dependent degradation: Implication of dysregulated mitochondrial dynamics in Parkinson disease. The Journal of Biological Chemistry, 286(13), 11649–11658.
Weetman, J., Wong, M. B., Sharry, S., Rcom-H’cheo-Gauthier, A., Gai, W. P., Meedeniya, A., et al. (2013). Increased SUMO-1 expression in the unilateral rotenone-lesioned mouse model of Parkinson’s disease. Neuroscience Letters.
Weger, S., Hammer, E., & Heilbronn, R. (2005). Topors acts as a SUMO-1 E3 ligase for p53 in vitro and in vivo. FEBS Letters, 579(22), 5007–5012.
Weinreb, P. H., Zhen, W., Poon, A. W., Conway, K. A., & Lansbury, P. T, Jr. (1996). NACP, a protein implicated in Alzheimer’s disease and learning, is natively unfolded. Biochemistry, 35(43), 13709–13715.
Wenzel, D. M., Lissounov, A., Brzovic, P. S., & Klevit, R. E. (2011). UBCH7 reactivity profile reveals parkin and HHARI to be RING/HECT hybrids. Nature, 474(7349), 105–108.
Wilhelmus, M. M., Nijland, P. G., Drukarch, B., de Vries, H. E., & van Horssen, J. (2012). Involvement and interplay of Parkin, PINK1, and DJ1 in neurodegenerative and neuroinflammatory disorders. Free Radical Biology & Medicine, 53(4), 983–992.
Wilkinson, K. A., & Henley, J. M. (2010). Mechanisms, regulation and consequences of protein SUMOylation. The Biochemical Journal, 428(2), 133–145.
Wilson, M. A., Collins, J. L., Hod, Y., Ringe, D., & Petsko, G. A. (2003). The 1.1-A resolution crystal structure of DJ-1, the protein mutated in autosomal recessive early onset Parkinson’s disease. Proceedings of the National Academy of Sciences of the United States of America, 100(16), 9256–9261.
Wiltshire, K. M., Dunham, C., Reid, S., Auer, R. N., & Suchowersky, O. (2010). Neuronal Intranuclear Inclusion Disease presenting as juvenile Parkinsonism. The Canadian Journal of Neurological Sciences. Le Journal Canadien des Sciences Neurologiques, 37(2), 213–218.
Wong, M. B., Goodwin, J., Norazit, A., Meedeniya, A. C., Richter-Landsberg, C., Gai, W. P., et al. (2013). SUMO-1 is associated with a subset of lysosomes in glial protein aggregate diseases. Neurotoxicity Research, 23(1), 1–21.
Xiong, H., Wang, D., Chen, L., Choo, Y. S., Ma, H., Tang, C., et al. (2009). Parkin, PINK1, and DJ-1 form a ubiquitin E3 ligase complex promoting unfolded protein degradation. The Journal of Clinical Investigation, 119(3), 650–660.
Yamashita, D., Yamaguchi, T., Shimizu, M., Nakata, N., Hirose, F., & Osumi, T. (2004). The transactivating function of peroxisome proliferator-activated receptor gamma is negatively regulated by SUMO conjugation in the amino-terminal domain. Genes to Cells: Devoted to Molecular & Cellular Mechanisms, 9(11), 1017–1029.
Yang, X., Khosravi-Far, R., Chang, H. Y., & Baltimore, D. (1997). Daxx, a novel Fas-binding protein that activates JNK and apoptosis. Cell, 89(7), 1067–1076.
Yang, S. H., & Sharrocks, A. D. (2004). SUMO promotes HDAC-mediated transcriptional repression. Molecular Cell, 13(4), 611–617.
Yang, W., Wang, L., & Paschen, W. (2013). Development of a high-throughput screening assay for inhibitors of small ubiquitin-like modifier proteases. Journal of Biomolecular Screening, 18(5), 621–628.
Yao, Q., Li, H., Liu, B. Q., Huang, X. Y., & Guo, L. (2011). SUMOylation-regulated protein phosphorylation, evidence from quantitative phosphoproteomics analyses. The Journal of Biological Chemistry, 286(31), 27342–27349.
Yeung, P. L., Chen, L. Y., Tsai, S. C., Zhang, A., & Chen, J. D. (2008). Daxx contains two nuclear localization signals and interacts with importin alpha3. Journal of Cellular Biochemistry, 103(2), 456–470.
Youle, R. J., & van der Bliek, A. M. (2012). Mitochondrial fission, fusion, and stress. Science, 337(6098), 1062–1065.
Zarranz, J. J., Alegre, J., Gomez-Esteban, J. C., Lezcano, E., Ros, R., Ampuero, I., et al. (2004). The new mutation, E46 K, of alpha-synuclein causes Parkinson and Lewy body dementia. Annals of Neurology, 55(2), 164–173.
Zhang, J., Pho, V., Bonasera, S. J., Holtzman, J., Tang, A. T., Hellmuth, J., et al. (2007). Essential function of HIPK2 in TGFbeta-dependent survival of midbrain dopamine neurons. Nature Neuroscience, 10(1), 77–86.
Zhang, L., Shimoji, M., Thomas, B., Moore, D. J., Yu, S. W., Marupudi, N. I., et al. (2005). Mitochondrial localization of the Parkinson’s disease related protein DJ-1: Implications for pathogenesis. Human Molecular Genetics, 14(14), 2063–2073.
Zhang, Q., Wu, J., Wu, R., Ma, J., Du, G., Jiao, R., et al. (2012). DJ-1 promotes the proteasomal degradation of Fis1: Implications of DJ-1 in neuronal protection. The Biochemical Journal, 447(2), 261–269.
Zhong, N., Kim, C. Y., Rizzu, P., Geula, C., Porter, D. R., Pothos, E. N., et al. (2006). DJ-1 transcriptionally up-regulates the human tyrosine hydroxylase by inhibiting the sumoylation of pyrimidine tract-binding protein-associated splicing factor. The Journal of Biological Chemistry, 281(30), 20940–20948.
Zhong, N., & Xu, J. (2008). Synergistic activation of the human MnSOD promoter by DJ-1 and PGC-1alpha: Regulation by SUMOylation and oxidation. Human Molecular Genetics, 17(21), 3357–3367.
Zhou, W., & Freed, C. R. (2005). DJ-1 up-regulates glutathione synthesis during oxidative stress and inhibits A53T alpha-synuclein toxicity. The Journal of Biological Chemistry, 280(52), 43150–43158.
Zhou, C., Huang, Y., Shao, Y., May, J., Prou, D., Perier, C., et al. (2008). The kinase domain of mitochondrial PINK1 faces the cytoplasm. Proceedings of the National Academy of Sciences of the United States of America, 105(33), 12022–12027.
Zucchelli, S., Vilotti, S., Calligaris, R., Lavina, Z. S., Biagioli, M., Foti, R., et al. (2009). Aggresome-forming TTRAP mediates pro-apoptotic properties of Parkinson’s disease-associated DJ-1 missense mutations. Cell Death and Differentiation, 16(3), 428–438.
Zungu, M., Schisler, J., & Willis, M. S. (2011). All the little pieces. -Regulation of mitochondrial fusion and fission by ubiquitin and small ubiquitin-like modifier and their potential relevance in the heart. Circulation Journal: Official Journal of the Japanese Circulation Society, 75(11), 2513–2521.
Zunino, R., Schauss, A., Rippstein, P., Andrade-Navarro, M., & McBride, H. M. (2007). The SUMO protease SENP5 is required to maintain mitochondrial morphology and function. Journal of Cell Science, 120(Pt 7), 1178–1188.
Acknowledgments
The author was funded through the Cluster of Excellence and DFG Research Center Nanoscale Microscopy and Molecular Physiology of the Brain (CNMPB).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eckermann, K. SUMO and Parkinson’s Disease. Neuromol Med 15, 737–759 (2013). https://doi.org/10.1007/s12017-013-8259-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8259-5