Abstract
Purpose
Abnormal expression of succinate dehydrogenase, (SDH), in particular of the B subunit (SDHB), is implicated in the pathogenesis of neuroendocrine tumors. This study evaluates the distribution of SDHB in WHO grading G1 and G2 intestinal, well-differentiated neuroendocrine tumors and corresponding lymph node or liver metastases.
Methods
We collected ileal well-differentiated neuroendocrine tumors specimens from consecutive patients with prior primary resection and distant synchronous or metachronous liver metastases. We obtained 195 specimens from primary tumors (n = 106) and metastases (n = 89). The expression (E) of SDHB and the immunostaining intensity (I) were evaluated semiquantitatively and combined into a single score. SDHB score was evaluated in primitive tumor and metastatic specimens.
Results
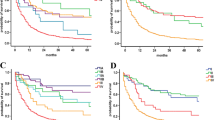
SDHB was found in all tumor cells. Mean SDHB expression was 72.7 % ± 17.1 % in primitive specimens and 27.9 % ± 24.6 % in metastatic specimens (p < 0.0001). SDH intensity was higher in primitive specimens (p < 0.0001). SDHB score was 9–12 in 96 specimens of the primitive group and 2 metastatic specimens (p < 0.0001). None of the analyzed parameters was predictive of overall survival in the primitive subset. In the metastatic subset, loss of SDHB expression, intensity, and score were prognostic factors for survival. Lower expression and intensity of SDHB in metastatic lesions were associated with longer overall survival. When combining SDHB score and Ki-67 % in the metastatic subset, a lower SDHB score was associated with prolonged overall survival, independently from Ki-67 %.
Conclusions
SDHB score was different in primitive and metastatic specimens. The combination of SDHB score and Ki-67 % was a stronger predictor of overall survival than Ki-67 % alone. This stratification might help predict survival.



Similar content being viewed by others
References
C. Dagohoy, C. Leary, J.E. Mares, E.K. Abdalla, J.B. Fleming, J.N. Vauthey, A. Rashid, D.B. Evans, J.C. Yao, M. Hassan, A. Phan, One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J. Clin. Oncol. 26, 3063–3072 (2008)
J. Calissendorff, E. Maret, A. Sundin, H. Falhammar, Ileal neuroendocrine tumors and heart: not only valvular consequences. Endocrine 48, 743–755 (2015)
M.B. Niederle, M. Hackl, K. Kaserer, B. Niederle, Gastroenteropancreatic neuroendocrine tumours: the current incidence and staging based on the WHO and European neuroendocrine tumour society classification: an analysis based on prospectively collected parameters. Endocr. Relat. Cancer. 17, 909–918 (2010)
I.M. Modlin, I. Latich, M. Kidd, M. Zikusoka, G. Eick, Therapeutic options for gastrointestinal carcinoids. Clin. Gastroenterol. Hepatol. 4, 526–547 (2006)
F. Grillo, M. Albertelli, F. Annunziata, M. Boschetti, A. Caff, S. Pigozzi, D. Ferone, L. Mastracci, Twenty years of gastroenteropancreatic neuroendocrine tumors: is reclassification worthwhile and feasible? Endocrine. 53(1), 58–62 (2016)
H. Ahlman, O. Nilsson, A.M. McNicol, P. Ruszniewski, B. Niederle, J. Ricke, R. Jensen, B. Kos-Kudla, K. Oberg, J.M. O’Connor, M.E. Pavel, M.P. Vullierme, Poorly-differentiated endocrine carcinomas of midgut and hindgut origin. Neuroendocrinology 87, 40–46 (2008)
O. Nilsson, C.E. Van, F.G. Delle, J.C. Yao, M.E. Pavel, A.M. McNicol, M.I. Sevilla Garcia, W.H. Knapp, F. Kelestimur, A. Sauvanet, S. Pauwels, D.J. Kwekkeboom, M. Caplin, Poorly differentiated carcinomas of the foregut (gastric, duodenal and pancreatic). Neuroendocrinology. 84, 212–215 (2006)
M. Milione, S. Pusceddu, P. Gasparini, F. Melotti, P. Maisonneuve, V. Mazzaferro, F.G. de Braud, G. Pelosi, Succinate dehydrogenase B subunit immunohistochemical expression predicts aggressiveness in well differentiated neuroendocrine tumorsof the ileum. Cancers (Basel) 4, 808–820 (2012)
A.J. Gill, D.E. Benn, A. Chou, A. Clarkson, A. Muljono, G.Y. Meyer-Rochow, A.L. Richardson, S.B. Sidhu, B.G. Robinson, R.J. Clifton-Bligh, Immunohistochemistry for SDHB triages genetic testing of SDHB, SDHC, and SDHD in paraganglioma- pheochromo-cytoma syndromes. Hum. Pathol. 34, 636–644 (2010)
A.J. Gill, A. Chou, R. Vilain, A. Clarkson, M. Lui, R. Jin, V. Tobias, J. Samra, D. Goldstein, C. Smith, L. Sioson, N. Parker, R.C. Smith, M. Sywak, S.,B. Sidhu, J.M. Wyatt, B.G. Robinson, R.P. Eckstein, D.E. Benn, R.J. Clifton-Bligh, Immunohistochemistry for SDHB divides gastrointestinal stromal tumors (GISTs) into 2 distinct types. Am. J. Surg. Pathol. 34, 805–814 (2010)
S.A. Boikos, C.A. Stratakis, The genetic landscape of gastrointestinal stromal tumor lacking KIT and PDGFRA mutations. Endocrine 47, 401–408 (2014)
N.D. Niemeijer, T.G. Papathomas, E. Korpershoek, R.R. de Krijger, L. Oudijk, H. Morreau, J.P. Bayley, F.J. Hes, J.C. Jansen, W.N. Dinjens, E.P. Corssmit, Succinate dehydrogenase (SDH)-deficient pancreatic neuroendocrine tumor expands the SDH-related tumor spectrum. J. Clin. Endocrinol. Metab. 100, E1386–E1393 (2015)
G. Capurso, S. Lattimore, T. Crnogorac-Jurcevic, F. Panzuto, M. Milione, V. Bhakta, N. Campanini, S.M. Swift, C. Bordi, G. Delle Fave, N.R. Lemoine, Gene expression profiles of progressive pancreatic endocrine tumours and their liver metastases reveal potential novel markers and therapeutic targets. Endocr. Relat. Cancer. 13, 541–558 (2006)
F. Andriani, G. Bertolini, F. Facchinetti, E. Baldoli, M. Moro, P. Casalini, R. Caserini, M. Milione, G. Leone, G. Pelosi, U. Pastorino, G. Sozzi, L. Roz, Conversion to stem-cell state in response to microenvironmental cues is regulated by balance between epithelial and mesenchymal features in lung cancer cells. Mol. Oncol. 10, 253–271 (2016)
B.E. Howitt, H.H. Sun, M.G. Roemer, A. Kelley, B. Chapuy, E. Aviki, C. Pak, C. Connelly, E. Gjini, Y. Shi, L. Lee, A. Viswanathan, N. Horowitz, D. Neuberg, C.P. Crum, N.L. Lindeman, F. Kuo, A.H. Ligon, G.J. Freeman, F.S. Hodi, M.A. Shipp, S.J. Rodig, Genetic basis for PD-L1 expression in squamous cell carcinomas of the cervix and vulva. JAMA Oncol 2, 518–522 (2016)
F. Grillo, M. Albertelli, M.P. Brisigotti, T. Borra, M. Boschetti, R. Fiocca, D. Ferone, L. Mastracci, Grade increases in gastro-entero-pancreatic neuroendocrine tumor metastases compared to the primary tumor. Neuroendocrinology. 103(5), 452–9 (2016)
W.M. Elshamy, R.J. Duhé, Overview: cellular plasticity, cancer stem cells and metastasis. Cancer Lett. 341, 2–8 (2013)
C. Bardella, P.J. Pollard, I. Tomlinson, SDH mutations in cancer. Biochim. Biophys. Acta 1807, 1432–1443 (2011)
L. Amar, E. Baudin, N. Burnichon, S. Peyrard, S. Silvera, J. Bertherat, X. Bertagna, M. Schlumberger, X. Jeunemaitre, A.P. Gimenez-Roqueplo, Succinate dehy- drogenase B gene mutations predict survival in patients with malignant pheochromocytomas or paragangliomas. J. Clin. Endocrinol. Metab. 92, 3822–3828 (2007)
M.S. Elston, G.Y. Meyer-Rochow, H.M. Conaglen, A. Clarkson, R.J. Clifton-Bligh, J.V. Conaglen, A.J. Gill, Increased SSTR2A and SSTR3 expression in succinate dehydrogenase-deficient pheochromocytomas and paragangliomas. Hum. Pathol. 46, 390–396 (2015)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Milione, M., Maisonneuve, P., Pellegrinelli, A. et al. Loss of succinate dehydrogenase subunit B (SDHB) as a prognostic factor in advanced ileal well-differentiated neuroendocrine tumors. Endocrine 57, 512–517 (2017). https://doi.org/10.1007/s12020-016-1180-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-1180-6