Abstract
Introduction
Skeletal fragility with high risk of vertebral fractures is an emerging complication of acromegaly in close relationship with duration of active disease. The aim of this cross-sectional study was to evaluate the prevalence and determinants of vertebral fractures in males and females with a history of long-standing active acromegaly undergoing treatment with Pegvisomant.
Subjects and methods
Thirty-eight patients (25 females, 13 males) with acromegaly under Pegvisomant therapy were evaluated for vertebral fractures and bone mineral density at lumbar spine and femoral neck. Gonadal status, serum IGF1 levels and growth hormone receptor genotype were also assessed.
Results
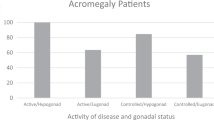
Vertebral fractures were detected in 12 patients (31.6%). Fractured patients had longer duration of active disease (p = 0.01) with higher frequency of active acromegaly (p = 0.04), received higher dose of Pegvisomant (p = 0.008), and were more frequently hypogonadic (p = 0.02) as compared to patients who did not fracture. Stratifying the patients for gender, vertebral fractures were significantly associated with Pegvisomant dose (p = 0.02) and untreated hypogonadism (p = 0.02) in males and with activity of disease (p = 0.03), serum insulin-like growth factor-I values (p = 0.01) and d3GHR polymorphism (p = 0.005) in females. No significant association was found between vertebral fractures and bone mineral density at either skeletal site.
Conclusion
Vertebral fractures are a frequent complication of long-standing active acromegaly. When patients are treated with Pegvisomant, vertebral fractures may occur in close relationship with active acromegaly and coexistent untreated hypogonadism.
Similar content being viewed by others
References
A. Giustina, G. Mazziotti, E. Canalis, Growth hormone, insuline-like growth factors, and the skeleton. Endocr. Rev. 29, 535–559 (2008)
C. Bima, S. Chiloiro, M. Mormando, S. Piacentini, E. Bracaccia, A. Giampietro, L. Tartaglione, A. Bianchi, L. De Marinis, Understanding the effect of acromegaly on the human skeleton. J. Expert Rev. Endocrinol. Metab. 11, 263–270 (2015)
G. Mazziotti, A. Bianchi, S. Bonadonna, V. Cimino, I. Patelli, A. Fusco, A. Pontecorvi, L. De Marinis, A. Giustina, Prevalence of vertebral fractures in men with acromegaly. J. Clin. Endocrinol. Metab. 93(12), 4649–4655 (2008)
G. Mazziotti, A. Bianchi, T. Porcelli, M. Mormando, F. Maffezzoni, A. Cristiano, A. Giampietro, L. De Marinis, A. Giustina, Vertebral fractures in patients with acromegaly: a 3-year prospective study. J. Clin. Endocrinol. Metab. 98, 3402–3410 (2013)
C. Battista, I. Chiodini, S. Muscarella, G. Guglielmi, M.L. Mascia, V. Carnevale, A. Scillitani, Spinal volumetric trabecular bone mass in acromegalic patients: a longitudinal study. Clin. Endocrinol. 70, 378–382 (2009)
M. Mormando, L.A. Nasto, A. Bianchi, G. Mazziotti, A. Giampietro, E. Pola, A. Pontecorvi, G. Giustina, L. De Marinis, GH receptor isoforms and skeletal fragility in acromegaly. Eur. J. Endocrinol. 171(2), 237–245 (2014)
K. Godang, N.C. Olarescu, J. Bollerslev, A. Heck, Treatment of acromegaly increases BMD but reduces Trabecular Bone Score – a longitudinal study. Eur. J. Endocrinol. 175(2), 155–164 (2016)
S. Grottoli, P. Maffei, F. Bogazzi, S. Cannavò, A. Colao, E. Ghigo, R. Gomez, E. Graziano, M. Monterubbianesi, P. Jonsson, L. De Marinis, ACROSTUDY: the Italian experience. Endocrine 48(1), 334–341 (2015). doi:10.1007/s12020-014-0393-9
A. Giustina Optimal use of pegvisomant in acromegaly: are we getting there?. Endocrine 48(1), 3–8 (2014) 10.1007/s12020-014-0462-0
A. Giustina, G. Arnaldi, F. Bogazzi, S. Cannavò, A. Colao, L. De Marinis, E. De Menis, E. Degli Uberti, F. Giorgino, S. Grottoli, A.G. Lania, P. Maffei, R. Pivonello, E. Ghigo, Pegvisomant in acromegaly: an update. J. Endocrinol. Invest. 40(6), 577–589 (2017). doi:10.1007/s40618-017-0614-1
S.E. Franck, A. Muhammad, A.J. van der Lely, S.J. Neggers, Combined treatment of somatostatin analogues with pegvisomant in acromegaly. Endocrine 52(2), 206–213 (2016). doi:10.1007/s12020-015-0810-8
G. Fairfield, L. Sesmilo, K. Katznelson, L. Katznelson, K. Pulaski, P.U. Freda, S. Stavrou, D. Kleinberg, A. Klibanski, Effects of a growth hormone receptor antagonist on bone markers in acromegaly. Clin. Endocrinol. 57, 385–390 (2002)
C. Parkinson, M. Kassem, A. Heickendorff, A. Flyvbjerg, P.J. Trainer, Pegvisomant-induced serum insulin-like growth factor-i normalization in patients with acromegaly returns elevated markers of bone turnover to normal. J. Clin. Endocrinol. Metab. 88(12), 5650–5655 (2003)
C. Jimenez, M. Ayala-Ramirez, J. Liu, R. Nunez, R.F. Gagel, Inhibition of growth hormone receptor activation by pegvisomant may increase bone density in acromegaly. Horm. Metab. Res. 43, 55–61 (2011)
G. Mazziotti, E. Canalis, A. Giustina, Drug-induced osteoporosis: mechanisms and clinical implications. Am. J. Med. 123, 877–884 (2010)
A. Giustina, P. Chanson, M.D. Bronstein, A. Klibanski, S. Lamberts, F.F. Casanueva, P. Trainer, E. Ghigo, K. Ho, S. Melmed, Acromegaly Consensus Group. A consensus on criteria for cure of acromegaly. J. Clin. Endocrinol. Metab. 95, 3141–3148 (2010)
H.R. Boquete, P.G. Sobrado, H.L. Fideleff, A.M. Sequera, A.V. Giaccio, M.G. Suárez, G.F. Ruibal, M. Miras, Evaluation of diagnostic accuracy of insulin-like growth factor (IGF)-I and IGF-binding protein-3 in growth hormone-deficient children and adults using ROC plot analysis. J. Clin. Endocrinol. Metab. 88, 4702–4708 (2003)
J.F. Griffith, H.K. Genant, New advances in imaging osteoporosis and its complications. Endocrine 42, 39–51 (2012)
H.K. Genant, M. Jergas, L. Palermo, M. Nevitt, R.S. Valentin, D. Black, S.R. Cummings, Comparison of semiquantitative visual and quantitative morphometric assessment of prevalent and incident vertebral fractures in osteoporosis. The Study of Osteoporotic Fractures Research Group. J. Bone Mineral Res. 11, 984–996 (1996)
G. Mazziotti, F. Maffezzoni, S. Frara, A. Giustina, Acromegalic osteopathy. Pituitary 20, 63–69 (2017)
G. Mazziotti, E. Biagioli, F. Maffezzoni, M. Spinelli, V. Serra, R. Maroldi, I. Floriani, A. Giustina, Bone turnover, bone mineral density, and fracture risk in acromegaly: a meta-analysis. J. Clin. Endocrinol. Metab. 100(2), 384–394 (2015). doi:10.1210/jc.2014-2937
F. Mirza, E. Canalis, Secondary osteoporosis: pathophysiology and management. Eur. J. Endocrinol. 173(3), R131–R151 (2016)
A. Bianchi, A. Giustina, V. Cimino, R. Pola, F. Angelini, A. Pontecorvi, L. De Marinis, Influence of growth hormone receptor d3 and full-length isoforms on biochemical treatment outcomes in acromegaly. J. Clin. Endocrinol. Metab. 94, 2015–2022 (2009)
A. Bianchi, G. Mazziotti, L. Tilaro, V. Cimino, F. Veltri, E. Gaetani, G. Pecorini, A. Pontecorvi, A. Giustina, L. De Marinis, Growth hormone receptor polymorphism and the effect of pegvisomant in acromegaly. Pituitary 12, 196–199 (2009)
Acknowledgements
This research received specific grant from Pfizer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Chiloiro, S., Mormando, M., Bianchi, A. et al. Prevalence of morphometric vertebral fractures in “difficult” patients with acromegaly with different biochemical outcomes after multimodal treatment. Endocrine 59, 449–453 (2018). https://doi.org/10.1007/s12020-017-1391-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-017-1391-5