Abstract
Purpose
To investigate whether exendin-4 (Ex-4), a glucagon-like peptide 1 receptor (GLP-1R) agonist, affects connexin 43 (Cx43) expression in osteoblasts, and determine the specific mechanism underlying Cx43 modulation by Ex-4.
Methods
Osteoblast-like MC3T3-E1 cells were treated with Ex-4 with or without GLP-1R antagonist. We assessed Cx43 expression using RT-PCR, western blotting, and confocal microscopy; visualized intercellular communication using Lucifer yellow dye transfer assay; evaluated osteoblast differentiation using alkaline phosphatase and Alizarin red S (ARS) staining. Cx43 silencing or overexpression was investigated via RNA-interference or adenovirus infection. The mechanism underlying Cx43 regulation by Ex-4 was determined via treatment with either Src kinase inhibitor, KX2–391, Akt activator, sc79, or inhibitor, LY294002.
Results
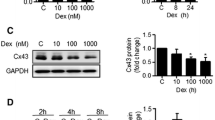
Ex-4 treatment enhanced Cx43 expression and gap junctional intercellular communication in MC3T3-E1 cells. GLP-1R antagonist pretreatment abrogated the induction of Cx43 expression. Cx43 silencing significantly decreased ARS staining intensity in Ex-4-treated cells, whereas overexpression enhanced cell differentiation. Treatment with KX2–391 reduced both the Ex-4-induced increase of Cx43 expression and p-Akt protein levels. sc79 upregulated Cx43 expression, while LY294002 attenuated Cx43 upregulation by Ex-4.
Conclusions
Induced Cx43 expression in osteoblasts via the Src-Akt signaling pathway illustrates the underlying mechanism for promoting osteoblast differentiation by Ex-4.





Similar content being viewed by others
References
T.D. Müller, B. Finan, S.R. Bloom, D. D’Alessio, D.J. Drucker, P.R. Flatt, A. Fritsche, F. Gribble, H.J. Grill, J.F. Habener, J.J. Holst, W. Langhans, J.J. Meier, M.A. Nauck, D. Perez-Tilve, A. Pocai, F. Reimann, D.A. Sandoval, T.W. Schwartz, R.J. Seeley, K. Stemmer, M. Tang-Christensen, S.C. Woods, R.D. DiMarchi, M.H. Tschöp, Glucagon-like peptide 1 (GLP-1). Mol. Metab. 30, 72–130 (2019). https://doi.org/10.1016/j.molmet.2019.09.010
H. Jiao, E. Xiao, D.T. Graves, Diabetes and its effect on bone and fracture healing. Curr. Osteoporos. Rep. 13, 327–335 (2015). https://doi.org/10.1007/s11914-015-0286-8
Y. Feng, L. Su, X. Zhong, W. Guohong, H. Xiao, Y. Li, L. Xiu, Exendin-4 promotes proliferation and differentiation of MC3T3-E1 osteoblasts by MAPKs activation. J. Mol. Endocrinol. 56, 189–199 (2016). https://doi.org/10.1530/JME-15-0264
J. Meng, X. Ma, N. Wang, M. Jia, L. Bi, Y. Wang, M. Li, H. Zhang, X. Xue, Z. Hou, Y. Zhou, Z. Yu, G. He, X. Luo, Activation of GLP-1 receptor promotes bone marrow stromal cell osteogenic differentiation through beta-catenin. Stem Cell Rep. 6, 633 (2016). https://doi.org/10.1016/j.stemcr.2016.03.010
C. Yamada, Y. Yamada, K. Tsukiyama, K. Yamada, N. Udagawa, N. Takahashi, K. Tanaka, D.J. Drucker, Y. Seino, N. Inagaki, The murine glucagon-like peptide-1 receptor is essential for control of bone resorption. Endocrinology 149, 574–579 (2008). https://doi.org/10.1210/en.2007-1292
L. Ma, R. Hua, Y. Tian, H. Cheng, R.J. Fajardo, J.J. Pearson, T. Guda, D.B. Shropshire, S. Gu, J.X. Jiang, Connexin 43 hemichannels protect bone loss during estrogen deficiency. Bone Res 7, 11 (2019). https://doi.org/10.1038/s41413-019-0050-2
A.E. Loiselle, E.M. Paul, G.S. Lewis, H.J. Donahue, Osteoblast and osteocyte-specific loss of Connexin43 results in delayed bone formation and healing during murine fracture healing. J. Orthop. Res. 31, 147–154 (2013). https://doi.org/10.1002/jor.22178
J.H. Kim, K. Kim, I. Kim, S. Seong, N. Kim, c-Src-dependent and -independent functions of matk in osteoclasts and osteoblasts. J. Immunol. 200, 2455–2463 (2018). https://doi.org/10.4049/jimmunol.1700582
Y.H. Choi, Y. Han, S.H. Lee, H. Cheong, K.H. Chun, C.Y. Yeo, K.Y. Lee, Src enhances osteogenic differentiation through phosphorylation of Osterix. Mol. Cell Endocrinol. 407, 85–97 (2015). https://doi.org/10.1016/j.mce.2015.03.010
S. Chen, M. Hu, M. Shen, S. Wang, C. Wang, F. Chen, Y. Tang, X. Wang, H. Zeng, M. Chen, J. Gao, F. Wang, Y. Su, Y. Xu, J. Wang, IGF-1 facilitates thrombopoiesis primarily through Akt activation. Blood 132, 210–222 (2018). https://doi.org/10.1182/blood-2018-01-825927
J. Rowlands, J. Heng, P. Newsholme, R. Carlessi, Pleiotropic effects of GLP-1 and analogs on cell signaling, metabolism, and function. Front. Endocrinol. (Lausanne) 9, 672 (2018). https://doi.org/10.3389/fendo.2018.00672
C. Shen, M.R. Kim, J.M. Noh, S.J. Kim, S.O. Ka, J.H. Kim, B.H. Park, J.H. Park, Glucocorticoid suppresses connexin 43 expression by inhibiting the Akt/mTOR signaling pathway in osteoblasts. Calcif. Tissue Int 99, 88–97 (2016). https://doi.org/10.1007/s00223-016-0121-y
J.E. Campbell, D.J. Drucker, Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 17, 819–837 (2013). https://doi.org/10.1016/j.cmet.2013.04.008
J. Kolic, P.E. MacDonald, cAMP-independent effects of GLP-1 on beta cells. J. Clin. Invest. 125, 4327–4330 (2015). https://doi.org/10.1172/jci85004
M. Pereira, J. Jeyabalan, C.S. Jorgensen, M. Hopkinson, A. Al-Jazzar, J.P. Roux, P. Chavassieux, I.R. Orriss, M.E. Cleasby, C. Chenu, Chronic administration of Glucagon-like peptide-1 receptor agonists improves trabecular bone mass and architecture in ovariectomised mice. Bone 81, 459–467 (2015). https://doi.org/10.1016/j.bone.2015.08.006
G. Mabilleau, M. Pereira, C. Chenu, Novel skeletal effects of glucagon-like peptide-1 (GLP-1) receptor agonists. J. Endocrinol. 236, R29–R42 (2018). https://doi.org/10.1530/joe-17-0278
J.Y. Kim, S.K. Lee, K.J. Jo, D.Y. Song, D.M. Lim, K.Y. Park, L.F. Bonewald, B.J. Kim, Exendin-4 increases bone mineral density in type 2 diabetic OLETF rats potentially through the down-regulation of SOST/sclerostin in osteocytes. Life Sci. 92, 533–540 (2013). https://doi.org/10.1016/j.lfs.2013.01.001
A. Garcia-Martin, P. Rozas-Moreno, R. Reyes-Garcia, S. Morales-Santana, B. Garcia-Fontana, J.A. Garcia-Salcedo, M. Munoz-Torres, Circulating levels of sclerostin are increased in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 97, 234–241 (2012). https://doi.org/10.1210/jc.2011-2186
L. Yang, G. Zhou, M. Li, Y. Li, L. Yang, Q. Fu, Y. Tian, High glucose downregulates connexin 43 expression and its gap junction and hemichannel function in osteocyte-like MLO-Y4 cells through activation of the p38MAPK/ERK signal pathway. Diabetes Metab. Syndr. Obes. 13, 545–557 (2020). https://doi.org/10.2147/dmso.S239892
M.C. Moorer, J.P. Stains, Connexin43 and the intercellular signaling network regulating skeletal remodeling. Curr. Osteoporos. Rep. 15, 24–31 (2017). https://doi.org/10.1007/s11914-017-0345-4
Funding
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2017R1D1A3B04028873), and by the Research Institute of Clinical Medicine of Jeonbuk National University-Biomedical Research Institute of Jeonbuk National University Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J.H., Shen, C., Oh, H. et al. Exendin-4, a glucagon-like peptide receptor agonist, facilitates osteoblast differentiation via connexin43. Endocrine 72, 672–680 (2021). https://doi.org/10.1007/s12020-021-02664-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02664-7