Abstract
Background
We hypothesized that the degree of preserved functional connectivity within the DMN during the first week after cardiopulmonary arrest (CPA) would be associated with functional outcome at hospital discharge.
Methods
Initially comatose CPA survivors with indeterminate prognosis at 72 h were enrolled. Seventeen CPA subjects between 4 and 7 days after CPA and 17 matched controls were studied with task-free fMRI. Independent component analysis was performed to delineate the DMN. Connectivity strength in the DMN was compared between CPA subjects and controls, as well as between CPA subjects with good outcome (discharge Cerebral Performance Category or CPC 1–2) and those with bad outcome (CPC 3–5). The relationship between connectivity strength in the posterior cingulate cortex (PCC) and precuneus (PC) within the DMN with discharge CPC was evaluated using linear regression.
Results
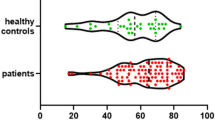
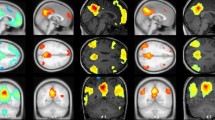
Compared to controls, CPA subjects had significantly lower connectivity strength in subregions of the DMN, the PCC and PC (p < 0.0001). Furthermore, connectivity strength in the PCC and PC was greater in CPA subjects with good outcome (n = 8) than those with bad outcome (n = 9) (p < 0.003). Among CPA subjects, the connectivity strength in the PCC and PC showed strong linear correlations with the discharge CPC (p < 0.005).
Conclusions
Among initially comatose CPA survivors with indeterminate prognosis, task-free fMRI demonstrated graded disruption of DMN connectivity, especially in those with bad outcomes. If confirmed, connectivity strength in the PC/PCC may provide a clinically useful prognostic marker for functional recovery after CPA.




Similar content being viewed by others
References
Bedell SE, Delbanco TL, Cook EF, Epstein EF. Survival after cardiopulmonary resuscitation in the hospital. N Engl J Med. 1983;309:569–76.
Edgren E, Kelsey S, Sutton K, Safar P. The presenting ECG pattern in survivors of cardiac arrest and its relation to the subsequent long-term survival. Brain Resuscitation Clinical Trial I Study Group. Acta Aneasthesiol Scand. 1989;33:265–71.
Berek K, Jeschow M, Aichner F. The prognostication of cerebral hypoxia after out-of-hospital cardiac arrest in adults. Eur Neurol. 1997;37:135–45.
Nichol G, Stiell IG, Herbert P, Wells GA, Vandemheen K, Laupacis A. What is the quality of life for survivors of cardiac arrest? A prospective study. Acad Emerg Med. 1999;6:95–102.
Neumar RW, Nolan JP, Adrie C, et al. Post-cardiac arrest syndrome. Epidemiology, pathophysiology, treatment, and prognostication. A consensus statement from the International Liaison Committee on resuscitation. Circulation. 2008;118:2452–83.
Wijdicks EFM, Hijdra A, Young GB, Bassetti CL, Wiebe S. Practice parameter: prediction of outcome in comatose survivors after cardiopulmonary resuscitation (an evidence-based review). Neurology. 2006;67:203–10.
Bunch TJ, White RD, Gersh BJ, et al. Long-term outcomes of out-of-hospital cardiac arrest after successful early defibrillation. N Engl J Med. 2003;348:2626–33.
van Alem AP, de Vos R, Schmand B, Koster RW. Cognitive impairment in survivors of out-of-hospital cardiac arrest. Am Heart J. 2004;148:416–21.
Moulaert VRMP, Verbunt JA, van Heugten CM, Wade DT. Cognitive impairments in survivors of out-of-hospital cardiac arrest: a systematic review. Resuscitation. 2009;80:297–305.
Wijdicks EF, Campeau NG, Miller GM. MR imaging in comatose survivors of cardiac resuscitation. Am J Neuroradiol. 2001;22:1561–5.
Els T, Kassubek J, Kubalek R, Klisch J. Diffusion-weighted MRI during early global cerebral hypoxia: a predictor for clinical outcome? Acta Neurol Scand. 2004;110:361–7.
Llinas R, Ribary U, Contreras D, Pedroarena C. The neuronal basis for consciousness. Philos Trans R Soc Lond B Biol Sci. 1998;353:1841–9.
Laureys S, Faymonville ME, Peigneux P, et al. Cortical processing of noxious somatosensory stimuli in the persistent vegetative state. Neuroimage. 2002;17:732–41.
Schiff ND, Rodriguez-Moreno D, Kamel A, et al. fMRI reveals large-scale network activation in minimally conscious patients. Neurology. 2005;64:514–23.
Biswal B, Yetkin FZ, Haughton VM, Hyde JS. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med. 1995;34:537–41.
Hampson M, Peterson BS, Skudlarski P, Gatenby JC, Gore JC. Detection of functional connectivity using temporal correlations in MR images. Hum Brain Mapp. 2002;15:513–9.
Schiff ND. Multimodal neuroimaging approaches to disorders of consciousness. J Head Trauma Rehabil. 2006;21:388–97.
He BJ, Shulman GL, Snyder AZ, Corbetta M. The role of impaired neuronal communication in neurological disorders. Curr Opin Neurol. 2007;20:655–60.
Ioannides AA. Dynamic functional connectivity. Curr Opin Neurobiol. 2007;17:161–70.
Raichle ME, Snyder AZ. A default mode of brain function: a brief history of an evolving idea. Neuroimage. 2007;37:1083–90.
Greicius MD, Krasnow B, Reiss AL, Menon V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci USA. 2003;100:253–8.
Fox MD, Corbetta M, Snyder AZ, Vincent JL, Raichle ME. Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc Natl Acad Sci USA. 2006;103:10046–51.
Fernández-Espejo D, Soddu A, Cruse D, et al. A role for the default mode network in the bases of disorders of consciousness. Ann Neurol. 2012;72:335–43.
Jacobs I, Nadkarni V. The ILCOR Task Force on Cardiac Arrest and Cardiopulmonary Resuscitation Outcomes. Update and simplification of the Utstein templates for resuscitation registries: a statement for healthcare professionals from a task force of the International Liaison Committee on Resuscitation. Circulation. 2004;110:3385–97.
Glover GH, Law CS. Spiral-in/out BOLD fMRI for increased SNR and reduced susceptibility artifacts. Magn Reson Med. 2001;46:515–22.
Smith SM, Jenkinson M, Woolrich MW, et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage. 2004;23(Suppl 1):S208–19.
Calhoun VD, Liu J, Adali T. A review of group ICA for fMRI data and ICA for joint inference of imaging, genetic, and ERP data. Neuroimage. 2009;45(Suppl 1):S163–72.
Beckmann CF, DeLuca M, Devlin JT, Smith SM. Investigations into resting-state connectivity using independent component analysis. Philos Trans R Soc Lond B Biol Sci. 2005;360:1001–13.
Smith SM, Fox PT, Miller KL, et al. Correspondence of the brain’s functional architecture during activation and rest. Proc Natl Acad Sci USA. 2009;106:13040–5.
Wang X, Foryt P, Ochs R, et al. Abnormalities in resting-state functional connectivity in early human immunodeficiency virus infection. Brain Connect. 2011;1:207–17.
Wijdicks EF, Bamlet WR, Maramatton BV, Manno EM, McClelland RL. Validation of a new coma scale: the FOUR score. Ann Neurol. 2005;58:585–93.
Cauda F, Micon BM, Sacco K, et al. Disrupted intrinsic functional connectivity in the vegetative state. J Neurol Neurosurg Psychiatry. 2009;80:429–31.
Vanhaudenhuyse A, Noirhomme Q, Tshibanda LJ, et al. Default network connectivity reflects the level of consciousness in non-communicative brain-damaged patients. Brain. 2010;133:161–71.
Norton L, Hutchison RM, Young GB, Lee DH, Sharpe MD, Mirsattari SM. Disruptions of functional connectivity in the default mode network of comatose patients. Neurology. 2012;78:175–81.
Rossetti AO, Oddo M, Logroscino G, Kaplan PW. Prognostication after cardiac arrest and hypothermia: a prospective study. Ann Neurol. 2010;67:301–7.
Fugate JE, Wijdicks EF, White RD, Rabinstein AA. Does therapeutic hypothermia affect time to awakening in cardiac arrest survivors? Neurology. 2011;77:1346–50.
Al Thenayan E, Savard M, Sharpe M, Norton L, Young B. Predictors of poor neurologic outcome after induced mild hypothermia following cardiac arrest. Neurology. 2008;71:1535–7.
Mhuircheartaigh RN, Rosenorn-Lanng D, Wise R, Jbabdi S, Rogers R, Tracey I. Cortical and subcortical connectivity changes during decreasing levels of consciousness in humans: a functional magnetic resonance imaging study using propofol. J Neurosci. 2010;30:9095–102.
Boveroux P, Vanhaudenhuyse A, Bruno MA, et al. Breakdown of within- and between-network resting state functional magnetic resonance imaging connectivity during propofol-induced loss of consciousness. Anesthesiology. 2010;113:1038–53.
Schaafsma A, de Jong BM, Bams JL, Haaxma-Reiche H, Pruim J, Zijlstra JG. Cerebral perfusion and metabolism in resuscitated patients with severe post-hypoxic encephalopathy. J Neurol Sci. 2003;210:23–30.
Acknowledgments
The authors thank Denise Dittrich, RN and Tracy Stern, RN from the Queen’s Medical Center Neuroscience Institute for study coordination; Todd Seto, MD from the Queen’s Medical Center Department of Cardiology for assistance with database design; and Ahnate Lim from the University of Hawaii MRI Research Center for assistance with data processing. Funding support for this study was provided by the Hawaii Community Foundation Grant 11ADVC-49231, Queen Emma Research Fund, and NIH Grants 1P30GM103341-01, U54NS56883, and K24DA16170.
Conflict of interest
Matthew Koenig declares that he has no conflict of interest. John Holt declares that he has no conflict of interest. Thomas Ernst consults for and holds patents through Kineticor, Inc. Steven Buchthal declares that he has no conflict of interest. Kazuma Nakagawa declares that he has no conflict of interest. Victor Stenger declares that he has no conflict of interest. Linda Chang declares that she has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koenig, M.A., Holt, J.L., Ernst, T. et al. MRI Default Mode Network Connectivity is Associated with Functional Outcome After Cardiopulmonary Arrest. Neurocrit Care 20, 348–357 (2014). https://doi.org/10.1007/s12028-014-9953-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-014-9953-3