Abstract
Background
Understanding the molecular mechanisms of colorectal cancer has evolved during the last decade ushering the era of personalized medicine. Alteration of BRAF and PI3K is common in colorectal cancer, and can affect several signaling pathways including EGFR (epidermal growth factor receptor). The aim of this meta-analysis is to evaluate the clinical role of PI3K and BRAF mutations in patients with KRAS wild-type metastatic colorectal cancer (MCRC) receiving an EGFR monoclonal antibody (anti-EGFR) inhibitor as first-line therapy.
Methods
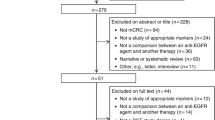
A literature search was performed to identify studies exploring the association between PI3K/BRAF mutations and clinical outcomes of KRAS wild-type mCRC patients treated with anti-EGFR as a first-line therapy. The primary clinical outcome was overall response rate (ORR). The secondary outcomes included progression-free survival (PFS) and overall survival (OS). The pooled relative risk (RR) or hazard ratio (HR) was estimated by using fixed-effect model or random effect model according to heterogeneity between studies.
Results
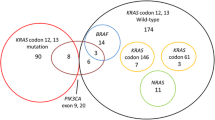
Ten studies with 1470 mCRC patients (357 for PI3K studies and 1113 from BRAF studies) met selection criteria. We observed a trend towards lower ORR in patients with PI3K mutations (3 studies, 357 patients; ORR = 14.3% in mutant-type PI3K vs. 52.4% in wild-type PIK3CA [95% CI − 0.12–0.02]; P = 0.13). Patients with mutant-type PI3K have significant shorter PFS (3 studies, 357 patients, 3.8 vs. 4.15 months, HR = 1.36; [95% CI 1.04–1.77]; P = 0.02]), and OS (3 studies, 357 patients, 14.17 vs. 16.3 months, HR = 1.50; [95% CI 1.14–1.97]; P = 0.004) compared to those with wild PI3K. For BRAF, patients with mutant type have significantly lower ORR (7 studies, 1113 patients; ORR = 33% vs. 39%; [95% CI − 0.16–0.01]; P = 0.03), shorter PFS (5 studies, 814 patients, 3.9 vs. 5.7 months, HR = 1.72; [95% CI 1.47–2.01]; P = 0.00001), and shorter OS (4 studies, 766 pts., 9.1 vs. 18.9 months, HR = 1.22; [95% CI 1.04–1.44]; P = 0.01) compared to those with wild-type.
Conclusion
This analysis suggests that patients with mCRC and either PI3K or BRAF mutation may have a lower response and worse outcome when treated with anti-EGFR in the first line. Given their worse outcome, routine testing for BRAF and PI3K mutational status should be considered. Novel therapeutic approaches are needed for patients with mutations in BRAF or PI3K.




Similar content being viewed by others
References
Haggar FA, Boushey RP. Colorectal cancer epidemiology: incidence, mortality, survival, and risk factors. Clin Colon Rectal Surg. 2009;22(4):191–7. https://doi.org/10.1055/s-0029-1242458.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30. https://doi.org/10.3322/caac.21387.
Walker F, Kato A, Gonez LJ, Hibbs ML, Pouliot N, Levitzki A, et al. Activation of the Ras/mitogen-activated protein kinase pathway by kinase-defective epidermal growth factor receptors results in cell survival but not proliferation. Mol Cell Biol. 1998;18(12):7192–204. https://doi.org/10.1128/MCB.18.12.7192.
Dienstmann R, Vilar E, Tabernero J. Molecular predictors of response to chemotherapy in colorectal cancer. Cancer J. 2011;17(2):114–26. https://doi.org/10.1097/PPO.0b013e318212f844.
Shen WD, Chen HL, Liu PF. EGFR gene copy number as a predictive biomarker for resistance to anti-EGFR monoclonal antibodies in metastatic colorectal cancer treatment: a meta-analysis. Chin J Cancer Res. 2014;26(1):59–71. https://doi.org/10.3978/j.issn.1000-9604.2014.01.10.
Nagahara H, Mimori K, Ohta M, Utsunomiya T, Inoue H, Barnard GF, et al. Somatic mutations of epidermal growth factor receptor in colorectal carcinoma. Clin Cancer Res. 2005;11(4):1368–71. https://doi.org/10.1158/1078-0432.CCR-04-1894.
Tan C, Du X. KRAS mutation testing in metastatic colorectal cancer. World J Gastroenterol. 2012;18(37):5171–80. https://doi.org/10.3748/wjg.v18.i37.5171.
Zhao B, Wang L, Qiu H, Zhang M, Sun L, Peng P, et al. Mechanisms of resistance to anti-EGFR therapy in colorectal cancer. Oncotarget. 2017;8(3):3980–4000. https://doi.org/10.18632/oncotarget.14012.
Clarke CN, Kopetz ES. BRAF mutant colorectal cancer as a distinct subset of colorectal cancer: clinical characteristics, clinical behavior, and response to targeted therapies. J Gastrointest Oncol. 2015;6(6):660–7. https://doi.org/10.3978/j.issn.2078-6891.2015.077.
Barras D. BRAF mutation in colorectal cancer: an update. Biomark Cancer. 2015;7(Suppl 1):9–12. https://doi.org/10.4137/BIC.S25248.
Zimmermann M, Zouhair A, Azria D, Ozsahin M. The epidermal growth factor receptor (EGFR) in head and neck cancer: its role and treatment implications. Radiat Oncol. 2006;1(1):11. https://doi.org/10.1186/1748-717X-1-11.
Brand TM, Iida M, Li C, Wheeler DL. The nuclear epidermal growth factor receptor signaling network and its role in cancer. Discov Med. 2011;12(66):419–32.
Seshacharyulu P, Ponnusamy MP, Haridas D, Jain M, Ganti AK, Batra SK. Targeting the EGFR signaling pathway in cancer therapy. Expert Opin Ther Targets. 2012;16(1):15–31. https://doi.org/10.1517/14728222.2011.648617.
Hsu HC, Thiam TK, Lu YJ, Yeh CY, Tsai WS, You JF, et al. Mutations of KRAS/NRAS/BRAF predict cetuximab resistance in metastatic colorectal cancer patients. Oncotarget. 2016;7(16):22257–70. https://doi.org/10.18632/oncotarget.8076.
Kishiki T, Ohnishi H, Masaki T, Ohtsuka K, Ohkura Y, Furuse J, et al. Overexpression of MET is a new predictive marker for anti-EGFR therapy in metastatic colorectal cancer with wild-type KRAS. Cancer Chemother Pharmacol. 2014;73(4):749–57. https://doi.org/10.1007/s00280-014-2401-4.
Pietrantonio F, Petrelli F, Coinu A, di Bartolomeo M, Borgonovo K, Maggi C, et al. Predictive role of BRAF mutations in patients with advanced colorectal cancer receiving cetuximab and panitumumab: a meta-analysis. Eur J Cancer. 2015;51(5):587–94. https://doi.org/10.1016/j.ejca.2015.01.054.
Sartore-Bianchi A, Martini M, Molinari F, Veronese S, Nichelatti M, Artale S, et al. PIK3CA mutations in colorectal cancer are associated with clinical resistance to EGFR-targeted monoclonal antibodies. Cancer Res. 2009;69(5):1851–7. https://doi.org/10.1158/0008-5472.CAN-08-2466.
Perrone F, Lampis A, Orsenigo M, di Bartolomeo M, Gevorgyan A, Losa M, et al. PI3KCA/PTEN deregulation contributes to impaired responses to cetuximab in metastatic colorectal cancer patients. Ann Oncol. 2009;20(1):84–90. https://doi.org/10.1093/annonc/mdn541.
Sood A, McClain D, Maitra R, Basu-Mallick A, Seetharam R, Kaubisch A, et al. PTEN gene expression and mutations in the PIK3CA gene as predictors of clinical benefit to anti-epidermal growth factor receptor antibody therapy in patients with KRAS wild-type metastatic colorectal cancer. Clin Colorectal Cancer. 2012;11(2):143–50. https://doi.org/10.1016/j.clcc.2011.12.001.
Wang F, Bai L, Liu TS, Yu YY, He MM, Liu KY, et al. Right-sided colon cancer and left-sided colorectal cancers respond differently to cetuximab. Chin J Cancer. 2015;34(9):384–93. https://doi.org/10.1186/s40880-015-0022-x.
Kim ST, Lee SJ, Lee J, Park SH, Park JO, Lim HY, et al. The impact of microsatellite instability status and sidedness of the primary tumor on the effect of cetuximab-containing chemotherapy in patients with metastatic colorectal cancer. J Cancer. 2017;8(14):2809–15. https://doi.org/10.7150/jca.18286.
Safaee Ardekani G, Jafarnejad SM, Tan L, Saeedi A, Li G. The prognostic value of BRAF mutation in colorectal cancer and melanoma: a systematic review and meta-analysis. PLoS One. 2012;7(10):e47054. https://doi.org/10.1371/journal.pone.0047054.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mohamed, A., Twardy, B., AbdAllah, N. et al. Clinical Impact of PI3K/BRAF Mutations in RAS Wild Metastatic Colorectal Cancer: Meta-analysis Results. J Gastrointest Canc 50, 269–275 (2019). https://doi.org/10.1007/s12029-018-0062-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-018-0062-y