Abstract
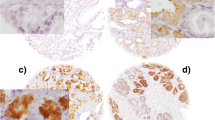
The aim of this study is to detect the expression levels of spindle assembly checkpoint protein-BubR1 and microtubule-associated protein-Tau in human prostate cancer tissues of different Gleason score, and to test whether there is a relationship between their expression levels and clinicopathologic parameters including response to docetaxel treatment, Gleason score, and overall survival (OS). Moreover, to test whether Tau protein expressed in the cancerous prostate tissue is phosphorylated. Thirty patients who received at least three cycles docetaxel for metastatic castrate-resistant prostate cancer were included into the trial. The patients’ formalin-fixed and paraffin-embedded prostate tissue specimens were retrospectively obtained from the pathology department archives of Ege University School of Medicine. The expression status of BubR1 protein was defined by immunohistochemical (IHC) using the anti-BubR1 antibody. The expression status of Tau protein was defined by IHC using the two types of Tau antibodies: anti-Tau-1 antibody (that recognizes Tau only in its dephosphorylated form) and anti-PHF-Tau antibody (that recognizes all isoforms of human Tau proteins independent of its phosphorylation status). The BubR1 and Tau were overexpressed in about 63 and 23 % of the study group, respectively. Tau overexpression was significantly associated with lower Gleason score. There was no significant association between the expression levels of BubR1 and Tau proteins, and docetaxel response. Reduced BubR1 expression was strongly associated with longer survival (P = 0.008), whereas Tau expression status did not effect survival. Moreover, the Tau expression of cancerous prostate tissue was highly dephosphorylated. In this clinicopathological study, our findings did not confirm the preclinical observations that low BubR1 and Tau expression confer selective sensitivity to microtubulisin drugs. Our data imply that reduced BubR1 expression was a predictor for longer OS, and the possibility that high Tau expression may be involved in better prognosis due to its relationship to the Gleason score. Furthermore, our data suggest that both Tau and BubR1 may be a promising prognostic marker rather than predictive marker in patients with prostate cancer.



Similar content being viewed by others
Abbreviations
- Bub1-3:
-
Budding uninhibited by benzimidazoles
- BubR1:
-
Budding uninhibited by benzimidazoles 1-related protein kinase
- IHC:
-
Immunohistochemical
- mCRPC:
-
Metastatic castrate-resistant prostate cancer
- RECIST:
-
Response evaluation criteria in solid tumors
- PSA:
-
Prostate-specific antigen
- MAPs:
-
Microtubule-associated proteins
- SAC:
-
Spindle assembly checkpoint
- Mad1–3:
-
Mitotic arrest deficient proteins
- OS:
-
Overall survival
- DFS:
-
Disease-free survival
- FFS:
-
Recurrence-free survival
- HR:
-
Hazard ratio
- NSABP:
-
National surgical breast and bowel project
- ER:
-
Estrogen receptor
- HER2:
-
Epidermal growth factor receptor 2
References
Petrylak DP, Tangen CM, Hussain MH, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20.
Tannock IF, de Wit R, Horti J, et al. Docetaxel plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12.
McGrogan BT, Gilmartin B, Carney DN, McCann A. Taxanes, microtubules and chemo resistant breast cancer. Biochim Biophys Acta. 2008;1785:96–132.
Orr G, Verdier PP. Mechanisms of taxol resistance related to microtubules. Oncogene. 2003;22:7280–95.
Honore S, Pasquier E, Braguer D. Understanding microtubule dynamics for improved cancer therapy. Cell Mol Life Sci. 2005;62:3039–56.
Dehmelt L, Halpain S. The MAP2/tau family of microtubule-associated Proteins. Genome Biol. 2004;6:204.
Musacchio A, Salmon ED. The spindle-assembly checkpoint in space and time. Nat Rev Mol Cell Biol. 2007;8:379–93.
Rouzier R, Rajan R, Wagnera P, et al. Microtubule-associated protein tau: a marker of paclitaxel sensitivity in breast cancer. Proc Natl Acad Sci USA. 2005;102:8315–20.
Munro A, Cameron D, Thomas J, Twelves C, Bartlett J. BUBR1 and MAD2: novel markers for predicting benefit from adjuvant anthracyclines? abstracts: thirty-second annual ctrc-aacr san antonio breast cancer symposium 2009; San Antonio. J Cancer Res. 2009;69(4):2124.
Rizzardi C, Torelli L, Barresi E, et al. Bubr1 expression in oral squamous cell carcinoma and its relationship to tumor stage and survival. Head Neck. 2011;33(5):727–33.
Lee YK, Choi E, Kim MA, Park PG, Park NH, Lee H. BubR1 as a prognostic marker for recurrence-free survival rates in epithelial ovarian cancers. Br J Cancer. 2009;101(3):504–10.
Shao YY, Kuo KT, Hu FC, et al. Predictive and prognostic values of tau and ERCC1 in advanced breast cancer patients treated with paclitaxel and cisplatin. J Clin Oncol. 2009;27(26):4287–92.
Pusztai L, Jeong JH, Gong Y, et al. Evaluation of microtubule-associated protein-Tau expression as a prognostic and predictive marker in the NSABP-B 28 randomized clinical trial. J Clin Oncol. 2009;27(26):4287–92.
Souter S, Lee G. Microtubule-associated protein tau in human prostate cancer cells: isoforms, phosphorylation, and interactions. J Cell Biochem. 2009;108(3):555–64.
Bubley GJ, Carducci M, Dahut W, et al. Eligibility and response guidelines for phase II clinical trials in androgen-independent prostate cancer: recommendations from the prostate-specific antigen working group. J Clin Oncol. 1999;17:3461–7.
Scher HI, Halabi S, Tannock I, et al. Prostate cancer clinical trials working group. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical TrialsWorking Group. J Clin Oncol. 2008;26:1148–59.
Nagao K, Yamamoto Y, Hara T, et al. Ki67 and BUBR1 May discriminate clinically insignificant prostate cancer in the PSA Range <4 ng/ml. Jpn J Clin Oncol. 2011;41(4):555–64.
Noguchi S. Predictive factors for response to docetaxel in human breast cancers. Cancer Sci. 2006;97:813–20.
Sudo T, Nitta M, Saya H, Ueno NT. Dependence of paclitaxel sensitivity on a functional spindle assembly checkpoint. Cancer Res. 2004;64:2502–8.
Cheung HW, Jin DY, Ling MT, et al. Mitotic arrest deficient 2 expression induces chemo sensitization to a DNA-damaging agent, cisplatin, in nasopharyngeal carcinoma cells. Cancer Res. 2005;65:1450–8.
Nitta M, Kobayashi O, Honda S, et al. Spindle checkpoint function is required for mitotic catastrophe induced by DNA damaging agents. Oncogene. 2004;23:6548–58.
Vogel C, Kienitz A, Muller R, Bastians H. The mitotic spindle checkpoint is a critical determinant for topoisomerase-based chemotherapy. J Biol Chem. 2005;280:4025–8.
Fang Y, Liu T, Wang X, et al. BubR1 is involved in regulation of DNA damage responses. Oncogene. 2006;25(25):3598–605.
Lee EA, Keutmann MK, Dowling ML, Harris E, Chan G, Kao GD. Inactivation of the mitotic checkpoint as a determinant of the efficacy of microtubule-targeted drugs in killing human cancer cells. Mol Cancer Ther. 2004;3:661–9.
Seike M, Gemma A, Hosoya Y, et al. The promoter region of the human BUBR1 gene and its expression analysis in lung cancer. Lung Cancer. 2002;38:229–34.
Shichiri M, Yoshinaga K, Hisatomi H, Sugihara K, Hirata Y. Genetic and epigenetic inactivation of mitotic checkpoint genes hBUB1 and hBUBR1 and their relationship to survival. Cancer Res. 2002;62:13–7.
Grabsch H, Takeno S, Parsons WJ, et al. Overexpression of the mitotic checkpoint genes BUB1, BUBR1, and BUB3 in gastric cancer-association with tumour cell proliferation. J Pathol. 2003;200:16–22.
Tanaka K, Mohri Y, Ohi M, et al. Mitotic checkpoint genes, hsMAD2 and BubR1, in oesophageal squamous cancer cells and their association with 5-fluorouracil and cisplatin-based radiochemotherapy. Clin Oncol (R Coll Radiol). 2008;20(8):639–46.
Sudo T, Nitta M, Saya H, Ueno NT. Dependence of paclitaxel sensitivity on a functional spindle assembly checkpoint. Cancer Res. 2004;64(7):2502–8.
Lee EA, Keutmann MK, Dowling ML, Harris E, Chan G, Kao GD. Inactivation of the mitotic checkpoint as a determinant of the efficacy of microtubule-targeted drugs in killing human cancer cells. Mol Cancer Ther. 2004;3(6):661–9.
Johnson GV, Stoothoff WH. Tau phosphorylation in neuronal cell function and dysfunction. J Cell Sci. 2004;117:5721–9.
Hernández F, Avila J. Tauopathies. Cell Mol Life Sci. 2007;64(17):2219–33.
Feinstein SC, Wilson L. Inability of tau to properly regulate neuronal microtubule dynamics: a loss-of-function mechanism by which tau might mediate neuronal cell death. Biochim Biophys Acta. 2005;1739(2–3):268–79.
Sangrajrang S, Denoulet P, Millot G, et al. Estramustine resistance correlates with tau over-expressıon in human prostatic carcınoma cells. Int J Cancer. 1998;77:626–31.
Mimori K, Sadanaga N, Yoshikawa Y, et al. Reduced tau expression in gastric cancer can identify candidates for successful Paclitaxel treatment. Br J Cancer. 2006;94(12):1894–7.
Jimeno A, Hallur G, Chan A, et al. Development of two novel benzoylphenylurea sulfur analogues and evidence that the microtubule-associated protein tau is predictive of their activity in pancreatic cancer. Mol Cancer Ther. 2007;6:1509–16.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cirak, Y., Sarsik, B., Cakar, B. et al. Predictive and prognostic values of Tau and BubR1 protein in prostate cancer and their relationship to the Gleason score. Med Oncol 30, 526 (2013). https://doi.org/10.1007/s12032-013-0526-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0526-7