Abstract
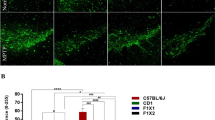
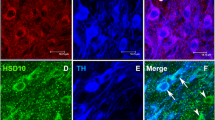
Disease genetics in admixed populations like Hispanic-Americans, African-Americans, etc. are gaining importance due to high disease burden in them. Furthermore, epidemiological studies conclusively prove ethnicity-based differential prevalence of Parkinson’s disease (PD), since the American-Caucasians are more susceptible than Asian-Indians and Africans. Contradictorily, Anglo-Indians, an admixture of Europeans and Asian-Indians are five-times less susceptible than Indians. We evaluated the neural basis of this phenomenon using the cytomorphological features of susceptibility to nigrostriatal neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). The nigral dopaminergic neuronal numbers, their size and tyrosine hydroxylase (TH), PitX3 and Nurr1 expression were compared in MPTP-susceptible C57BL/6J mice, MPTP-resistant CD-1 mice and their crossbreds using stereology, morphometry and densitometry. Apoptotic index was evaluated by TUNEL-assay and caspase-3 expression. Striatal volume, TH and glial derived neurotrophic factor (GDNF) expression were studied. The normal CD-1 and crossbreds had significantly more, although smaller, nigral dopaminergic neurons than C57BL/6J, and a larger striatum. The crossbreds had higher TH, Nurr1 and PitX3 levels. MPTP administration caused loss of ~50–60 % nigral dopaminergic neurons in C57BL/6J and ~15 % in CD-1, but none in crossbreds. MPTP-induced cellular shrinkage in C57BL/6J was contrasted by nuclear enlargement without somal alterations in resistant strains. MPTP lowered the striatal TH and GDNF in C57BL/6J. Elevated striatal GDNF in CD-1 and crossbreds could be of compensatory nature and complemented the reduced nigral caspase-3 expression to attenuate and/or block apoptosis. Similar neural correlates of resilience are envisaged in the Anglo-Indian population. Thus, we present the core neuroanatomical features of resilience against PD and evidence for ethnicity-based differential prevalence.






Similar content being viewed by others
References
Seldin MF, Pasaniuc B, Price AL (2011) New approaches to disease mapping in admixed populations. Nature reviews. Genetics 12(8):523–528. doi:10.1038/nrg3002
Strickland D, Bertoni JM (2004) Parkinson’s prevalence estimated by a state registry. Movement disorders : official journal of the Movement Disorder Society 19(3):318–323. doi:10.1002/mds.10619
Totaro R, Marini C, Pistoia F, Sacco S, Russo T, Carolei A (2005) Prevalence of Parkinson’s disease in the L’Aquila district, central Italy. Acta neurologica. Scandinavica 112(1):24–28. doi:10.1111/j.1600-0404.2005.00426.x
Satake W, Nakabayashi Y, Mizuta I, Hirota Y, Ito C, Kubo M, Kawaguchi T, Tsunoda T, Watanabe M, Takeda A, Tomiyama H, Nakashima K, Hasegawa K, Obata F, Yoshikawa T, Kawakami H, Sakoda S, Yamamoto M, Hattori N, Murata M, Nakamura Y, Toda T (2009) Genome-wide association study identifies common variants at four loci as genetic risk factors for Parkinson’s disease. Nat Genet 41(12):1303–1307. doi:10.1038/ng.485
Simon-Sanchez J, Schulte C, Bras JM, Sharma M, Gibbs JR, Berg D, Paisan-Ruiz C, Lichtner P, Scholz SW, Hernandez DG, Kruger R, Federoff M, Klein C, Goate A, Perlmutter J, Bonin M, Nalls MA, Illig T, Gieger C, Houlden H, Steffens M, Okun MS, Racette BA, Cookson MR, Foote KD, Fernandez HH, Traynor BJ, Schreiber S, Arepalli S, Zonozi R, Gwinn K, van der Brug M, Lopez G, Chanock SJ, Schatzkin A, Park Y, Hollenbeck A, Gao J, Huang X, Wood NW, Lorenz D, Deuschl G, Chen H, Riess O, Hardy JA, Singleton AB, Gasser T (2009) Genome-wide association study reveals genetic risk underlying Parkinson’s disease. Nat Genet 41(12):1308–1312. doi:10.1038/ng.487
Das SK, Misra AK, Ray BK, Hazra A, Ghosal MK, Chaudhuri A, Roy T, Banerjee TK, Raut DK (2010) Epidemiology of Parkinson disease in the city of Kolkata, India: a community-based study. Neurology 75(15):1362–1369. doi:10.1212/WNL.0b013e3181f735a7
Schoenberg BS, Osuntokun BO, Adeuja AO, Bademosi O, Nottidge V, Anderson DW, Haerer AF (1988) Comparison of the prevalence of Parkinson’s disease in black populations in the rural United States and in rural Nigeria: door-to-door community studies. Neurology 38(4):645–646
Ragothaman M, Murgod UA, Gururaj G, Kumaraswamy SD, Muthane U (2003) Lower risk of Parkinson’s disease in an admixed population of European and Indian origins. Movement disorders : official journal of the Movement Disorder Society 18(8):912–914. doi:10.1002/mds.10449
Jellinger K (1986) Recent developments in Parkinson’s disease. Raven, New York
Fahn S (1989) The history of parkinsonism. Movement disorders: official journal of the Movement Disorder Society 4(Suppl 1):S2–10
Chu Y, Kompoliti K, Cochran EJ, Mufson EJ, Kordower JH (2002) Age-related decreases in Nurr1 immunoreactivity in the human substantia nigra. J Comp Neurol 450(3):203–214. doi:10.1002/cne.10261
Alladi PA, Mahadevan A, Yasha TC, Raju TR, Shankar SK, Muthane U (2009) Absence of age-related changes in nigral dopaminergic neurons of Asian Indians: relevance to lower incidence of Parkinson’s disease. Neuroscience 159(1):236–245. doi:10.1016/j.neuroscience.2008.11.051
Cabello CR, Thune JJ, Pakkenberg H, Pakkenberg B (2002) Ageing of substantia nigra in humans: cell loss may be compensated by hypertrophy. Neuropathol Appl Neurobiol 28(4):283–291
Ma SY, Rinne JO, Collan Y, Roytta M, Rinne UK (1996) A quantitative morphometrical study of neuron degeneration in the substantia nigra in Parkinson’s disease. J Neurol Sci 140(1–2):40–45
Muthane U, Yasha TC, Shankar SK (1998) Low numbers and no loss of melanized nigral neurons with increasing age in normal human brains from India. Ann Neurol 43(3):283–287. doi:10.1002/ana.410430304
Muthane UB, Chickabasaviah YT, Henderson J, Kingsbury AE, Kilford L, Shankar SK, Subbakrishna DK, Lees AJ (2006) Melanized nigral neuronal numbers in Nigerian and British individuals. Movement disorders : official journal of the Movement Disorder Society 21(8):1239–1241. doi:10.1002/mds.20917
Boyd JD, Jang H, Shepherd KR, Faherty C, Slack S, Jiao Y, Smeyne RJ (2007) Response to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) differs in mouse strains and reveals a divergence in JNK signaling and COX-2 induction prior to loss of neurons in the substantia nigra pars compacta. Brain Res 1175:107–116. doi:10.1016/j.brainres.2007.07.067
Jones BC, Miller DB, O'Callaghan JP, Lu L, Unger EL, Alam G, Williams RW (2013) Systems analysis of genetic variation in MPTP neurotoxicity in mice. Neurotoxicology 37:26–34. doi:10.1016/j.neuro.2013.03.010
Baker H, Joh TH, Reis DJ (1980) Genetic control of number of midbrain dopaminergic neurons in inbred strains of mice: relationship to size and neuronal density of the striatum. Proc Natl Acad Sci U S A 77(7):4369–4373
Rosen GD, Williams RW (2001) Complex trait analysis of the mouse striatum: independent QTLs modulate volume and neuron number. BMC Neurosci 2:5
Vadasz C, Smiley JF, Figarsky K, Saito M, Toth R, Gyetvai BM, Oros M, Kovacs KK, Mohan P, Wang R (2007) Mesencephalic dopamine neuron number and tyrosine hydroxylase content: genetic control and candidate genes. Neuroscience 149(3):561–572. doi:10.1016/j.neuroscience.2007.06.049
Schwarting RK, Sedelis M, Hofele K, Auburger GW, Huston JP (1999) Strain-dependent recovery of open-field behavior and striatal dopamine deficiency in the mouse MPTP model of Parkinson’s disease. Neurotox Res 1(1):41–56
Hamre K, Tharp R, Poon K, Xiong X, Smeyne RJ (1999) Differential strain susceptibility following 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) administration acts in an autosomal dominant fashion: quantitative analysis in seven strains of Mus musculus. Brain Res 828(1–2):91–103
Cook R, Lu L, Gu J, Williams RW, Smeyne RJ (2003) Identification of a single QTL, Mptp1, for susceptibility to MPTP-induced substantia nigra pars compacta neuron loss in mice. Brain Res Mol Brain Res 110(2):279–288
Sedelis M, Hofele K, Schwarting RK, Huston JP, Belknap JK (2003) Chromosomal loci influencing the susceptibility to the parkinsonian neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. The Journal of neuroscience: the official journal of the Society for Neuroscience 23(23):8247–8253
Jiao Y, Lu L, Williams RW, Smeyne RJ (2012) Genetic dissection of strain dependent paraquat-induced neurodegeneration in the substantia nigra pars compacta. PLoS One 7 (1):e29447. doi:10.1371/journal.pone.0029447
Fu Y, Yuan Y, Halliday G, Rusznak Z, Watson C, Paxinos G (2012) A cytoarchitectonic and chemoarchitectonic analysis of the dopamine cell groups in the substantia nigra, ventral tegmental area, and retrorubral field in the mouse. Brain Struct Funct 217(2):591–612. doi:10.1007/s00429-011-0349-2
Baquet ZC, Williams D, Brody J, Smeyne RJ (2009) A comparison of model-based (2D) and design-based (3D) stereological methods for estimating cell number in the substantia nigra pars compacta (SNpc) of the C57BL/6J mouse. Neuroscience 161(4):1082–1090. doi:10.1016/j.neuroscience.2009.04.031
Vidyadhara DJ, Yarreiphang H, Abhilash PL, Raju TR, Alladi PA (2016) Differential expression of calbindin in nigral dopaminergic neurons in two mice strains with differential susceptibility to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. J Chem Neuroanat. doi:10.1016/j.jchemneu.2016.01.001
Alladi PA, Mahadevan A, Shankar SK, Raju TR, Muthane U (2010) Expression of GDNF receptors GFRalpha1 and RET is preserved in substantia nigra pars compacta of aging Asian Indians. J Chem Neuroanat 40(1):43–52. doi:10.1016/j.jchemneu.2010.03.007
Jyothi HJ, Vidyadhara DJ, Mahadevan A, Philip M, Parmar SK, Manohari SG, Shankar SK, Raju TR, Alladi PA (2015) Aging causes morphological alterations in astrocytes and microglia in human substantia nigra pars compacta. Neurobiol Aging 36(12):3321–3333. doi:10.1016/j.neurobiolaging.2015.08.024
Alladi PA, Wadhwa S, Singh N (2002) Effect of prenatal auditory enrichment on developmental expression of synaptophysin and syntaxin 1 in chick brainstem auditory nuclei. Neuroscience 114(3):577–590
Franklin GPaKBJ (2013) The mouse brain in stereotaxic coordinates. Fourth edn. Academic Press, USA
Alladi PA, Mahadevan A, Vijayalakshmi K, Muthane U, Shankar SK, Raju TR (2010) Ageing enhances alpha-synuclein, ubiquitin and endoplasmic reticular stress protein expression in the nigral neurons of Asian Indians. Neurochem Int 57(5):530–539. doi:10.1016/j.neuint.2010.06.018
Sundstrom E, Samuelsson EB (1997) Comparison of key steps in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxicity in rodents. Pharmacology & toxicology 81(5):226–231
Przedborski S, Jackson-Lewis V, Naini AB, Jakowec M, Petzinger G, Miller R, Akram M (2001) The parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP): a technical review of its utility and safety. J Neurochem 76(5):1265–1274
Muthane U, Ramsay KA, Jiang H, Jackson-Lewis V, Donaldson D, Fernando S, Ferreira M, Przedborski S (1994) Differences in nigral neuron number and sensitivity to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in C57/bl and CD-1 mice. Exp Neurol 126(2):195–204. doi:10.1006/exnr.1994.1058
Sedelis M, Hofele K, Auburger GW, Morgan S, Huston JP, Schwarting RK (2000) Evidence for resistance to MPTP in C57BL/6 x BALB/c F1 hybrids as compared with their progenitor strains. Neuroreport 11(5):1093–1096
Das SK, Ghosh B, Das G, Biswas A, Ray J (2013) Movement disorders: Indian scenario: a clinico-genetic review. Neurol India 61(5):457–466. doi:10.4103/0028-3886.121908
Surathi P, Jhunjhunwala K, Yadav R, Pal PK (2016) Research in Parkinson's disease in India: a review. Annals of Indian Academy of Neurology 19(1):9–20. doi:10.4103/0972-2327.167713
West MJ (1993) New stereological methods for counting neurons. Neurobiol Aging 14(4):275–285
Ikeda M, Levitt M, Udenfriend S (1965) Hydroxylation of phenylalanine by purified preparations of adrenal and brain tyrosine hydroxylase. Biochem Biophys Res Commun 18:482–488
Veenvliet JV, Smidt MP (2014) Molecular mechanisms of dopaminergic subset specification: fundamental aspects and clinical perspectives. Cellular and molecular life sciences : CMLS 71(24):4703–4727. doi:10.1007/s00018-014-1681-5
Decressac M, Volakakis N, Bjorklund A, Perlmann T (2013) NURR1 in Parkinson disease—from pathogenesis to therapeutic potential. Nature reviews. Neurology 9(11):629–636. doi:10.1038/nrneurol.2013.209
Wang X, Michaelis EK (2010) Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci 2:12. doi:10.3389/fnagi.2010.00012
Itoh K, Weis S, Mehraein P, Muller-Hocker J (1997) Defects of cytochrome c oxidase in the substantia nigra of Parkinson’s disease: and immunohistochemical and morphometric study. Movement disorders: official journal of the Movement Disorder Society 12(1):9–16. doi:10.1002/mds.870120104
Langston JW, Forno LS, Rebert CS, Irwin I (1984) Selective nigral toxicity after systemic administration of 1-methyl-4-phenyl-1,2,5,6-tetrahydropyrine (MPTP) in the squirrel monkey. Brain Res 292(2):390–394
Jackson-Lewis V, Jakowec M, Burke RE, Przedborski S (1995) Time course and morphology of dopaminergic neuronal death caused by the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Neurodegeneration: a journal for neurodegenerative disorders, neuroprotection, and neuroregeneration 4(3):257–269
Harris GJ, Codori AM, Lewis RF, Schmidt E, Bedi A, Brandt J (1999) Reduced basal ganglia blood flow and volume in pre-symptomatic, gene-tested persons at-risk for Huntington’s disease. Brain: a journal of neurology 122(Pt 9):1667–1678
Rosas HD, Goodman J, Chen YI, Jenkins BG, Kennedy DN, Makris N, Patti M, Seidman LJ, Beal MF, Koroshetz WJ (2001) Striatal volume loss in HD as measured by MRI and the influence of CAG repeat. Neurology 57(6):1025–1028
Voelbel GT, Bates ME, Buckman JF, Pandina G, Hendren RL (2006) Caudate nucleus volume and cognitive performance: are they related in childhood psychopathology? Biol Psychiatry 60(9):942–950. doi:10.1016/j.biopsych.2006.03.071
Kreczmanski P, Heinsen H, Mantua V, Woltersdorf F, Masson T, Ulfig N, Schmidt-Kastner R, Korr H, Steinbusch HW, Hof PR, Schmitz C (2007) Volume, neuron density and total neuron number in five subcortical regions in schizophrenia. Brain : a journal of neurology 130(Pt 3):678–692. doi:10.1093/brain/awl386
Reiss AL, Faruque F, Naidu S, Abrams M, Beaty T, Bryan RN, Moser H (1993) Neuroanatomy of Rett syndrome: a volumetric imaging study. Ann Neurol 34(2):227–234. doi:10.1002/ana.410340220
Rosen GD, Pung CJ, Owens CB, Caplow J, Kim H, Mozhui K, Lu L, Williams RW (2009) Genetic modulation of striatal volume by loci on Chrs 6 and 17 in BXD recombinant inbred mice. Genes Brain Behav 8(3):296–308. doi:10.1111/j.1601-183X.2009.00473.x
Wu DC, Jackson-Lewis V, Vila M, Tieu K, Teismann P, Vadseth C, Choi DK, Ischiropoulos H, Przedborski S (2002) Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. The Journal of neuroscience: the official journal of the Society for Neuroscience 22(5):1763–1771
Benner EJ, Mosley RL, Destache CJ, Lewis TB, Jackson-Lewis V, Gorantla S, Nemachek C, Green SR, Przedborski S, Gendelman HE (2004) Therapeutic immunization protects dopaminergic neurons in a mouse model of Parkinson’s disease. Proc Natl Acad Sci U S A 101(25):9435–9440. doi:10.1073/pnas.0400569101
Aguirre JA, Kehr J, Yoshitake T, Liu FL, Rivera A, Fernandez-Espinola S, Andbjer B, Leo G, Medhurst AD, Agnati LF, Fuxe K (2005) Protection but maintained dysfunction of nigral dopaminergic nerve cell bodies and striatal dopaminergic terminals in MPTP-lesioned mice after acute treatment with the mGluR5 antagonist MPEP. Brain Res 1033(2):216–220. doi:10.1016/j.brainres.2004.11.040
Tande D, Hoglinger G, Debeir T, Freundlieb N, Hirsch EC, Francois C (2006) New striatal dopamine neurons in MPTP-treated macaques result from a phenotypic shift and not neurogenesis. Brain: a journal of neurology 129(Pt 5):1194–1200. doi:10.1093/brain/awl041
Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F (1993) GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science (New York, NY) 260(5111):1130–1132
Tomac A, Lindqvist E, Lin LF, Ogren SO, Young D, Hoffer BJ, Olson L (1995) Protection and repair of the nigrostriatal dopaminergic system by GDNF in vivo. Nature 373(6512):335–339. doi:10.1038/373335a0
Kordower JH, Emborg ME, Bloch J, Ma SY, Chu Y, Leventhal L, McBride J, Chen EY, Palfi S, Roitberg BZ, Brown WD, Holden JE, Pyzalski R, Taylor MD, Carvey P, Ling Z, Trono D, Hantraye P, Deglon N, Aebischer P (2000) Neurodegeneration prevented by lentiviral vector delivery of GDNF in primate models of Parkinson’s disease. Science (New York, NY) 290(5492):767–773
Burke RE, Antonelli M, Sulzer D (1998) Glial cell line-derived neurotrophic growth factor inhibits apoptotic death of postnatal substantia nigra dopamine neurons in primary culture. J Neurochem 71(2):517–525
Oo TF, Kholodilov N, Burke RE (2003) Regulation of natural cell death in dopaminergic neurons of the substantia nigra by striatal glial cell line-derived neurotrophic factor in vivo. The Journal of neuroscience: the official journal of the Society for Neuroscience 23(12):5141–5148
Li X, Peng C, Li L, Ming M, Yang D, Le W (2007) Glial cell-derived neurotrophic factor protects against proteasome inhibition-induced dopamine neuron degeneration by suppression of endoplasmic reticulum stress and caspase-3 activation. J Gerontol A Biol Sci Med Sci 62(9):943–950
Hartmann A, Hunot S, Michel PP, Muriel MP, Vyas S, Faucheux BA, Mouatt-Prigent A, Turmel H, Srinivasan A, Ruberg M, Evan GI, Agid Y, Hirsch EC (2000) Caspase-3: a vulnerability factor and final effector in apoptotic death of dopaminergic neurons in Parkinson’s disease. Proc Natl Acad Sci U S A 97(6):2875–2880. doi:10.1073/pnas.040556597
Kramer ER, Aron L, Ramakers GM, Seitz S, Zhuang X, Beyer K, Smidt MP, Klein R (2007) Absence of Ret signaling in mice causes progressive and late degeneration of the nigrostriatal system. PLoS Biol 5(3):e39. doi:10.1371/journal.pbio.0050039
Pascual A, Hidalgo-Figueroa M, Piruat JI, Pintado CO, Gomez-Diaz R, Lopez-Barneo J (2008) Absolute requirement of GDNF for adult catecholaminergic neuron survival. Nat Neurosci 11(7):755–761. doi:10.1038/nn.2136
Boger HA, Middaugh LD, Zaman V, Hoffer B, Granholm AC (2008) Differential effects of the dopamine neurotoxin MPTP in animals with a partial deletion of the GDNF receptor, GFR alpha1, gene. Brain Res 1241:18–28. doi:10.1016/j.brainres.2008.09.011
Winkler C, Sauer H, Lee CS, Bjorklund A (1996) Short-term GDNF treatment provides long-term rescue of lesioned nigral dopaminergic neurons in a rat model of Parkinson's disease. The Journal of neuroscience: the official journal of the Society for Neuroscience 16(22):7206–7215
Kirik D, Rosenblad C, Bjorklund A, Mandel RJ (2000) Long-term rAAV-mediated gene transfer of GDNF in the rat Parkinson's model: intrastriatal but not intranigral transduction promotes functional regeneration in the lesioned nigrostriatal system. The Journal of neuroscience: the official journal of the Society for Neuroscience 20(12):4686–4700
Garcia-Valenzuela E, Gorczyca W, Darzynkiewicz Z, Sharma SC (1994) Apoptosis in adult retinal ganglion cells after axotomy. J Neurobiol 25(4):431–438. doi:10.1002/neu.480250408
Jackson-Lewis V, Przedborski S (2007) Protocol for the MPTP mouse model of Parkinson’s disease. Nat Protoc 2(1):141–151. doi:10.1038/nprot.2006.342
Acknowledgments
The study was funded by the Science and Engineering Research Board, Department of Science and Technology, Govt. of India to PAA (Project no. SR/SO/HS-0121/2012). VDJ is a NIMHANS fellow; HY is a UGC fellow. The authors are grateful to Dr. Bindu M. Kutty, Prof. and Head, Dept. of Neurophysiology, NIMHANS for extending laboratory facilities, Dr. G.H. Mohan, Head Veterinarian at National Centre for Biological Sciences, Bengaluru for providing breeding colonies of CD-1 mice strain, Mr. Niranjan S. for standardizing immunoblots and Ms. Vaidehi Kelkar for densitometric quantification of few striatal samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vidyadhara, D.J., Yarreiphang, H., Raju, T.R. et al. Admixing of MPTP-Resistant and Susceptible Mice Strains Augments Nigrostriatal Neuronal Correlates to Resist MPTP-Induced Neurodegeneration. Mol Neurobiol 54, 6148–6162 (2017). https://doi.org/10.1007/s12035-016-0158-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-0158-y