Abstract
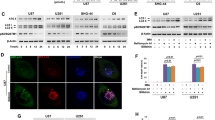
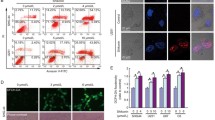
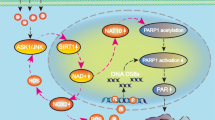
Parthanatos is a form of PARP-1-dependent programmed cell death. The induction of parthanatos is emerging as a new strategy to kill gliomas which are the most common type of primary malignant brain tumor. Oxidative stress is thought to be a critical factor triggering parthanatos, but its underlying mechanism is poorly understood. In this study, we used glioma cell lines and H2O2 to investigate the role of JNK in glioma cell parthanatos induced by oxidative stress. We found that exposure to H2O2 not only induced intracellular accumulation of ROS but also resulted in glioma cell death in a concentration- and incubation time-dependent manner, which was accompanied with cytoplasmic formation of PAR polymer, expressional upregulation of PARP-1, mitochondrial depolarization, and AIF translocation to nucleus. Pharmacological inhibition of PARP-1 with 3AB or genetic knockdown of its level with siRNA rescued glioma cell death, as well as suppressed cytoplasmic accumulation of PAR polymer and nuclear translocation of AIF, which were consistent with the definition of parthanatos. Moreover, the phosphorylated level of JNK increased markedly with the extension of H2O2 exposure time. Either attenuation of intracellular ROS with antioxidant NAC or inhibition of JNK phosphorylation with SP600125 or JNK siRNA could significantly prevent H2O2-induced parthanatos in glioma cells. Additionally, inhibition of JNK with SP600125 alleviated intracellular accumulation of ROS and attenuated mitochondrial generation of superoxide. Thus, we demonstrated that JNK activation contributes to glioma cell parthanatos caused by oxidative stress via increase of intracellular ROS generation.









Similar content being viewed by others
Abbreviations
- PARP-1:
-
Poly(ADP-ribose) synthetase 1
- PAR:
-
Poly(ADP-ribose)
- NAD+:
-
Nicotinamide adenine dinucleotide
- MAPK:
-
Mitogen-activated protein kinase
- JNK:
-
c-Jun-N-terminal protein kinase
- ERK1/2:
-
Extracellular signal regulated kinases 1 and 2
- AIF:
-
Apoptosis inducing factor
- RIP-1:
-
Receptor interacting protein-1
- ROS:
-
Reactive oxygen species
- LDH:
-
Lactate dehydrogenase
- Nec-1:
-
Necrostatin-1
- 3AB:
-
3-Aminobenzamide
- NAC:
-
N-acetyl-l-cysteine
References
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507
Beck C, Robert I, Reina-San-Martin B, Schreiber V, Dantzer F (2014) Poly(ADP-ribose) polymerases in double-strand break repair: focus on PARP1, PARP2 and PARP3. Exp Cell Res 329:18–25
Fatokun AA, Dawson VL, Dawson TM (2014) Parthanatos: mitochondrial-linked mechanisms and therapeutic opportunities. Br J Pharmacol 171:2000–2016
Galia A, Calogero AE, Condorelli R, Fraggetta F, La Corte A, Ridolfo F, Bosco P, Castiglione R et al (2012) PARP-1 protein expression in glioblastoma multiforme. Eur J Histochem 56, e9
Kase M, Vardja M, Lipping A, Asser T, Jaal J (2011) Impact of PARP-1 and DNA-PK expression on survival in patients with glioblastoma multiforme. Radiother Oncol 101:127–131
Karpel-Massler G, Pareja F, Aimé P, Shu C, Chau L, Westhoff MA, Halatsch ME, Crary JF et al (2014) PARP inhibition restores extrinsic apoptotic sensitivity in glioblastoma. PLoS One 9, e114583
Tentori L, Ricci-Vitiani L, Muzi A, Ciccarone F, Pelacchi F, Calabrese R, Runci D, Pallini R et al (2014) Pharmacological inhibition of poly(ADP-ribose) polymerase-1 modulates resistance of human glioblastoma stem cells to temozolomide. BMC Cancer 14:151
Ma D, Lu B, Feng C, Wang C, Wang Y, Luo T, Feng J, Jia H et al (2016) Deoxypodophyllotoxin triggers parthanatos in glioma cells via induction of excessive ROS. Cancer Lett 371:194–204
Zhao N, Mao Y, Han G, Ju Q, Zhou L, Liu F, Xu Y, Zhao X (2015) YM155, a survivin suppressant, triggers PARP-dependent cell death (parthanatos) and inhibits esophageal squamous-cell carcinoma xenografts in mice. Oncotarget 6:18445–18459
Lee Y, Kang HC, Lee BD, Lee YI, Kim YP, Shin JH (2014) Poly (ADP-ribose) in the pathogenesis of Parkinson’s disease. BMB Rep 47:424–432
Mohammad G, Siddiquei MM, Abu El-Asrar AM (2013) Poly (ADP-ribose) polymerase mediates diabetes-induced retinal neuropathy. Mediat Inflamm 2013:510451
Yang Z, Li L, Chen L, Yuan W, Dong L, Zhang Y, Wu H, Wang C (2014) PARP-1 mediates LPS-induced HMGB1 release by macrophages through regulation of HMGB1 acetylation. J Immunol 193:6114–6123
Lu P, Kamboj A, Gibson SB, Anderson CM (2014) Poly(ADP-ribose) polymerase-1 causes mitochondrial damage and neuron death mediated by Bnip3. J Neurosci 34:15975–15987
Wu P, Zhu X, Jin W, Hao S, Liu Q (2015) Oxaliplatin triggers necrosis as well as apoptosis in gastric cancer SGC-7901 cells. Biochem Biophys Res Commun 460:183–190
Chiu LY, Ho FM, Shiah SG, Chang Y, Lin WW (2011) Oxidative stress initiates DNA damager MNNG-induced poly(ADP-ribose)polymerase-1-dependent parthanatos cell death. Biochem Pharmacol 81:459–470
Akhiani AA, Werlenius O, Aurelius J, Movitz C, Martner A, Hellstrand K, Thorén FB (2014) Role of the ERK pathway for oxidant-induced parthanatos in human lymphocytes. PLoS One 9, e89646
Zhang L, Wang H, Xu J, Zhu J, Ding K (2014) Inhibition of cathepsin S induces autophagy and apoptosis in human glioblastoma cell lines through ROS-mediated PI3K/AKT/mTOR/p70S6K and JNK signaling pathways. Toxicol Lett 228:248–259
Barbouti A, Doulias PT, Nousis L, Tenopoulou M, Galaris D (2002) DNA damage and apoptosis in hydrogen peroxide-exposed Jurkat cells: bolus addition versus continuous generation of H(2)O(2). Free Radic Biol Med 33:691–702
Mao Y, Song G, Cai Q, Liu M, Luo H, Shi M, Ouyang G, Bao S (2006) Hydrogen peroxide-induced apoptosis in human gastric carcinoma MGC803 cells. Cell Biol Int 30:332–337
Singh M, Sharma H, Singh N (2007) Hydrogen peroxide induces apoptosis in HeLa cells through mitochondrial pathway. Mitochondrion 7:367–373
Wang X, Wang J, Lin S, Geng Y, Wang J, Jiang B (2008) Sp1 is involved in H2O2-induced PUMA gene expression and apoptosis in colorectal cancer cells. J Exp Clin Cancer Res 27:44
McKeague AL, Wilson DJ, Nelson J (2008) Staurosporine-induced apoptosis and hydrogen peroxide-induced necrosis in two human breast cell lines. Br J Cancer 88:125–131
Datta K, Babbar P, Srivastava T, Sinha S, Chattopadhyay P (2002) p53 dependent apoptosis in glioma cell lines in response to hydrogen peroxide induced oxidative stress. Int J Biochem Cell Biol 34:148–157
Byun YJ, Kim SK, Kim YM, Chae GT, Jeong SW, Lee SB (2009) Hydrogen peroxide induces autophagic cell death in C6 glioma cells via BNIP3-mediated suppression of the mTOR pathway. Neurosci Lett 461:131–135
Chen Y, McMillan-Ward E, Kong J, Israels SJ, Gibson SB (2008) Oxidative stress induces autophagic cell death independent of apoptosis in transformed and cancer cells. Cell Death Differ 15:171–182
Zhang H, Kong X, Kang J, Su J, Li Y, Zhong J, Sun L (2009) Oxidative stress induces parallel autophagy and mitochondria dysfunction in human glioma U251 cells. Toxicol Sci 110:376–388
Lennicke C, Rahn J, Lichtenfels R, Wessjohann LA, Seliger B (2015) Hydrogen peroxide-production, fate and role in redox signaling of tumor cells. Cell Commun Signal 13:39
Wang Y, Dawson VL, Dawson TM (2009) Poly(ADP-ribose) signals to mitochondrial AIF: a key event in parthanatos. Exp Neurol 218:193–202
Mashimo M, Kato J, Moss J (2013) ADP-ribosyl-acceptor hydrolase 3 regulates poly (ADP-ribose) degradation and cell death during oxidative stress. Proc Natl Acad Sci U S A 110:18964–18969
Zhao H, Ning J, Lemaire A, Koumpa FS, Sun JJ, Fung A, Gu J, Yi B et al (2015) Necroptosis and parthanatos are involved in remote lung injury after receiving ischemic renal allografts in rats. Kidney Int 87:738–748
Andrabi SA, Kim NS, Yu S, Wang H, Koh DW, Sasaki M, Klaus JA, Otsuka T et al (2006) Poly(ADP-ribose) (PAR) polymer is a death signal. Proc Natl Acad Sci U S A 103:18308–18313
Andrabi SA, Umanah GK, Chang C, Stevens DA, Karuppagounder SS, Gagné JP, Poirier GG, Dawson VL et al (2014) Poly(ADP-ribose) polymerase-dependent energy depletion occurs through inhibition of glycolysis. Proc Natl Acad Sci U S A 28:10209–10214
Paul M, Hemshekhar M, Thushara RM, Sundaram MS, NaveenKumar SK, Naveen S, Devaraja S, Somyajit K et al (2015) Methotrexate promotes platelet apoptosis via JNK-mediated mitochondrial damage: alleviation by N-acetylcysteine and N-acetylcysteine amide. PLoS One 10, e0127558
Dixit D, Ghildiyal R, Anto NP, Sen E (2014) Chaetocin-induced ROS-mediated apoptosis involves ATM-YAP1 axis and JNK-dependent inhibition of glucose metabolism. Cell Death Dis 5, e1212
Zhang C, Yang L, Wang XB, Wang JS, Geng YD, Yang CS, Kong LY (2013) Calyxin Y induces hydrogen peroxide-dependent autophagy and apoptosis via JNK activation in human non-small cell lung cancer NCI-H460 cells. Cancer Lett 340:51–62
Palit S, Kar S, Sharma G, Das PK (2015) Hesperetin induces apoptosis in breast carcinoma by triggering accumulation of ROS and activation of ASK1/JNK pathway. J Cell Physiol 230:1729–1739
Nomura J, Busso N, Ives A, Tsujimoto S, Tamura M, So A, Yamanaka Y (2013) Febuxostat, an inhibitor of xanthine oxidase, suppresses lipopolysaccharide-induced MCP-1 production via MAPK phosphatase-1-mediated inactivation of JNK. PLoS One 8, e75527
Zhou JY, Liu Y, Wu GS (2006) The role of mitogen-activated protein kinase phosphatase-1 in oxidative damage-induced cell death. Cancer Res 66:4888–4894
Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N (2008) Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem 283:13565–13577
Win S, Than TA, Le BH, García-Ruiz C, Fernandez-Checa JC, Kaplowitz N (2015) Sab (Sh3bp5) dependence of JNK mediated inhibition of mitochondrial respiration in palmitic acid induced hepatocyte lipotoxicity. J Hepatol 62:1367–1374
Acknowledgments
This work was supported by the National Nature Science Foundation of China (81171234, 81372697, 11432016, and 11272134), the Changbaishan Scholar Project of Jilin province (2013026), the Scientific Research Foundation of Jilin province (20150414013GH, 20121809), and the Bethune project B of Jilin University (no.2012203).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Zheng, L., Wang, C., Luo, T. et al. JNK Activation Contributes to Oxidative Stress-Induced Parthanatos in Glioma Cells via Increase of Intracellular ROS Production. Mol Neurobiol 54, 3492–3505 (2017). https://doi.org/10.1007/s12035-016-9926-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9926-y