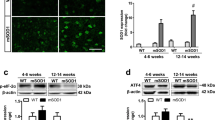
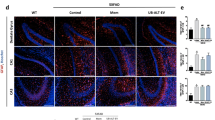
Abstract
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease of unknown cause. Absence of specific targets and biomarkers compromise the development of new therapeutic strategies and of innovative tools to stratify patients and assess their responses to treatment. Here, we investigate changes in neuroprotective-neuroinflammatory actions in the spinal cord of SOD1 G93A mice, at presymptomatic and symptomatic stages to identify stage-specific biomarkers and potential targets. Results showed that in the presymptomatic stage, there are alterations in both astrocytes and microglia, which comprise decreased expression of GFAP and S100B and upregulation of GLT-1, as well as reduced expression of CD11b, M2-phenotype markers, and a set of inflammatory mediators. Reduced levels of Connexin-43, Pannexin-1, CCL21, and CX3CL1 further indicate the existence of a compromised intercellular communication. In contrast, in the symptomatic stage, increased markers of inflammation became evident, such as NF-κB/Nlrp3-inflammasome, Iba1, pro-inflammatory cytokines, and M1-polarizion markers, together with a decreased expression of M2-phenotypic markers. We also observed upregulation of the CX3CL1-CX3CR1 axis, Connexin-43, Pannexin-1, and of microRNAs (miR)-124, miR-125b, miR-146a and miR-21. Reduced motor neuron number and presence of reactive astrocytes with decreased GFAP, GLT-1, and GLAST further characterized this inflammatory stage. Interestingly, upregulation of miR-155 and downregulation of MFG-E8 appear as consistent biomarkers of both presymptomatic and symptomatic stages. We hypothesize that downregulated cellular interplay at the early stages may represent neuroprotective mechanisms against inflammation, SOD1 aggregation, and ALS onset. The present study identified a set of inflamma-miRNAs, NLRP3-inflammasome, HMGB1, CX3CL1-CX3CR1, Connexin-43, and Pannexin-1 as emerging candidates and promising pharmacological targets that may represent potential neuroprotective strategies in ALS therapy.







Similar content being viewed by others
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- Arg1:
-
Arginase 1
- Cebpa:
-
CCAAT/enhancer-binding protein alpha
- CCL21:
-
Chemokine (C-C motif) ligand 21
- CCR7:
-
Chemokine (C-C motif) receptor type 7
- CSF:
-
Cerebrospinal fluid
- CX3CL1:
-
Chemokine (C-X3-C motif) ligand 1
- CX3CR1:
-
Chemokine (C-X3-C motif) receptor 1
- Cx43:
-
Connexin-43
- GFAP:
-
Glial fibrillary acidic protein
- GLT-1:
-
Glutamate transporter 1
- Fizz1:
-
Found in inflammatory zone 1
- HMGB1:
-
High mobility group box 1
- H&E:
-
hematoxylin–eosin
- Iba1:
-
Ionized calcium-binding adapter molecule 1
- IL:
-
Interleukin
- miR:
-
MicroRNA
- MFG-E8:
-
Milk fat globule-EGF factor 8
- NeuN:
-
Neuronal nuclei
- NF-κB:
-
Nuclear factor kappa B
- NLRP3:
-
NOD-like receptor family pyrin domain containing 3
- Panx1:
-
Pannexin-1
- PBS:
-
Phosphate buffer saline
- pNF-κB:
-
Phosphorylated NF-κB
- qRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- RT:
-
Room temperature
- S100B:
-
S100 calcium-binding protein B
- SC:
-
Spinal cord
- SOCS1:
-
Suppressor of cytokine signaling 1
- SOD1:
-
Superoxide dismutase 1
- TLRs:
-
Toll-like receptors
- TGF-β:
-
Transforming growth factor beta
- TNF-α:
-
Tumor necrosis factor alfa
- WB:
-
Western blot
- WT:
-
Wild type
References
Benatar M, Wuu J (2012) Presymptomatic studies in ALS: rationale, challenges, and approach. Neurology 79(16):1732–1739. doi:10.1212/WNL.0b013e31826e9b1d
Rocha MC, Pousinha PA, Correia AM, Sebastiao AM, Ribeiro JA (2013) Early changes of neuromuscular transmission in the SOD1(G93A) mice model of ALS start long before motor symptoms onset. PLoS One 8(9):e73846. doi:10.1371/journal.pone.0073846
Nascimento F, Pousinha PA, Correia AM, Gomes R, Sebastiao AM, Ribeiro JA (2014) Adenosine A2A receptors activation facilitates neuromuscular transmission in the pre-symptomatic phase of the SOD1(G93A) ALS mice, but not in the symptomatic phase. PLoS One 9(8):e104081. doi:10.1371/journal.pone.0104081
Nascimento F, Sebastiao AM, Ribeiro JA (2015) Presymptomatic and symptomatic ALS SOD1(G93A) mice differ in adenosine A1 and A2A receptor-mediated tonic modulation of neuromuscular transmission. Purinergic Signal 11(4):471–480. doi:10.1007/s11302-015-9465-4
Gravel M, Beland LC, Soucy G, Abdelhamid E, Rahimian R, Gravel C, Kriz J (2016) IL-10 controls early microglial phenotypes and disease onset in ALS caused by misfolded superoxide dismutase 1. J Neurosci 36(3):1031–1048. doi:10.1523/JNEUROSCI.0854-15.2016
Tefera TW, Wong Y, Barkl-Luke ME, Ngo ST, Thomas NK, McDonald TS, Borges K (2016) Triheptanoin protects motor neurons and delays the onset of motor symptoms in a mouse model of amyotrophic lateral sclerosis. PLoS One 11(8):e0161816. doi:10.1371/journal.pone.0161816
Apolloni S, Fabbrizio P, Amadio S, Volonte C (2016) Actions of the antihistaminergic clemastine on presymptomatic SOD1-G93A mice ameliorate ALS disease progression. J Neuroinflammation 13(1):191. doi:10.1186/s12974-016-0658-8
Gaiottino J, Norgren N, Dobson R, Topping J, Nissim A, Malaspina A, Bestwick JP, Monsch AU et al (2013) Increased neurofilament light chain blood levels in neurodegenerative neurological diseases. PLoS One 8(9):e75091. doi:10.1371/journal.pone.0075091
Chio A, Mora G, Leone M, Mazzini L, Cocito D, Giordana MT, Bottacchi E, Mutani R (2002) Early symptom progression rate is related to ALS outcome: a prospective population-based study. Neurology 59(1):99–103
Turner M, Al-Chalabi A (2002) Early symptom progression rate is related to ALS outcome: a prospective population-based study. Neurology 59(12):2012–2013 author reply 2013
Benatar M, Stanislaw C, Reyes E, Hussain S, Cooley A, Fernandez MC, Dauphin DD, Michon SC et al (2016) Presymptomatic ALS genetic counseling and testing: experience and recommendations. Neurology 86(24):2295–2302. doi:10.1212/WNL.0000000000002773
Turner MR, Cagnin A, Turkheimer FE, Miller CC, Shaw CE, Brooks DJ, Leigh PN, Banati RB (2004) Evidence of widespread cerebral microglial activation in amyotrophic lateral sclerosis: an [11C](R)-PK11195 positron emission tomography study. Neurobiol Dis 15(3):601–609. doi:10.1016/j.nbd.2003.12.012
Brites D, Vaz AR (2014) Microglia centered pathogenesis in ALS: insights in cell interconnectivity. Front Cell Neurosci 8:117. doi:10.3389/fncel.2014.00117
Hensley K, Floyd RA, Gordon B, Mou S, Pye QN, Stewart C, West M, Williamson K (2002) Temporal patterns of cytokine and apoptosis-related gene expression in spinal cords of the G93A-SOD1 mouse model of amyotrophic lateral sclerosis. J Neurochem 82(2):365–374
Casula M, Iyer AM, Spliet WG, Anink JJ, Steentjes K, Sta M, Troost D, Aronica E (2011) Toll-like receptor signaling in amyotrophic lateral sclerosis spinal cord tissue. Neuroscience 179:233–243. doi:10.1016/j.neuroscience.2011.02.001
Lee JY, Lee JD, Phipps S, Noakes PG, Woodruff TM (2015) Absence of toll-like receptor 4 (TLR4) extends survival in the hSOD1 G93A mouse model of amyotrophic lateral sclerosis. J Neuroinflammation 12:90. doi:10.1186/s12974-015-0310-z
Lo Coco D, Veglianese P, Allievi E, Bendotti C (2007) Distribution and cellular localization of high mobility group box protein 1 (HMGB1) in the spinal cord of a transgenic mouse model of ALS. Neurosci Lett 412(1):73–77. doi:10.1016/j.neulet.2006.10.063
Johann S, Heitzer M, Kanagaratnam M, Goswami A, Rizo T, Weis J, Troost D, Beyer C (2015) NLRP3 inflammasome is expressed by astrocytes in the SOD1 mouse model of ALS and in human sporadic ALS patients. Glia 63(12):2260–2273. doi:10.1002/glia.22891
Campos-Melo D, Droppelmann CA, He Z, Volkening K, Strong MJ (2013) Altered microRNA expression profile in amyotrophic lateral sclerosis: a role in the regulation of NFL mRNA levels. Molecular brain 6:26. doi:10.1186/1756-6606-6-26
Koval ED, Shaner C, Zhang P, du Maine X, Fischer K, Tay J, Chau BN, Wu GF et al (2013) Method for widespread microRNA-155 inhibition prolongs survival in ALS-model mice. Hum Mol Genet 22(20):4127–4135. doi:10.1093/hmg/ddt261
Butovsky O, Jedrychowski MP, Cialic R, Krasemann S, Murugaiyan G, Fanek Z, Greco DJ, Wu PM et al (2015) Targeting miR-155 restores abnormal microglia and attenuates disease in SOD1 mice. Ann Neurol 77(1):75–99. doi:10.1002/ana.24304
ALSA (2017) miRagen therapeutics receives ALS Association grant to advance the development of MRG-107. http://www.alsa.org/news/media/press-releases/miragen-therapeutics-060216.html.
Gurney ME, Pu H, Chiu AY, Dal Canto MC, Polchow CY, Alexander DD, Caliendo J, Hentati A et al (1994) Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation. Science 264(5166):1772–1775
Cardoso FL, Herz J, Fernandes A, Rocha J, Sepodes B, Brito MA, McGavern DB, Brites D (2015) Systemic inflammation in early neonatal mice induces transient and lasting neurodegenerative effects. J Neuroinflammation 12:82. doi:10.1186/s12974-015-0299-3
Simões AE, Pereira DM, Amaral JD, Nunes AF, Gomes SE, Rodrigues PM, Lo AC, D'Hooge R et al (2013) Efficient recovery of proteins from multiple source samples after TRIzol((R)) or TRIzol((R))LS RNA extraction and long-term storage. BMC Genomics 14:181. doi:10.1186/1471-2164-14-181
Fernandes A, Falcão AS, Silva RF, Gordo AC, Gama MJ, Brito MA, Brites D (2006) Inflammatory signalling pathways involved in astroglial activation by unconjugated bilirubin. J Neurochem 96(6):1667–1679. doi:10.1111/j.1471-4159.2006.03680.x
Caldeira C, Oliveira AF, Cunha C, Vaz AR, Falcão AS, Fernandes A, Brites D (2014) Microglia change from a reactive to an age-like phenotype with the time in culture. Front Cell Neurosci 8:152. doi:10.3389/fncel.2014.00152
Cunha C, Gomes C, Vaz AR, Brites D (2016) Exploring new inflammatory biomarkers and pathways during LPS-induced M1 polarization. Mediat Inflamm 2016:6986175. doi:10.1155/2016/6986175
Pathak S, Grillo AR, Scarpa M, Brun P, D'Inca R, Nai L, Banerjee A, Cavallo D et al (2015) MiR-155 modulates the inflammatory phenotype of intestinal myofibroblasts by targeting SOCS1 in ulcerative colitis. Exp Mol Med 47:e164. doi:10.1038/emm.2015.21
de Jong EK, Dijkstra IM, Hensens M, Brouwer N, van Amerongen M, Liem RS, Boddeke HW, Biber K (2005) Vesicle-mediated transport and release of CCL21 in endangered neurons: a possible explanation for microglia activation remote from a primary lesion. J Neurosci 25(33):7548–7557. doi:10.1523/JNEUROSCI.1019-05.2005
Díaz-Amarilla P, Olivera-Bravo S, Trias E, Cragnolini A, Martinez-Palma L, Cassina P, Beckman J, Barbeito L (2011) Phenotypically aberrant astrocytes that promote motoneuron damage in a model of inherited amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A 108(44):18126–18131. doi:10.1073/pnas.1110689108
Vargas MR, Johnson JA (2010) Astrogliosis in amyotrophic lateral sclerosis: role and therapeutic potential of astrocytes. Neurotherapeutics 7(4):471–481. doi:10.1016/j.nurt.2010.05.012
Awada R, Rondeau P, Gres S, Saulnier-Blache JS, Lefebvre d'Hellencourt C, Bourdon E (2012) Autotaxin protects microglial cells against oxidative stress. Free Radic Biol Med 52(2):516–526. doi:10.1016/j.freeradbiomed.2011.11.014
Chiu IM, Morimoto ET, Goodarzi H, Liao JT, O'Keeffe S, Phatnani HP, Muratet M, Carroll MC et al (2013) A neurodegeneration-specific gene-expression signature of acutely isolated microglia from an amyotrophic lateral sclerosis mouse model. Cell Rep 4(2):385–401. doi:10.1016/j.celrep.2013.06.018
Li X, Kong D, Chen H, Liu S, Hu H, Wu T, Wang J, Chen W, Ning Y, Li Y, Lu Z (2016) miR-155 acts as an anti-inflammatory factor in atherosclerosis-associated foam cell formation by repressing calcium-regulated heat stable protein 1. Scientific reports 6:21789. doi:10.1038/srep21789
Raymond A, Ensslin MA, Shur BD (2009) SED1/MFG-E8: a bi-motif protein that orchestrates diverse cellular interactions. J Cell Biochem 106(6):957–966. doi:10.1002/jcb.22076
Liu Y, Yang X, Guo C, Nie P, Liu Y, Ma J (2013) Essential role of MFG-E8 for phagocytic properties of microglial cells. PLoS One 8(2):e55754. doi:10.1371/journal.pone.0055754
Suzuki M, El-Hage N, Zou S, Hahn YK, Sorrell ME, Sturgill JL, Conrad DH, Knapp PE et al (2011) Fractalkine/CX3CL1 protects striatal neurons from synergistic morphine and HIV-1 Tat-induced dendritic losses and death. Mol Neurodegener 6:78. doi:10.1186/1750-1326-6-78
Sheridan GK, Murphy KJ (2013) Neuron-glia crosstalk in health and disease: fractalkine and CX3CR1 take centre stage. Open biology 3(12):130181. doi:10.1098/rsob.130181
Butovsky O, Siddiqui S, Gabriely G, Lanser AJ, Dake B, Murugaiyan G, Doykan CE, Wu PM et al (2012) Modulating inflammatory monocytes with a unique microRNA gene signature ameliorates murine ALS. J Clin Invest 122(9):3063–3087. doi:10.1172/JCI62636
Oak S, Mandrekar P, Catalano D, Kodys K, Szabo G (2006) TLR2- and TLR4-mediated signals determine attenuation or augmentation of inflammation by acute alcohol in monocytes. J Immunol 176(12):7628–7635
Volonté C, Apolloni S, Parisi C (2015) MicroRNAs: newcomers into the ALS picture. CNS Neurol Disord Drug Targets 14(2):194–207
Ponomarev ED, Veremeyko T, Weiner HL (2013) MicroRNAs are universal regulators of differentiation, activation, and polarization of microglia and macrophages in normal and diseased CNS. Glia 61(1):91–103. doi:10.1002/glia.22363
Saba R, Gushue S, Huzarewich RL, Manguiat K, Medina S, Robertson C, Booth SA (2012) MicroRNA 146a (miR-146a) is over-expressed during prion disease and modulates the innate immune response and the microglial activation state. PLoS One 7(2):e30832. doi:10.1371/journal.pone.0030832
Ponomarev ED, Veremeyko T, Barteneva N, Krichevsky AM, Weiner HL (2011) MicroRNA-124 promotes microglia quiescence and suppresses EAE by deactivating macrophages via the C/EBP-alpha-PU.1 pathway. Nat Med 17(1):64–70. doi:10.1038/nm.2266
Limatola C, Ransohoff RM (2014) Modulating neurotoxicity through CX3CL1/CX3CR1 signaling. Front Cell Neurosci 8:229. doi:10.3389/fncel.2014.00229
Zabel MK, Zhao L, Zhang Y, Gonzalez SR, Ma W, Wang X, Fariss RN, Wong WT (2016) Microglial phagocytosis and activation underlying photoreceptor degeneration is regulated by CX3CL1-CX3CR1 signaling in a mouse model of retinitis pigmentosa. Glia 64(9):1479–1491. doi:10.1002/glia.23016
Vu LT, Bowser R (2016) Fluid-based biomarkers for amyotrophic lateral sclerosis. Neurotherapeutics. doi:10.1007/s13311-016-0503-x
Cardoso AL, Guedes JR, Pereira de Almeida L, Pedroso de Lima MC (2012) miR-155 modulates microglia-mediated immune response by down-regulating SOCS-1 and promoting cytokine and nitric oxide production. Immunology 135(1):73–88. doi:10.1111/j.1365-2567.2011.03514.x
Guedes JR, Custodia CM, Silva RJ, de Almeida LP, Pedroso de Lima MC, Cardoso AL (2014) Early miR-155 upregulation contributes to neuroinflammation in Alzheimer’s disease triple transgenic mouse model. Hum Mol Genet 23(23):6286–6301. doi:10.1093/hmg/ddu348
Czochor JR, Sulkowski P, Glazer PM (2016) miR-155 overexpression promotes genomic instability by reducing high-fidelity polymerase delta expression and activating error-prone DSB repair. Mol Cancer Res 14(4):363–373. doi:10.1158/1541-7786.MCR-15-0399
Yuan K, Zhang X, Lv L, Zhang J, Liang W, Wang P (2016) Fine-tuning the expression of microRNA-155 controls acetaminophen-induced liver inflammation. Int Immunopharmacol 40:339–346. doi:10.1016/j.intimp.2016.09.011
Cardona AE, Pioro EP, Sasse ME, Kostenko V, Cardona SM, Dijkstra IM, Huang D, Kidd G et al (2006) Control of microglial neurotoxicity by the fractalkine receptor. Nat Neurosci 9(7):917–924. doi:10.1038/nn1715
Fricker M, Neher JJ, Zhao JW, Thery C, Tolkovsky AM, Brown GC (2012) MFG-E8 mediates primary phagocytosis of viable neurons during neuroinflammation. J Neurosci 32(8):2657–2666. doi:10.1523/JNEUROSCI.4837-11.2012
Hanayama R, Tanaka M, Miwa K, Shinohara A, Iwamatsu A, Nagata S (2002) Identification of a factor that links apoptotic cells to phagocytes. Nature 417(6885):182–187. doi:10.1038/417182a
Das A, Ghatak S, Sinha M, Chaffee S, Ahmed NS, Parinandi NL, Wohleb ES, Sheridan JF et al (2016) Correction of MFG-E8 resolves inflammation and promotes cutaneous wound healing in diabetes. J Immunol 196(12):5089–5100. doi:10.4049/jimmunol.1502270
Parisi C, Napoli G, Amadio S, Spalloni A, Apolloni S, Longone P, Volonte C (2016) MicroRNA-125b regulates microglia activation and motor neuron death in ALS. Cell Death Differ 23(3):531–541. doi:10.1038/cdd.2015.153
He X, Jing Z, Cheng G (2014) MicroRNAs: new regulators of Toll-like receptor signalling pathways. Biomed Res Int 2014:945169. doi:10.1155/2014/945169
Sun Y, Luo ZM, Guo XM, Su DF, Liu X (2015) An updated role of microRNA-124 in central nervous system disorders: a review. Front Cell Neurosci 9:193. doi:10.3389/fncel.2015.00193
Pinto S, Cunha C, Barbosa M, Vaz AR, Brites D (2017) Exosomes from NSC-34 cells transfected with hSOD1-G93A are enriched in miR-124 and drive alterations in microglia phenotype. Front Neurosci 11:273. doi:10.3389/fnins.2017.00273
Thomsen GM, Gowing G, Latter J, Chen M, Vit JP, Staggenborg K, Avalos P, Alkaslasi M et al (2014) Delayed disease onset and extended survival in the SOD1G93A rat model of amyotrophic lateral sclerosis after suppression of mutant SOD1 in the motor cortex. J Neurosci 34(47):15587–15600. doi:10.1523/JNEUROSCI.2037-14.2014
Kuo JJ, Schonewille M, Siddique T, Schults AN, Fu R, Bar PR, Anelli R, Heckman CJ et al (2004) Hyperexcitability of cultured spinal motoneurons from presymptomatic ALS mice. J Neurophysiol 91(1):571–575. doi:10.1152/jn.00665.2003
Vucic S, Nicholson GA, Kiernan MC (2008) Cortical hyperexcitability may precede the onset of familial amyotrophic lateral sclerosis. Brain 131(Pt 6):1540–1550. doi:10.1093/brain/awn071
Ciryam P, Kundra R, Freer R, Morimoto RI, Dobson CM, Vendruscolo M (2016) A transcriptional signature of Alzheimer’s disease is associated with a metastable subproteome at risk for aggregation. Proc Natl Acad Sci U S A 113(17):4753–4758. doi:10.1073/pnas.1516604113
Boillée S, Yamanaka K, Lobsiger CS, Copeland NG, Jenkins NA, Kassiotis G, Kollias G, Cleveland DW (2006) Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312(5778):1389–1392. doi:10.1126/science.1123511
Frakes AE, Ferraiuolo L, Haidet-Phillips AM, Schmelzer L, Braun L, Miranda CJ, Ladner KJ, Bevan AK et al (2014) Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron 81(5):1009–1023. doi:10.1016/j.neuron.2014.01.013
Nikodemova M, Small AL, Smith SMC, Mitchell GS, Watters JJ (2014) Spinal but not cortical microglia acquire an atypical phenotype with high VEGF, galectin-3 and osteopontin, and blunted inflammatory responses in ALS rats. Neurobiol Dis 69:43–53. doi:10.1016/j.nbd.2013.11.009
Montero TD, Orellana JA (2015) Hemichannels: new pathways for gliotransmitter release. Neuroscience 286:45–59. doi:10.1016/j.neuroscience.2014.11.048
Orellana JA, Froger N, Ezan P, Jiang JX, Bennett MV, Naus CC, Giaume C, Saez JC (2011) ATP and glutamate released via astroglial connexin 43 hemichannels mediate neuronal death through activation of pannexin 1 hemichannels. J Neurochem 118(5):826–840. doi:10.1111/j.1471-4159.2011.07210.x
Chen SC, Leach MW, Chen Y, Cai XY, Sullivan L, Wiekowski M, Dovey-Hartman BJ, Zlotnik A et al (2002) Central nervous system inflammation and neurological disease in transgenic mice expressing the CC chemokine CCL21 in oligodendrocytes. J Immunol 168(3):1009–1017
Wolf Y, Yona S, Kim KW, Jung S (2013) Microglia, seen from the CX3CR1 angle. Front Cell Neurosci 7:26. doi:10.3389/fncel.2013.00026
Wu J, Bie B, Yang H, Xu JJ, Brown DL, Naguib M (2013) Suppression of central chemokine fractalkine receptor signaling alleviates amyloid-induced memory deficiency. Neurobiol Aging 34(12):2843–2852. doi:10.1016/j.neurobiolaging.2013.06.003
Lopez-Lopez A, Gamez J, Syriani E, Morales M, Salvado M, Rodriguez MJ, Mahy N, Vidal-Taboada JM (2014) CX3CR1 is a modifying gene of survival and progression in amyotrophic lateral sclerosis. PLoS One 9(5):e96528. doi:10.1371/journal.pone.0096528
Chapman GA, Moores K, Harrison D, Campbell CA, Stewart BR, Strijbos PJ (2000) Fractalkine cleavage from neuronal membranes represents an acute event in the inflammatory response to excitotoxic brain damage. J Neurosci 20(15):RC87
Zahednasab H, Balood M, Harirchian MH, Mesbah-Namin SA, Rahimian N, Siroos B (2014) Increased autotaxin activity in multiple sclerosis. J Neuroimmunol 273(1–2):120–123. doi:10.1016/j.jneuroim.2014.06.006
Acknowledgements
This work was funded by the Research Grant of the Santa Casa Scientific Research Program on ALS, by Santa Casa da Misericórdia de Lisboa (SCML), Portugal, Project Ref. ELA-2015-002 (to DB), by the project PTDC/SAU-FAR/118787/2010 (to D.B.) and, in part, by iMed.ULisboa (UID/DTP/04138/2013) from Fundação para a Ciência e a Tecnologia (FCT). A.R.V. holds a postdoctoral research fellowship (SFRH/BPD/76590/2011), and C.C. and C.G. are recipients of PhD fellowships (SFRH/BD/91316/2012 and SFRH/BD/102718/2014, respectively), all from FCT. The funding organization had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We thank to Dr. Rui Gomes (Instituto de Medicina Molecular, Faculdade de Medicina de Lisboa, Universidade de Lisboa, Portugal) for genotyping mice.
Author information
Authors and Affiliations
Contributions
D.B, conceived the study; D.B. and A.R.V. designed the experiments; A.R.V. gave technical support; C.C., C.G., A.R.V., A.F. and C.S. acquired data; A.S. and A.M.C. contributed to conceptual advice and data acquisition on mice related with its usage, management, and maintenance; A.R.V., C.C., and D.B. analyzed data; C.C. and A.R.V. drafted figures and participated in the manuscript writing; D.B. wrote and edited the final version. The manuscript has been read and approved by all named authors.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Supplementary Table 1
(DOC 53 kb)
Supplementary Table 2
(DOC 66 kb)
Rights and permissions
About this article
Cite this article
Cunha, C., Santos, C., Gomes, C. et al. Downregulated Glia Interplay and Increased miRNA-155 as Promising Markers to Track ALS at an Early Stage. Mol Neurobiol 55, 4207–4224 (2018). https://doi.org/10.1007/s12035-017-0631-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0631-2