Abstract
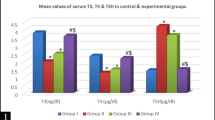
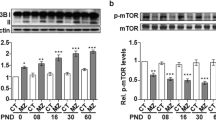
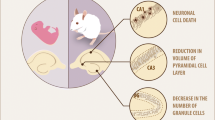
Hypothyroidism is a condition that becomes more prevalent with age. Patients with untreated hypothyroidism have consistently reported symptoms of severe cognitive impairments. In patients suffering hypothyroidism, thyroid hormone supplementation offers the prospect to alleviate the cognitive consequences of hypothyroidism; however, the therapeutic value of TH supplementation remains at present uncertain and the link between cellular modifications associated with hypothyroidism and neurodegeneration remains to be elucidated. In the present study, we therefore evaluated the molecular and behavioral consequences of T3 hormone replacement in an animal model of hypothyroidism. We have previously reported that the antithyroid molecule propylthiouracil (PTU) given in the drinking water favors cerebral atrophy, brain neuroinflammation, Aβ production, Tau hyperphosphorylation, and altered plasticity-related cell-signaling pathways in the hippocampus in association with hippocampal-dependent spatial memory deficits. In the present study, our aim was to explore, in this model, the effect of hippocampal T3 signaling normalization on various molecular mechanisms involved in learning and memory that goes awry under conditions of hypothyroidism and to evaluate its potential for recovery of hippocampal-dependent memory deficits. We report that T3 supplementation can alleviate hippocampal-dependent memory impairments displayed by hypothyroid rats and normalize key markers of thyroid status in the hippocampus, of neuroinflammation, Aβ production, and of cell-signaling pathways known to be involved in synaptic plasticity and memory function. Together, these findings suggest that normalization of hippocampal T3 signaling is sufficient to reverse molecular and cognitive dysfunctions associated with hypothyroidism.






Similar content being viewed by others
References
Morte B, Bernal J (2014) Thyroid hormone action: astrocyte-neuron communication. Front Endocrinol 5:82. https://doi.org/10.3389/fendo.2014.00082
Raymaekers SR, Darras VM (2017) Thyroid hormones and learning-associated neuroplasticity. Gen Comp Endocrinol 247:26–33. https://doi.org/10.1016/j.ygcen.2017.04.001
Schroeder AC, Privalsky ML (2014) Thyroid hormones, t3 and t4, in the brain. Front Endocrinol 5:40. https://doi.org/10.3389/fendo.2014.00040
Ritchie M, Yeap BB (2015) Thyroid hormone: influences on mood and cognition in adults. Maturitas 81(2):266–275. https://doi.org/10.1016/j.maturitas.2015.03.016
Aggarwal N, Razvi S (2013) Thyroid and aging or the aging thyroid? An evidence-based analysis of the literature. J Thyroid Res 2013:481287–481288. https://doi.org/10.1155/2013/481287
Tan ZS, Vasan RS (2009) Thyroid function and Alzheimer’s disease. J Alzheimers Dis 16(3):503–507
Ganguli M, Burmeister LA, Seaberg EC, Belle S, DeKosky ST (1996) Association between dementia and elevated TSH: a community-based study. Biol Psychiatry 40(8):714–725. https://doi.org/10.1016/0006-3223(95)00489-0
van Osch LA, Hogervorst E, Combrinck M, Smith AD (2004) Low thyroid-stimulating hormone as an independent risk factor for Alzheimer disease. Neurology 62(11):1967–1971
Alzoubi KH, Alkadhi KA (2014) Levothyroxin replacement therapy restores hypothyroidism induced impairment of L-LTP induction: critical role of CREB. Brain Res Bull 100:29–37. https://doi.org/10.1016/j.brainresbull.2013.10.011
McAninch EA, Bianco AC (2016) The history and future of treatment of hypothyroidism. Ann Intern Med 164(1):50–56. https://doi.org/10.7326/M15-1799
Freitas BC, Gereben B, Castillo M, Kallo I, Zeold A, Egri P, Liposits Z, Zavacki AM et al (2010) Paracrine signaling by glial cell-derived triiodothyronine activates neuronal gene expression in the rodent brain and human cells. J Clin Invest 120(6):2206–2217. https://doi.org/10.1172/JCI41977
Cooke GE, Mullally S, Correia N, O'Mara SM, Gibney J (2014) Hippocampal volume is decreased in adults with hypothyroidism. Thyroid 24(3):433–440. https://doi.org/10.1089/thy.2013.0058
Gerges NZ, Alzoubi KH, Park CR, Diamond DM, Alkadhi KA (2004) Adverse effect of the combination of hypothyroidism and chronic psychosocial stress on hippocampus-dependent memory in rats. Behav Brain Res 155(1):77–84. https://doi.org/10.1016/j.bbr.2004.04.003
Artis AS, Bitiktas S, Taskin E, Dolu N, Liman N, Suer C (2012) Experimental hypothyroidism delays field excitatory post-synaptic potentials and disrupts hippocampal long-term potentiation in the dentate gyrus of hippocampal formation and Y-maze performance in adult rats. J Neuroendocrinol 24(3):422–433. https://doi.org/10.1111/j.1365-2826.2011.02253.x
Alzoubi KH, Gerges NZ, Alkadhi KA (2005) Levothyroxin restores hypothyroidism-induced impairment of LTP of hippocampal CA1: electrophysiological and molecular studies. Exp Neurol 195(2):330–341. https://doi.org/10.1016/j.expneurol.2005.05.007
Iniguez MA, Rodriguez-Pena A, Ibarrola N, Aguilera M, Munoz A, Bernal J (1993) Thyroid hormone regulation of RC3, a brain-specific gene encoding a protein kinase-C substrate. Endocrinology 133(2):467–473. https://doi.org/10.1210/endo.133.2.8344193
Husson M, Enderlin V, Alfos S, Boucheron C, Pallet V, Higueret P (2004) Expression of neurogranin and neuromodulin is affected in the striatum of vitamin A-deprived rats. Brain Res Mol Brain Res 123(1-2):7–17. https://doi.org/10.1016/j.molbrainres.2003.12.012
Guadano-Ferraz A, Escamez MJ, Morte B, Vargiu P, Bernal J (1997) Transcriptional induction of RC3/neurogranin by thyroid hormone: differential neuronal sensitivity is not correlated with thyroid hormone receptor distribution in the brain. Brain Res Mol Brain Res 49(1–2):37–44
Piosik PA, van Groenigen M, Baas F (1996) Effect of thyroid hormone deficiency on RC3/neurogranin mRNA expression in the prenatal and adult caprine brain. Brain Res Mol Brain Res 42(2):227–235
Marambaud P, Dreses-Werringloer U, Vingtdeux V (2009) Calcium signaling in neurodegeneration. Mol Neurodegener 4:20. https://doi.org/10.1186/1750-1326-4-20
Koromilas C, Liapi C, Schulpis KH, Kalafatakis K, Zarros A, Tsakiris S (2010) Structural and functional alterations in the hippocampus due to hypothyroidism. Metab Brain Dis 25(3):339–354. https://doi.org/10.1007/s11011-010-9208-8
Bozon B, Davis S, Laroche S (2002) Regulated transcription of the immediate-early gene Zif268: mechanisms and gene dosage-dependent function in synaptic plasticity and memory formation. Hippocampus 12(5):570–577. https://doi.org/10.1002/hipo.10100
Davis S, Bozon B, Laroche S (2003) How necessary is the activation of the immediate early gene zif268 in synaptic plasticity and learning? Behav Brain Res 142(1–2):17–30
Besnard A, Laroche S, Caboche J (2014) Comparative dynamics of MAPK/ERK signalling components and immediate early genes in the hippocampus and amygdala following contextual fear conditioning and retrieval. Brain Struct Funct 219(1):415–430. https://doi.org/10.1007/s00429-013-0505-y
Bianco AC, Salvatore D, Gereben B, Berry MJ, Larsen PR (2002) Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocr Rev 23(1):38–89
Cooper DS (2005) Antithyroid drugs. N Engl J Med 352(9):905–917
Ghenimi N, Alfos S, Redonnet A, Higueret P, Pallet V, Enderlin V (2010) Adult-onset hypothyroidism induces the amyloidogenic pathway of amyloid precursor protein processing in the rat hippocampus. J Neuroendocrinol 22(8):951–959. https://doi.org/10.1111/j.1365-2826.2010.02002.x
Chaalal A, Poirier R, Blum D, Gillet B, Le Blanc P, Basquin M, Buee L, Laroche S et al (2014) PTU-induced hypothyroidism in rats leads to several early neuropathological signs of Alzheimer’s disease in the hippocampus and spatial memory impairments. Hippocampus 24(11):1381–1393. https://doi.org/10.1002/hipo.22319
Sui L, Wang F, Li BM (2006) Adult-onset hypothyroidism impairs paired-pulse facilitation and long-term potentiation of the rat dorsal hippocampo-medial prefrontal cortex pathway in vivo. Brain Res 1096(1):53–60
Weiss RE, Forrest D, Pohlenz J, Cua K, Curran T, Refetoff S (1997) Thyrotropin regulation by thyroid hormone in thyroid hormone receptor beta-deficient mice. Endocrinology 138(9):3624–3629. https://doi.org/10.1210/endo.138.9.5412
Weiss RE, Murata Y, Cua K, Hayashi Y, Seo H, Refetoff S (1998) Thyroid hormone action on liver, heart, and energy expenditure in thyroid hormone receptor beta-deficient mice. Endocrinology 139(12):4945–4952. https://doi.org/10.1210/endo.139.12.6412
Feart C, Pallet V, Boucheron C, Higueret D, Alfos S, Letenneur L, Dartigues JF, Higueret P (2005) Aging affects the retinoic acid and the triiodothyronine nuclear receptor mRNA expression in human peripheral blood mononuclear cells. Eur J Endocrinol 152(3):449–458
Vallortigara J, Alfos S, Micheau J, Higueret P, Enderlin V (2008) T3 administration in adult hypothyroid mice modulates expression of proteins involved in striatal synaptic plasticity and improves motor behavior. Neurobiol Dis 31(3):378–385
Sapin R, Schlienger JL (2003) Thyroxine (T4) and tri-iodothyronine (T3) determinations: techniques and value in the assessment of thyroid function. Ann Biol Clin (Paris) 61(4):411–420
Diamond DM, Park CR, Heman KL, Rose GM (1999) Exposing rats to a predator impairs spatial working memory in the radial arm water maze. Hippocampus 9(5):542–552. https://doi.org/10.1002/(SICI)1098-1063(1999)9:5<542::AID-HIPO8>3.0.CO;2-N
Lin HB, Yang XM, Li TJ, Cheng YF, Zhang HT, Xu JP (2009) Memory deficits and neurochemical changes induced by C-reactive protein in rats: implication in Alzheimer’s disease. Psychopharmacology 204(4):705–714. https://doi.org/10.1007/s00213-009-1499-2
Kelly A, Laroche S, Davis S (2003) Activation of mitogen-activated protein kinase/extracellular signal-regulated kinase in hippocampal circuitry is required for consolidation and reconsolidation of recognition memory. J Neurosci 23(12):5354–5360
Iniguez MA, Rodriguez-Pena A, Ibarrola N, Morreale de Escobar G, Bernal J (1992) Adult rat brain is sensitive to thyroid hormone. Regulation of RC3/neurogranin mRNA. J Clin Invest 90(2):554–558. https://doi.org/10.1172/JCI115894
Ge JF, Peng L, Hu CM, Wu TN (2012) Impaired learning and memory performance in a subclinical hypothyroidism rat model induced by hemi-thyroid electrocauterisation. J Neuroendocrinol 24(6):953–961. https://doi.org/10.1111/j.1365-2826.2012.02297.x
Rivas M, Naranjo JR (2007) Thyroid hormones, learning and memory. Genes Brain Behav 6(Suppl 1):40–44. https://doi.org/10.1111/j.1601-183X.2007.00321.x
Yu D, Zhou H, Yang Y, Jiang Y, Wang T, Lv L, Zhou Q, Yang Y et al (2015) The bidirectional effects of hypothyroidism and hyperthyroidism on anxiety- and depression-like behaviors in rats. Horm Behav 69:106–115. https://doi.org/10.1016/j.yhbeh.2015.01.003
Gustafson C (2014) Antonio C. Bianco, md, phd: is T4 enough for patients with hypothyroid dysfunction? Integr Med (Encinitas) 13(3):20–22
Bocco BM, Werneck-de-Castro JP, Oliveira KC, Fernandes GW, Fonseca TL, Nascimento BP, McAninch EA, Ricci E et al (2016) Type 2 deiodinase disruption in astrocytes results in anxiety-depressive-like behavior in male mice. Endocrinology 157(9):3682–3695. https://doi.org/10.1210/en.2016-1272
Boelen A, Kwakkel J, Alkemade A, Renckens R, Kaptein E, Kuiper G, Wiersinga WM, Visser TJ (2005) Induction of type 3 deiodinase activity in inflammatory cells of mice with chronic local inflammation. Endocrinology 146(12):5128–5134
Park CR, Zoladz PR, Conrad CD, Fleshner M, Diamond DM (2008) Acute predator stress impairs the consolidation and retrieval of hippocampus-dependent memory in male and female rats. Learn Mem 15(4):271–280. https://doi.org/10.1101/lm.721108
Alzoubi KH, Gerges NZ, Aleisa AM, Alkadhi KA (2009) Levothyroxin restores hypothyroidism-induced impairment of hippocampus-dependent learning and memory: behavioral, electrophysiological, and molecular studies. Hippocampus 19(1):66–78. https://doi.org/10.1002/hipo.20476
Bourtchuladze R, Frenguelli B, Blendy J, Cioffi D, Schutz G, Silva AJ (1994) Deficient long-term memory in mice with a targeted mutation of the cAMP-responsive element-binding protein. Cell 79(1):59–68
Sweatt JD (2001) The neuronal MAP kinase cascade: a biochemical signal integration system subserving synaptic plasticity and memory. J Neurochem 76(1):1–10
Davis S, Vanhoutte P, Pages C, Caboche J, Laroche S (2000) The MAPK/ERK cascade targets both Elk-1 and cAMP response element-binding protein to control long-term potentiation-dependent gene expression in the dentate gyrus in vivo. J Neurosci 20(12):4563–4572
Lisman J, Schulman H, Cline H (2002) The molecular basis of CaMKII function in synaptic and behavioural memory. Nat Rev Neurosci 3(3):175–190. https://doi.org/10.1038/nrn753
Jones MW, Errington ML, French PJ, Fine A, Bliss TV, Garel S, Charnay P, Bozon B et al (2001) A requirement for the immediate early gene Zif268 in the expression of late LTP and long-term memories. Nat Neurosci 4(3):289–296. https://doi.org/10.1038/85138
Veyrac A, Besnard A, Caboche J, Davis S, Laroche S (2014) The transcription factor Zif268/Egr1, brain plasticity, and memory. Prog Mol Biol Transl Sci 122:89–129. https://doi.org/10.1016/B978-0-12-420170-5.00004-0
Thomas GM, Huganir RL (2004) MAPK cascade signalling and synaptic plasticity. Nat Rev Neurosci 5(3):173–183. https://doi.org/10.1038/nrn1346
Bitiktas S, Kandemir B, Tan B, Kavraal S, Liman N, Dursun N, Donmez-Altuntas H, Aksan-Kurnaz I et al (2016) Adult-onset hyperthyroidism impairs spatial learning: possible involvement of mitogen-activated protein kinase signaling pathways. Neuroreport 27(11):802–808. https://doi.org/10.1097/WNR.0000000000000612
Mancini A, Di Segni C, Raimondo S, Olivieri G, Silvestrini A, Meucci E, Curro D (2016) Thyroid hormones, oxidative stress, and inflammation. Mediat Inflamm 2016:6757154. https://doi.org/10.1155/2016/6757154 12
Noda M (2015) Possible role of glial cells in the relationship between thyroid dysfunction and mental disorders. Front Cell Neurosci 9:194. https://doi.org/10.3389/fncel.2015.00194
Cortes C, Eugenin E, Aliaga E, Carreno LJ, Bueno SM, Gonzalez PA, Gayol S, Naranjo D et al (2012) Hypothyroidism in the adult rat causes incremental changes in brain-derived neurotrophic factor, neuronal and astrocyte apoptosis, gliosis, and deterioration of postsynaptic density. Thyroid 22(9):951–963. https://doi.org/10.1089/thy.2010.0400
Yirmiya R, Goshen I (2011) Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav Immun 25(2):181–213. https://doi.org/10.1016/j.bbi.2010.10.015
Delpech JC, Madore C, Nadjar A, Joffre C, Wohleb ES, Laye S (2015) Microglia in neuronal plasticity: influence of stress. Neuropharmacology 96(Pt A):19–28. https://doi.org/10.1016/j.neuropharm.2014.12.034
Barrientos RM, Higgins EA, Sprunger DB, Watkins LR, Rudy JW, Maier SF (2002) Memory for context is impaired by a post context exposure injection of interleukin-1 beta into dorsal hippocampus. Behav Brain Res 134(1–2):291–298
Labrousse VF, Costes L, Aubert A, Darnaudery M, Ferreira G, Amedee T, Laye S (2009) Impaired interleukin-1beta and c-Fos expression in the hippocampus is associated with a spatial memory deficit in P2X(7) receptor-deficient mice. PLoS One 4(6):e6006. https://doi.org/10.1371/journal.pone.0006006
Sparkman NL, Buchanan JB, Heyen JR, Chen J, Beverly JL, Johnson RW (2006) Interleukin-6 facilitates lipopolysaccharide-induced disruption in working memory and expression of other proinflammatory cytokines in hippocampal neuronal cell layers. J Neurosci 26(42):10709–10716. https://doi.org/10.1523/JNEUROSCI.3376-06.2006
O'Barr SA, Oh JS, Ma C, Brent GA, Schultz JJ (2006) Thyroid hormone regulates endogenous amyloid-beta precursor protein gene expression and processing in both in vitro and in vivo models. Thyroid 16(12):1207–1213
Jacobsen JS, Wu CC, Redwine JM, Comery TA, Arias R, Bowlby M, Martone R, Morrison JH et al (2006) Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci U S A 103(13):5161–5166. https://doi.org/10.1073/pnas.0600948103
Brouillette J, Caillierez R, Zommer N, Alves-Pires C, Benilova I, Blum D, De Strooper B, Buee L (2012) Neurotoxicity and memory deficits induced by soluble low-molecular-weight amyloid-beta1–42 oligomers are revealed in vivo by using a novel animal model. J Neurosci 32(23):7852–7861. https://doi.org/10.1523/JNEUROSCI.5901-11.2012
Cai Z, Liu N, Wang C, Qin B, Zhou Y, Xiao M, Chang L, Yan LJ et al (2016) Role of RAGE in Alzheimer’s disease. Cell Mol Neurobiol 36(4):483–495. https://doi.org/10.1007/s10571-015-0233-3
Bjerke SN, Bjoro T, Heyerdahl S (2001) Psychiatric and cognitive aspects of hypothyroidism. Tidsskr Nor Laegeforen 121(20):2373–2376
Samuels MH, Schuff KG, Carlson NE, Carello P, Janowsky JS (2007) Health status, psychological symptoms, mood, and cognition in L-thyroxine-treated hypothyroid subjects. Thyroid 17(3):249–258. https://doi.org/10.1089/thy.2006.0252
Correia N, Mullally S, Cooke G, Tun TK, Phelan N, Feeney J, Fitzgibbon M, Boran G et al (2009) Evidence for a specific defect in hippocampal memory in overt and subclinical hypothyroidism. J Clin Endocrinol Metab 94(10):3789–3797. https://doi.org/10.1210/jc.2008-2702
Escobar-Morreale HF, Botella-Carretero JI, Escobar del Rey F, Morreale de Escobar G (2005) REVIEW: treatment of hypothyroidism with combinations of levothyroxine plus liothyronine. J Clin Endocrinol Metab 90(8):4946–4954. https://doi.org/10.1210/jc.2005-0184
Wiersinga WM (2017) Therapy of endocrine disease: T4+T3 combination therapy: is there a true effect? Eur J Endocrinol 177:R287–R296. https://doi.org/10.1530/EJE-17-0645
Escobar-Morreale HF, Obregon MJ, Escobar del Rey F, Morreale de Escobar G (1999) Tissue-specific patterns of changes in 3,5,3′-triiodo-L-thyronine concentrations in thyroidectomized rats infused with increasing doses of the hormone. Which are the regulatory mechanisms? Biochimie 81(5):453–462
Escobar-Morreale HF, del Rey FE, Obregon MJ, de Escobar GM (1996) Only the combined treatment with thyroxine and triiodothyronine ensures euthyroidism in all tissues of the thyroidectomized rat. Endocrinology 137(6):2490–2502. https://doi.org/10.1210/endo.137.6.8641203
Davis JD, Podolanczuk A, Donahue JE, Stopa E, Hennessey JV, Luo LG, Lim YP, Stern RA (2008) Thyroid hormone levels in the prefrontal cortex of post-mortem brains of Alzheimer’s disease patients. Curr Aging Sci 1(3):175–181
Johansson P, Almqvist EG, Johansson JO, Mattsson N, Hansson O, Wallin A, Blennow K, Zetterberg H et al (2013) Reduced cerebrospinal fluid level of thyroxine in patients with Alzheimer’s disease. Psychoneuroendocrinology 38(7):1058–1066. https://doi.org/10.1016/j.psyneuen.2012.10.012
Luo L, Stopa EG (2004) Thyrotropin releasing hormone inhibits tau phosphorylation by dual signaling pathways in hippocampal neurons. J Alzheimers Dis 6(5):527–536
Sampaolo S, Campos-Barros A, Mazziotti G, Carlomagno S, Sannino V, Amato G, Carella C, Di Iorio G (2005) Increased cerebrospinal fluid levels of 3,3′,5′-triiodothyronine in patients with Alzheimer’s disease. J Clin Endocrinol Metab 90(1):198–202
Acknowledgements
We would like to thank Pascale Gisquet-Verrier for her help with behavioral equipment and Fabien Lhericel for animal care.
Funding
This work was supported by Centre National de la Recherche Scientifique (France), University Paris-Sud (France) and by the France Alzheimer/Fédération pour la Recherche sur le Cerveau (AAP France Alzheimer 2013) to V Enderlin. A Chaalal was supported by a doctoral fellowship from the French Ministry of Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chaalal, A., Poirier, R., Blum, D. et al. Thyroid Hormone Supplementation Restores Spatial Memory, Hippocampal Markers of Neuroinflammation, Plasticity-Related Signaling Molecules, and β-Amyloid Peptide Load in Hypothyroid Rats. Mol Neurobiol 56, 722–735 (2019). https://doi.org/10.1007/s12035-018-1111-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1111-z