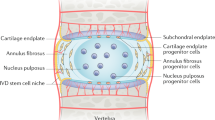
Abstract
Low back pain due to degeneration of intervertebral disc (IVD) is a major health problem resulting in significant disability as well as adding to the economic burden. Discectomy is a very common procedure done worldwide to relieve this pain. At present all the surgically removed disc tissue is mostly discarded. However, there are reports that state that progenitor cells in the IVD can be grown ex vivo and have the potential to be used for IVD repair and regeneration. We report here that viable cells can be harvested from surgically removed, herniated disc tissue and can be potentially used in cell based therapy. Further, we have successfully replaced xenogenic supplements such as foetal bovine serum with either autologous serum or human platelet lysate for culturing IVD cells from patient’s surgically removed disc tissue, without loss of any cell characteristics, including cell surface markers, growth factor secretion in the conditioned medium and osteogenic and chondrogenic differentiation potential in vitro. The present work will not only contribute to overcoming some of the major barriers in carrying out human clinical trials, but also provide a cheap, alternate source of proteins and growth factors for growing IVD cells ex vivo for therapy.



Similar content being viewed by others
References
Altaie A, Owston H and Jones E 2016 Use of platelet lysate for bone regeneration are we ready for clinical translation? World J. Stem Cells 8 47–55
Atashi F, Jaconi ME, Pittet-Cuénod B and Modarressi A 2015 Autologous platelet-rich plasma: a biological supplement to enhance adipose-derived mesenchymal stem cell expansion. Tissue Eng. Methods 21 253–262
Bari E, Perteghella S, Marrubini G, et al. 2018 In vitro efficacy of silk sericin microparticles and platelet lysate for intervertebral disk regeneration. Int. J. Biol. Macromol. 118 792–799
Becherucci V, Piccini L, Casamassima S, et al. 2018 Human platelet lysate in mesenchymal stromal cell expansion according to a GMP grade protocol: a cell factory experience. Stem Cell Res. Ther. 9 124
Bertolo A, Häfner S, Taddei AR, Baur M, Pötzel T, Steffen F and Stoyanov J 2015 Injectable microcarriers as human mesenchymal stem cell support and their application for cartilage and degenerated intervertebral disc repair. Eur. Cells Mate. 29 70–81
Bieback K, Fernandez-Munoz B, Pati S and Scahefer R 2019 Gaps in the knowledge of human platelet lysate as a cell culture supplement for cell therapy: a joint publication from the AABB and the International Society for Cell & Gene Therapy. Transfusion 59 3448–3460
Blanco JF, Graciani IF, Sanchez-Guijo FM, et al. 2010 Isolation and characterization of mesenchymal stromal cells from human degenerated nucleus pulposus comparison with bone marrow mesenchymal stromal cells from the same subjects. Spine 35 2259–2265
Centeno C, Markle J, Dodson E, Stemper I, Williams CJ, Hyzy M, Ichim T and Freeman M 2017 Treatment of lumbar degenerative disc disease-associated radicular pain with culture-expanded autologous mesenchymal stem cells: a pilot study on safety and efficacy. J. Transl. Med. 15 197
Choi J, Chung J-H, Kwon G-Y, Kim K-W, Kim S and Chang K 2013 Effectiveness of autologous serum as an alternative to fetal bovine serum in adipose-derived stem cell engineering. Cell Tissue Bank 14 413–422
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini Fc, Krause Ds, Deans Rj, Keating A, Prockop DJ and Horwitz EM 2006 Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8 315–317
Ganey T, Libera J, Moos V, Alasevic O, Fritsch KG, Meisel HJ and Hutton WC 2003 Disc chondrocyte transplantation in a canine model: a treatment for degenerated or damaged intervertebral disc. Spine 28 2609
Gruber HE, Johnson TL, Leslie K, et al. 2002 Autologous intervertebral disc cell implantation: a model using Psammomys obesus, the sand rat. Spine 27 1626
Kandoi S, Kumar PL, Patra B, Vidyasekar P, Sivanesan D, Vijayalakshmi S, Rajagopal K and Verma RS 2018 Evaluation of platelet lysate as a substitute for FBS in explant and enzymatic isolation methods of human umbilical cord MSCs. Sci. Rep. 8 12439
Leung VYL, Chan D and Cheung KMC 2006 Regeneration of intervertebral disc by mesenchymal stem cells: potentials, limitations, and future direction. Eur. Spine J. 15 S406–S413
Mochida J, Sakai D, Nakamura Y, Watanabe T, Yamamoto Y and Kato S 2015 Intervertebral Disc repair with activated nucleus pulposus cell transplantation: A three-year prospective clinical study of its safety. Eur. Cells Mater. 29 202–212
Risbud MV, Guttapalli A, Tsai T-T, et al. 2007 Evidence for skeletal progenitor cells in the degenerate human intervertebral disc. Spine 32 2537–2544
Sakai D and Andersson GB 2015 Stem cell therapy for intervertebral disc regeneration: obstacles and solutions. Nat. Rev. Rheumatol. 11 243–256
Sakai D, Mochida J and Iwahina T 2003 Transplantation of mesenchymal stem cells embedded in Atelocollagen gel to the intervertebral disc: a potential therapeutic model for disc degeneration. Biomaterials 24 3531–3541
Sakai D, Mochida J, Iwashina T, Watanabe T, Nakai T, Ando K and Hotta T 2005 Differentiation of mesenchymal stem cells transplanted to a rabbit degenerative disc model: potential and limitations for stem cell therapy in disc regeneration. Spine 30 2379–2387
Sakai D and Schol J 2017 Cell therapy for intervertebral disc repair: Clinical perspective. J. Orthopaedic Transl. 9 8–18
Shu CC, Dart A, Bell R, Dart C, Clarke E, Smith MM, Little CB and Melrose J 2018 Efficacy of administered mesenchymal stem cells in the initiation and co-ordination of repair processes by resident disc cells in an ovine (Ovis aries) large destabilizing lesion model of experimental disc degeneration. JOR Spine 1 e1037
Strandberg G, Sellberg F, Sommar P, Ronaghi M, Lubenow N, Knutson F and Berglund D 2017 Standardizing the freeze-thaw preparation of growth factors from platelet lysate. Transfusion 57 1058–1065
Trombi L, Danti S, Savelli S, Moscato S, D’Alessandro D, Ricci C, Giannotti S and Petrini M 2016 Mesenchymal stromal cell culture and delivery in autologous conditions: a smart approach for orthopedic applications. J. Vis. Exp. 118 e54845
Tschugg A, Michnacs F, Strowitzki M, Meisel HJ and Thomé C 2016 A prospective multicenter phase I/II clinical trial to evaluate safety and efficacy of NOVOCART disc plus autologous disc chondrocyte transplantation in the treatment of nucleotomized and degenerative lumbar disc to avoid secondary disease: study protocol for a randomized controlled trial. Trials 17 108
van der Valk J, Bieback K, Buta C, et al. 2018 Fetal bovine serum (FBS): past – present – future. Altex 35 99–118
Vos T, Flaxman AD, Naghavi M, et al. 2012 Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380 2163–2196
Acknowledgements
Biotechnology Industry Research Assistance Council, Delhi, is acknowledged for funding SN under the Biotechnology Ignition Grant (BIRAC/Venture0261/BIG-11/17). Help received from staff of BKL Walawalkar Rural Hospital, Sawarde, India, as well as from Ms. Sonali Mhatre and Ms. Seema Rajwade, in setting up the tissue culture lab and doing the initial experiments, is acknowledged. Assistance received from Dr. N. Vyas, Institute of Applied Biological Research and Development, Pune, and Dr. M. Wani, NCCS, Pune, in carrying out flow cytometry and differentiation assays is acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Corresponding editor: BJ Rao
Rights and permissions
About this article
Cite this article
Mujawar, S., Iyengar, K., Nadkarni, S. et al. Expansion and characterization of cells from surgically removed intervertebral disc fragments in xenogen-free medium. J Biosci 45, 108 (2020). https://doi.org/10.1007/s12038-020-00091-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12038-020-00091-w