Abstract
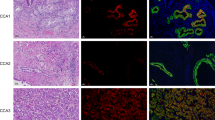
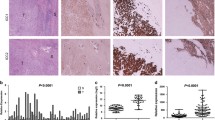
Notch signaling has been reported to correlate with tumor progression and metastasis in several types of cancer. In cholangiocarcinoma (CCA), it has recently been shown that NOTCH1 is overexpressed in both nucleus and cytoplasm of CCA cells; however, the complete understanding of Notch signaling in CCA is still lacking. Here, we aimed to understand the functions of NOTCH1 in CCA cells and the molecular mechanisms that underlie those functions. We used retroviral vectors to overexpress active forms of NOTCH1, the NOTCH1 intracellular domain (N1ICD) and N1ICD that lacks the RBP-J-associated module (RAM), in human CCA cell lines RMCCA-1 and HuCCA-1. Our results showed that activation of Notch signaling by both N1ICD variants enhanced CCA cell proliferation and survival via upregulation of pro-survival protein Mcl-1 and Bcl-xL. Moreover, our LC-MS/MS proteomic studies demonstrated that NOTCH1 may cooperate with 14-3-3 theta to promote CCA cell survival. Knockdown of 14-3-3 theta in RMCCA-1 cells overexpressing N1ICD, diminished pro-survival effects of N1ICD under gemcitabine treatment. In conclusion, these data demonstrated that NOTCH1 plays a role in CCA cell proliferation and survival via the regulation of 14-3-3 theta in a RAM-independent fashion.




Similar content being viewed by others
References
Aster JC, Xu L, Karnell FG, Patriub V, Pui JC, Pear WS (2000) Essential roles for ankyrin repeat and transactivation domains in induction of T-cell leukemia by notch1. Mol Cell Biol 20:7505–7515
Aster JC, Blacklow SC, Pear WS (2011) Notch signalling in T-cell lymphoblastic leukaemia/lymphoma and other haematological malignancies. J Pathol 223:262–273. https://doi.org/10.1002/path.2789
Dar A, Wu D, Lee N, Shibata E, Dutta A (2014) 14-3-3 proteins play a role in the cell cycle by shielding cdt2 from ubiquitin-mediated degradation. Mol Cell Biol 34:4049–4061. https://doi.org/10.1128/MCB.00838-14
Davis R, Turner D (2001). Vertebrate hairy and Enhancer of split related proteins: transcriptional repressors regulating cellular differentiation and embryonic patterning. Oncogene 20(58):8342–8357. https://doi.org/10.1038/sj.onc.1205094
Fan B, Malato Y, Calvisi DF, Naqvi S, Razumilava N, Ribback S, Gores GJ, Dombrowski F, Evert M, Chen X, Willenbring H (2012) Cholangiocarcinomas can originate from hepatocytes in mice. J Clin Invest 122:2911–2915. https://doi.org/10.1172/JCI63212
Gebäck T, Schulz MMP, Koumoutsakos P, Detmar M (2009) TScratch: a novel and simple software tool for automated analysis of monolayer wound healing assays. BioTechniques 46:265–274. https://doi.org/10.2144/000113083
Grabher C, Boehmer von H, Look AT (2006) Notch 1 activation in the molecular pathogenesis of T-cell acute lymphoblastic leukaemia. Nat Rev Cancer 6:347–359. https://doi.org/10.1038/nrc1880
Greenwald I (1998) LIN-12/Notch signaling: lessons from worms and flies. Genes Dev 12:1751–1762
Greenwald I (2005) LIN-12/Notch signaling in C. elegans. WormBook : the online review of C elegans biology 1–16. https://doi.org/10.1895/wormbook.1.10.1
Haonon O, Rucksaken R, Pinlaor P, Pairojkul C, Chamgramol Y, Intuyod K, Onsurathum S, Khuntikeo N, Pinlaor S (2016) Upregulation of 14-3-3 eta in chronic liver fluke infection is a potential diagnostic marker of cholangiocarcinoma. Proteom Clin Appl 10:248–256. https://doi.org/10.1002/prca.201500019
Hermeking H (2003) The 14-3-3 cancer connection. Nat Rev Cancer 3:931–943. https://doi.org/10.1038/nrc1230
Kapuria S, Karpac J, Biteau B, Hwangbo DS, Jasper H (2012) Notch-mediated suppression of TSC2 expression regulates cell differentiation in the Drosophila intestinal stem cell lineage. PLoS Genet 8:e1003045–e1003014. https://doi.org/10.1371/journal.pgen.1003045
Kovall RA, Blacklow SC (2010) Chapter two - mechanistic insights into Notch receptor signaling from structural and biochemical studies. Curr Top Dev Biol 92(92):31–71. https://doi.org/10.1016/S0070-2153(10)92002-4
Kurooka H, Honjo T (2000) Functional interaction between the mouse notch1 intracellular region and histone acetyltransferases PCAF and GCN5. J Biol Chem 275:17211–17220. https://doi.org/10.1074/jbc.M000909200
Li Y, Inoki K, Yeung R, Guan K-L (2002) Regulation of TSC2 by 14-3-3 binding. J Biol Chem 277:44593–44596. https://doi.org/10.1074/jbc.C200510200
Meier-Stiegen F, Schwanbeck R, Bernoth K, Martini S, Hieronymus T, Ruau D, Zenke M, Just U (2010) Activated Notch1 target genes during embryonic cell differentiation depend on the cellular context and include lineage determinants and inhibitors. PLoS One 5:e11481. https://doi.org/10.1371/journal.pone.0011481
Meurette O, Stylianou S, Rock R, Collu GM, Gilmore AP, Brennan K (2009) Notch activation induces Akt signaling via an autocrine loop to prevent apoptosis in breast epithelial cells. Cancer Res 69:5015–5022. https://doi.org/10.1158/0008-5472.CAN-08-3478
Nakagawa O, McFadden DG, Nakagawa M et al (2000) Members of the HRT family of basic helix-loop-helix proteins act as transcriptional repressors downstream of Notch signaling. Proc Natl Acad Sci U S A 97:13655–13660. https://doi.org/10.1073/pnas.250485597
Paemanee A, Wikan N, Roytrakul S, Smith DR (2016) Application of GelC-MS/MS to proteomic profiling of Chikungunya virus infection: preparation of peptides for analysis. In: Application of GelC-MS/MS to proteomic profiling of Chikungunya virus infection: preparation of peptides for analysis. Springer New York, New York, pp 179–193
Palaga T, Ratanabunyong S, Pattarakankul T, Sangphech N, Wongchana W, Hadae Y, Kueanjinda P (2013) Notch signaling regulates expression of Mcl-1 and apoptosis in PPD-treated macrophages. Cell Mol Immunol 10:444–452. https://doi.org/10.1038/cmi.2013.22
Perumalsamy LR, Nagala M, Sarin A (2010) Notch-activated signaling cascade interacts with mitochondrial remodeling proteins to regulate cell survival. Proc Natl Acad Sci U S A 107:6882–6887. https://doi.org/10.1073/pnas.0910060107
Ranganathan P, Weaver KL, Capobianco AJ (2011) Notch signalling in solid tumours: a little bit of everything but not all the time. Nat Rev Cancer 11:338–351. https://doi.org/10.1038/nrc3035
Rattanasinganchan P, Leelawat K, Treepongkaruna S-A, Tocharoentanaphol C, Subwongcharoen S, Suthiphongchai T, Tohtong R (2006) Establishment and characterization of a cholangiocarcinoma cell line (RMCCA-1) from a Thai patient. World J Gastroenterol 12:6500–6506. https://doi.org/10.3748/wjg.v12.i40.6500
Reinhardt HC, Yaffe MB (2013) Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response. Nat Rev Mol Cell Biol 14:563–580. https://doi.org/10.1038/nrm3640
Sade H, Krishna S, Sarin A (2004) The anti-apoptotic effect of Notch-1 requires p56lck-dependent, Akt/PKB-mediated signaling in T cells. J Biol Chem 279:2937–2944. https://doi.org/10.1074/jbc.M309924200
Sirisinha S, Tengchaisri T, Boonpucknavig S, Prempracha N, Ratanarapee S, Pausawasdi A (1991) Establishment and characterization of a cholangiocarcinoma cell line from a Thai patient with intrahepatic bile duct cancer. Asian Pac J Allergy Immunol 9:153–157
Stylianou S, Clarke R, Brennan K (2006) Aberrant activation of Notch signaling in human breast cancer. Cancer Res 66:1517–1525
Tit-oon P, Chokchaichamnankit D, Khongmanee A et al (2014) Comparative secretome analysis of cholangiocarcinoma cell line in three-dimensional culture. Int J Oncol 45:2108–2116. https://doi.org/10.3892/ijo.2014.2636
Zender S, Nickeleit I, Wuestefeld T, Sörensen I, Dauch D, Bozko P, el-Khatib M, Geffers R, Bektas H, Manns MP, Gossler A, Wilkens L, Plentz R, Zender L, Malek NP (2013) A critical role for Notch signaling in the formation of cholangiocellular carcinomas. Cancer Cell 23:784–795. https://doi.org/10.1016/j.ccr.2013.04.019
Zhang C, Liu L-X, Dong Z-R, Shi GM, Cai JB, Zhang PF, Ke AW, Yu JX, Zhou J, Fan J (2015) Up-regulation of 14-3-3ζ expression in intrahepatic cholangiocarcinoma and its clinical implications. Tumour Biol 36:1781–1789. https://doi.org/10.1007/s13277-014-2780-5
Acknowledgments
The work was supported by the Thailand Research Fund (TRG5780200) and Mahidol University (T.K.). The authors thank the Central Instrument Facility, Center of Nanoimaging, Faculty of Science, Mahidol University and Sucheewin Krobthong for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singrang, N., Kittisenachai, S., Roytrakul, S. et al. NOTCH1 regulates the viability of cholangiocarcinoma cells via 14-3-3 theta. J. Cell Commun. Signal. 13, 245–254 (2019). https://doi.org/10.1007/s12079-018-0488-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-018-0488-9