Abstract
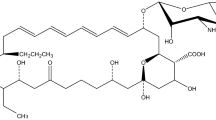
Pravastatin is one of the most popular cholesterol-lowering drugs. Its industrial production represents a two-stage process including the microbial production of compactin and its further biocatalytic conversion to pravastatin. To increase a conversion rate, a higher compactin content in fermentation medium should be used; however, high compactin concentrations inhibit microbial growth. Therefore, the improvement of the compactin resistance of a producer still remains a relevant problem. A multi-step random UV mutagenesis of a Streptomyces xanthochromogenes strain RIA 1098 and the further selection of high-yield compactin-resistant mutants have resulted in a highly productive compactin-resistant strain S 33-1. After the fermentation medium improvement, the maximum bioconversion rate of this strain has reached 91 % at the daily compactin dose equal to 1 g/L and still remained high (83 %) even at the doubled dose (2 g/L). A 1-year study of the mutant strain stability has proved a stable inheritance of its characteristics that provides this strain to be very promising for the pravastatin-producing industry.

Similar content being viewed by others
References
Dulak J, Józkowicz A (2005) Anti-angiogenic and anti-inflammatory effects of statins: relevance to anti-cancer therapy. Curr Cancer Drug Targets 5:579–594. doi:10.2174/156800905774932824
Kataria P, Kaur J, Parvez E, Maurya RP (2014) Statins: the paradigm shift in periodontal regeneration. SRM J Res Dent Sci 5:26–30. doi:10.4103/0976-433X.129069
dos Santos LF, de Carvalho JC, Rubel R, Soccol CR (2013) Microbial statins. In: Brar SK, Dhillon GS, Soccol CR (eds) Biotransformation of waste biomass into high value biochemicals. Springer, New York, pp 313–335. doi:10.1007/978-1-4614-8005-1_13
del Sol AI, Nanayakkara PWB (2008) Pravastatin: An evidence-based statin? Expert Opin Drug Metab Toxicol 4:821–825. doi:10.1517/17425255.4.6.821
Al-Badr AA, Mostafa GAE (2014) Pravastatin sodium. In: Brittain HG (ed) Profiles of drug substances, excipients, and related methodology. Elsevier, San Diego, pp 433–513. doi:10.1016/B978-0-12-800173-8.00008-8
Salat J, Mozes J, Bidlo M, Horvath G, Boros S, Barta I, Birincsik L, Albrecht K, Ambrus G, Konya A, Andor A, Jekkel A, Lang I, Ilkoy E, Szabo I (2005) Hydroxylation of compactin to pravastatin by Micromonospora. US Patent 6,905,851
Salat J, Mozes NSJ, Bidlo NIG, Horvath G, Boros S, Barta I, Somogyi G, Birincsik L, Albrecht K, Ambrus G, Konya A, Andor A, Jekkel A, Lang I, Ilkoy E, Szabo I (2007) Microbial process for preparing pravastatin. Canadian Patent 2361701
Lin C-L, Tang Y-L, Lin S-M (2011) Efficient bioconversion of compactin to pravastatin by the quinoline-degrading microorganism Pseudonocardia carboxydivorans isolated from petroleum-contaminated soil. Bioresour Technol 102:10187–10193. doi:10.1016/j.biortech.2011.09.029
Mei M, Ji X, Gao X, Chen Y, Li Y, Yao Y, Zhuo Z, Xu J (2009) Microorganism and the process for preparation of pravastatin sodium. US Patent 7,582,464
Peng YL, Demain AL (2000) Bioconversion of compactin to pravastatin by Actinomadura sp. ATCC 55678. J Mol Catal B Enzym 10:151–156. doi:10.1016/s1381-1177(00)00123-5
Zhang L, Zhang J, Yang W, Bai G (2008) Classification of Streptomyces strain Z314 and purification of its product pravastatin. Wei Sheng Wu Xue Bao 48:33–37
Park JW, Lee JK, Kwon TJ, Yi DH, Kim YJ, Moon SH, Suh HH, Kang SM, Park YI (2003) Bioconversion of compactin into pravastatin by Streptomyces sp. Biotechnol Lett 25:1827–1831. doi:10.1023/A:1026281914301
Ukraintseva SN, Voinova TM, Dzhavakhiya VG (2004) Penicillium citrinum strain improvement for compactin production by induced-mutagenesis and optimization of obtained mutant cultivation conditions. In: Zaikov GE (ed) Biotechnology and medicine. Nova Science Publisher Inc., New York, pp 71–78
Endo A, Kuroda M, Tsujita Y (1976) ML-236A, ML-236B and ML-236C, new inhibitors of cholesterogenesis produced by Penicillium citrinum. J Antibiot (Tokyo) 29:1346–1348. doi:10.7164/antibiotics.29.1346
Dzhavakhiya VV, Voinova TM (2004) Optimization of fermentation conditions for high lovastatin producing mutant 45–50 of fungus Aspergillus terreus. In: Zaikov GE (ed) Biotechnology and industry. Nova Science Publisher Inc., New York, pp 81–87
Lane DJ (1991) 16S/23S sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York, pp 115–175
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 84:5463–5467. doi:10.1073/pnas.74.12.5463
Stackebrandt E, Ebers J (2006) Taxonomic parameters revisited: tarnished gold standards. Microbiol Today 33:152–155
Lee C-K, Kim D-Y, Suh J-W, Chang J-H (2004) Streptomyces sp. CJPV975652 capable of converting compactin to pravastatin and method for producing pravastatin using the same. International Patent Application WO2004111205
Lee J-K, Park J-W, Seo D-J, Lee S-C, Kim J-Y (2001) Microorganism Streptomyces exfoliatus YJ-118 and a method for producing pravastatin sodium by using the strain. US Patent 6,306,629
Chen C-H, Hu H-Y, Cho Y-C, Hsu W-H (2006) Screening of compactin-resistant microorganisms capable of converting compactin to pravastatin. Curr Microbiol 53:108–112. doi:10.1007/s00284-005-0276-7
Szabo L, Tchelet R (2010) Process for constructing strain having compactin hydroxylation ability. US Patent 7,700,337
Fujii T, Fujii Y, Machida K, Ochiai A, Ito M (2009) Efficient biotransformations using Escherichia coli with tolC acrAB mutations expressing cytochrome P450 genes. Biosci Biotechnol Biochem 73:805–810. doi:10.1271/bbb.80627
Fujii Y, Norihisa K, Fujii T, Aritoku Y, Kagawa Y, Sallam KI, Johdo O, Arisawa A, Tamura T (2011) Construction of a novel expression vector in Pseudonocardia autotrophica and its application to efficient biotransformation of compactin to pravastatin, a specific HMG-CoA reductase inhibitor. Biochem Biophys Res Commun 404:511–516. doi:10.1016/j.bbrc.2010.12.013
McLean KJ, Hans M, Meijrink B, van Scheppingen WB, Vollebregt A, Tee KL, van der Laan J-M, Leys D, Munro AW, van den Berg MA (2015) Single-step fermentative production of the cholesterol-lowering drug pravastatin via reprogramming of Penicillium chrysogenum. Proc Natl Acad Sci 112:2847–2852. doi:10.1073/pnas.1419028112
Kostova I, Ivanova N, Losev V, Dimitrova A, Vasileva R, Todorova D (2004) Method for production of pravastatin by fermentation. European Patent 1452602
Lee F-Y, Lee M-L, Hong AC, Chiu S-C (2007) Strains of Saccharotrix, process for producing pravastatin using the strains and isolation process of (HMG-CoA) reductase. US Patent 7,202,062
Choi N-H, Tak K-T, Lee K-W, Kim N-H, Jun J-C, Kong Y-L, Lee K-M (2007) Process for preparing pravastatin sodium. US Patent 7,223,590
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dzhavakhiya, V.V., Voinova, T.M., Glagoleva, E.V. et al. Strain Improvement of Streptomyces xanthochromogenes RIA 1098 for Enhanced Pravastatin Production at High Compactin Concentrations. Indian J Microbiol 55, 440–446 (2015). https://doi.org/10.1007/s12088-015-0537-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-015-0537-5