Abstract
Purpose
This prospective observational study assessed the efficacy of bevacizumab in combination with chemotherapy as preoperative treatment to downsize tumours for radical resection in patients with unresectable metastatic colorectal cancer (mCRC).
Patients/methods
Patients with mCRC initially unresectable according to predefined criteria were included. Preoperative treatment consisted of bevacizumab (5 mg/kg) combined with oxaliplatin- or irinotecan-based chemotherapy, which was followed by surgery in patients showing clinical benefit. Resection rate was the primary endpoint. Response rate (RR) and clinical benefit of preoperative chemotherapy, and overall survival (OS) were secondary endpoints.
Results
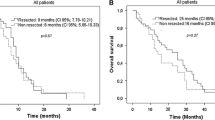
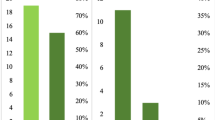
A total of 120 eligible patients were included and received preoperative treatment. Chemotherapy was irinotecan-based in 73 (61 %) patients, oxaliplatin-based in 25 (21 %) and 22 (18 %) patients received more than one line. A RR of 30 % and a clinical benefit rate of 73 % were observed with preoperative chemotherapy. Metastatic resection was possible in 61 (51 %) patients. Median OS was 33 months (95 % CI 31–NA months) for patients undergoing surgery, and 15 months (95 % CI 11–25 months) in non-operated patients. Thirty-five patients experienced 59 postoperative complications (morbidity rate 57 %).
Conclusion
Preoperative bevacizumab-based chemotherapy offers a high surgical rescue rate in patients with initially unresectable mCRC.

Similar content being viewed by others
References
Jemal A, Siegel R, Ward E et al (2009) Cancer statistics. CA Cancer J Clin 59:225–249
Scheele J, Stangl R, Tendorf-Hofmann A (1990) Hepatic metastases from colorectal carcinoma: impact of surgical resection on the natural history. Br J Surg 77:1241–1246
Mayo SC, Pawlik TM (2009) Current management of colorectal hepatic metastasis. Expert Rev Gastroenterol Hepatol 3:131–144
Fong Y, Fortner J, Sun RL et al (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230:309–318
Figueras J, Valls C, Rafecas A et al (2001) Resection rate and effect of postoperative chemotherapy on survival after surgery for colorectal liver metastases. Br J Surg 88:980–985
Stangl R, Tendorf-Hofmann A, Charnley RM et al (1994) Factors influencing the natural history of colorectal liver metastases. Lancet 343:1405–1410
Figueras J, Torras J, Valls C et al (2007) Surgical resection of colorectal liver metastases in patients with expanded indications: a single-center experience with 501 patients. Dis Colon Rectum 50:478–488
Neeff H, Horth W, Makowiec F et al (2009) Outcome after resection of hepatic and pulmonary metastases of colorectal cancer. J Gastrointest Surg 13:1813–1820
Shah SA, Haddad R, Al-Sukhni W et al (2006) Surgical resection of hepatic and pulmonary metastases from colorectal carcinoma. J Am Coll Surg 202:468–475
Elias D, Liberale G, Vernerey D et al (2005) Hepatic and extrahepatic colorectal metastases: when resectable, their localization does not matter, but their total number has a prognostic effect. Ann Surg Oncol 12:900–909
Adam R, Delvart V, Pascal G et al (2004) Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg 240:644–657
Nordlinger B, Van CE, Rougier P et al (2007) Does chemotherapy prior to liver resection increase the potential for cure in patients with metastatic colorectal cancer? A report from the European Colorectal Metastases treatment group. Eur J Cancer 43:2037–2045
Masi G, Loupakis F, Pollina L et al (2009) Long-term outcome of initially unresectable metastatic colorectal cancer patients treated with 5-fluorouracil/leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) followed by radical surgery of metastases. Ann Surg 249:420–425
Blazer DG III, Kishi Y, Maru DM et al (2008) Pathologic response to preoperative chemotherapy: a new outcome end point after resection of hepatic colorectal metastases. J Clin Oncol 26:5344–5351
Nordlinger B, Van CE, Gruenberger T et al (2009) Combination of surgery and chemotherapy and the role of targeted agents in the treatment of patients with colorectal liver metastases: recommendations from an expert panel. Ann Oncol 20:985–992
Gruenberger B, Tamandl D, Schueller J et al (2008) Bevacizumab, capecitabine, and oxaliplatin as neoadjuvant therapy for patients with potentially curable metastatic colorectal cancer. J Clin Oncol 26:1830–1835
Ribero D, Wang H, Donadon M et al (2007) Bevacizumab improves pathologic response and protects against hepatic injury in patients treated with oxaliplatin-based chemotherapy for colorectal liver metastases. Cancer 110:2761–2767
Okines A, Puerto OD, Cunningham D et al (2009) Surgery with curative-intent in patients treated with first-line chemotherapy plus bevacizumab for metastatic colorectal cancer First BEAT and the randomised phase-III NO16966 trial. Br J Cancer 101:1033–1038
D’Angelica M, Kornprat P, Gonen M et al (2007) Lack of evidence for increased operative morbidity after hepatectomy with perioperative use of bevacizumab: a matched case-control study. Ann Surg Oncol 14:759–765
Kesmodel SB, Ellis LM, Lin E et al (2008) Preoperative bevacizumab does not significantly increase postoperative complication rates in patients undergoing hepatic surgery for colorectal cancer liver metastases. J Clin Oncol 26:5254–5260
Reddy SK, Morse MA, Hurwitz HI et al (2008) Addition of bevacizumab to irinotecan- and oxaliplatin-based preoperative chemotherapy regimens does not increase morbidity after resection of colorectal liver metastases. J Am Coll Surg 206:96–106
Mahfud M, Breitenstein S, El-Badry AM et al (2010) Impact of preoperative bevacizumab on complications after resection of colorectal liver metastases: case-matched control study. World J Surg 34:92–100
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid Tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Couinaud C (1954) Anatomic principles of left and right regulated hepatectomy: technics. J Chir (Paris) 70:933–966
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Van Cutsem E, Rivera F, Berry S et al (2009) Safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study. Ann Oncol 20:1842–1847
Saltz LB, Clarke S, Diaz-Rubio E et al (2008) Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol 26:2013–2019
Baumgaertner I, Ratziu V, Vaillant JC et al (2010) Hepatotoxicity of metastatic colorectal cancer chemotherapy: systematic review. Bull Cancer 97:559–569
Chun YS, Vauthey JN, Boonsirikamchai P et al (2009) Association of computed tomography morphologic criteria with pathologic response and survival in patients treated with bevacizumab for colorectal liver metastases. JAMA 302:2338–2344
Cleary JM, Tanabe KT, Lauwers GY et al (2009) Hepatic toxicities associated with the use of preoperative systemic therapy in patients with metastatic colorectal adenocarcinoma to the liver. Oncologist 14:1095–1105
Acknowledgments
We thank Carmen Carmona MD for helping to collect data. This work was supported by Roche Farma S.A.
Conflict of interest
Dr J Figueras is a member of the Speakers’ Bureau for Avastin.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Figueras, J., Lopez-Ben, S., Alsina, M. et al. Preoperative treatment with bevacizumab in combination with chemotherapy in patients with unresectable metastatic colorectal carcinoma. Clin Transl Oncol 15, 460–466 (2013). https://doi.org/10.1007/s12094-012-0952-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-012-0952-6