Abstract
Objective
Although peroxiredoxin 3 (PRX3) was reported to be overexpressed in liver cancer, the precise function of PRX3 in the development and/or progression of liver cancer remained to be obscure. The present study was conducted to investigate the response of PRX3 to oxidative stress in hepatocellular carcinoma (HCC) cells.
Methods
After successful knockdown of PRX3 expression by small interfering RNA, we treated HCC cell lines Hep-3b and Hep-G2 with gradient concentrations of H2O2 and detected cell proliferation, apoptosis, and the level of reactive oxygen species (ROS) in the cells.
Results
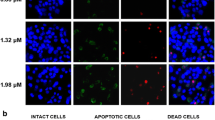
After low-dose (5–20 μmol/l) H2O2 treatment, the ROS level was significantly higher in PRX3-knockdown Hep-3b cells than in controls. In addition, PRX3 down-regulation resulted in decreased proliferation, increased apoptosis, and increased caspase 3 activity of Hep-3b cells. We did not notice significant difference between PrxIII knockdown and control Hep-G2 cells in ROS level, cell viability or apoptosis.
Conclusion
Our results suggest that PRX3 is an indispensable ROS scavenger that protects tumor cells against oxidative damage and subsequent apoptosis, which provides a clue that PRX3 may be involved in the chemotherapeutic resistance of liver cancer. The underlying mechanism for PRX3 function needs further investigation.




Similar content being viewed by others
References
World Health Organization. Global alert and response (GAR). In: Hepatitis B: surveillance and control. 2009. http://www.who.int/csr/disease/hepatitis/whocdscsrlyo20022/en/index4.html. Accessed 23 Oct 2009.
Lu FM, Zhuang H. Management of hepatitis B in China. Chin Med J (Engl). 2009;122:3–4.
Song P, Feng X, Zhang K, Song T, Ma K, Kokudo N, et al. Screening for and surveillance of high-risk patients with HBV-related chronic liver disease: promoting the early detection of hepatocellular carcinoma in China. Biosci Trends. 2013;7:1–6.
Yeh CT, Chen HC, Sung CM, Hsu CL, Lin CC, Pan KT, et al. Retrospective comparison between a regular and a split-dose protocol of 5-fluorouracil, cisplatin, and mitoxantrone for the treatment of far advanced hepatocellular carcinoma. BMC Cancer. 2011;11:117.
Zhang ZM, Guo JX, Zhang ZC, Jiang N, Zhang ZY, Pan LJ. Therapeutic options for intermediate-advanced hepatocellular carcinoma. World J Gastroenterol. 2011;17:1685–9.
Srinivas P, Gopinath G, Banerji A, Dinakar A, Srinivas G. Plumbagin induces reactive oxygen species, which mediate apoptosis in human cervical cancer cells. Mol Carcinog. 2004;40:201–11.
Alexandre J, Batteux F, Nicco C, Chéreau C, Laurent A, Guillevin L, et al. Accumulation of hydrogen peroxide is an early and crucial step for paclitaxel-induced cancer cell death both in vitro and in vivo. Int J Cancer. 2006;119:41–8.
Chung YM, Yoo YD, Park JK, Kim YT, Kim HJ. Increased expression of peroxiredoxin II confers resistance to cisplatin. Anticancer Res. 2001;21:1129–33.
Zhang B, Su Y, Ai G, Wang Y, Wang T, Wang F. Involvement of peroxiredoxin I in protecting cells from radiation-induced death. J Radiat Res (Tokyo). 2005;46:305–12.
Wang T, Tamae D, LeBon T. The role of peroxiredoxin II in radiation-resistant MCF-7 breast cancer cells. Cancer Res. 2005;65:10338–46.
Watabe S, Hiroi T, Yamamoto Y, Fujioka Y, Hasegawa H, Yago N, et al. SP-22 is a thioredoxin-dependent peroxide reductase in mitochondria. Eur J Biochem. 1997;249:52–60.
Chae HZ, Kim HJ, Kang SW, Rhee SG. Characterization of three isoforms of mammalian peroxiredoxin that reduce peroxides in the presence of thioredoxin. Diabetes Res Clin Pract. 1999;45:101–12.
Chang TS, Cho CS, Park S. Peroxiredoxin III, a mitochondrion-specific peroxidase, regulates apoptotic signaling by mitochondria. J Biol Chem. 2004;279:41975–84.
Nonn L, Berggren M, Powis G. Increased expression of mitochondrial peroxiredoxin-3 (thioredoxin peroxidase-2) protects cancer cells against hypoxia and drug-induced hydrogen peroxide-dependent apoptosis. Mol Cancer Res. 2003;1:682–9.
Chua PJ, Lee EH, Yu Y, Yip GW, Tan PH, Bay BH. Silencing the Peroxiredoxin III gene inhibits cell proliferation in breast cancer. Int J Oncol. 2010;36:359–64.
Duan J, Lang Y, Song C, Xiong J, Wang Y, Yan Y. siRNA targeting of PRDX3 enhances cisplatin-induced apoptosis in ovarian cancer cells through the suppression of the NF-κB signaling pathway. Mol Med Rep. 2013;7:1688–94.
Li L, Kaifu T, Obinata M, Takai T. Peroxiredoxin III-deficiency sensitizes macrophages to oxidative stress. J Biochem. 2009;145:425–7.
Shih SF, Wu YH, Hung CH, Yang HY, Lin JY. Abrin triggers cell death by inactivating a thiol-specific antioxidant protein. J Biol Chem. 2001;276:21870–7.
Wang XY, Wang HJ, Li XQ. Peroxiredoxin III protein expression is associated with platinum resistance in epithelial ovarian cancer. Tumour Biol. 2013;34:2275–81.
Whitaker HC, Patel D, Howat WJ, Warren AY, Kay JD, Sangan T, et al. Peroxiredoxin-3 is overexpressed in prostate cancer and promotes cancer cell survival by protecting cells from oxidative stress. Br J Cancer. 2013. doi:10.1038/bjc.2013.396.
Choi JH, Kim TN, Kim S. Overexpression of mitochondrial thioredoxin reductase and peroxiredoxin III in hepatocellular carcinomas. Anticancer Res. 2002;22:3331–5.
Qiao B, Wang J, Xie J, Niu Y, Ye S, Wan Q, et al. Detection and identification of peroxiredoxin 3 as a biomarker in hepatocellular carcinoma by a proteomic approach. Int J Mol Med. 2012;29:832–40.
Li L, Zhang YG, Chen CL. Anti-apoptotic role of peroxiredoxin III in cervical cancer cells. FEBS Open Bio. 2013;3:51–4.
Shibata E, Nanri H, Ejima K, Araki M, Fukuda J, Yoshimura K, et al. Enhancement of mitochondrial oxidative stress and up-regulation of antioxidant protein peroxiredoxin III/SP-22 in the mitochondria of human pre-eclamptic placentae. Placenta. 2003;24:698–705.
Li L, Shoji W, Oshima H, Obinata M, Fukumoto M, Kanno N. Crucial role of peroxiredoxin III in placental antioxidant defense of mice. FEBS Lett. 2008;582:2431–4.
Shen C, Nathan C. Nonredundant antioxidant defense by multiple two-cysteine peroxiredoxins in human prostate cancer cells. Mol Med. 2002;8:95–102.
Karihtala P, Mäntyniemi A, Kang SW, Kinnula VL, Soini Y. Peroxiredoxins in breast carcinoma. Clin Cancer Res. 2003;9:3418–24.
Park JH, Kim YS, Lee HL, Shim JY, Lee KS, Oh YJ, et al. Expression of peroxiredoxin and thioredoxin in human lung cancer and paired normal lung. Respirology. 2006;11:269–75.
Wu XY, Fu ZX, Wang XH. Peroxiredoxins in colorectal neoplasms. Histol Histopathol. 2010;25:1297–303.
Safaeian M, Hildesheim A, Gonzalez P, Yu K, Porras C, Li Q, et al. Single nucleotide polymorphisms in the PRDX3 and RPS19 and risk of HPV persistence and cervical precancer/cancer. PLoS ONE. 2012;7:e33619.
Conflict of interest
The authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, YG., Li, L., Liu, CH. et al. Peroxiredoxin 3 is resistant to oxidation-induced apoptosis of Hep-3b cells. Clin Transl Oncol 16, 561–566 (2014). https://doi.org/10.1007/s12094-013-1117-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-013-1117-y