Abstract
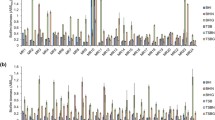
Staphylococcus hominis is the third species of coagulase-negative staphylococci (CoNS) most frequently isolated from specimens of patients with hospital-acquired infections. Many infections caused by CoNS appeared to be associated with biofilms. Nevertheless, the knowledge of the ability of S. hominis to form a biofilm is limited. The aim of this study was to analyze the formation of the biofilm by 56 S. hominis strains isolated from clinical cases. The biofilm three-dimensional structure was reconstructed by confocal laser scanning microscopy. We found that most of S. hominis strains carried icaADBC genes encoding polysaccharide intercellular adhesin (PIA), which plays a crucial role in the formation of biofilms in staphylococci strains. However, only a half of the ica-positive strains had an ability to form a biofilm in vitro. In this study, we also accessed the sensitivity of biofilms of S. hominis strains to sodium metaperiodate, proteinase K and DNase. We found that polysaccharides and proteins are the major components of the extracellular matrix of the biofilm formed by S. hominis. DNase did not have a significant effect on biofilms, which suggested that nucleic acid plays a minor role in the mature biofilm.


Similar content being viewed by others
References
Bouchami O, Ben Hassen A, de Lencastre H, Miragaia M (2011) Molecular epidemiology of methicillin-resistant Staphylococcus hominis (MRSHo): low clonality and reservoirs of SCCmec structural elements. PLoS ONE 6:e21940
Chaves F, García-Álvarez M, Sanz F, Alba C, Otero JR (2005) Nosocomial spread of a Staphylococcus hominis subsp. novobiosepticus strain causing sepsis in a neonatal intensive care unit. J Clin Microbiol 43:4877–4879
Chokr A, Watier D, Eleaume H, Pangon B, Ghnassia J-C, Mack D, Jabbouri S (2006) Correlation between biofilm formation and production of polysaccharide intercellular adhesin in clinical isolates of coagulase-negative staphylococci. Int J Med Microbiol 296:381–388
Christensen GD, Simpson A, Younger JJ, Baddour LM, Barrett FF, Melton DM, Beachey EH (1985) Adherence of coagulase-negative staphylococci to plastic tissue culture plates: a quantitative model for the adherence of staphylococci to medical devices. J Clin Microbiol 22:996–1006
De Silvia GDI, Kantzanou M, Justice A, Massey RC, Wilkinson AR, Day NPJ, Peacock SJ (2002) The ica operon and biofilm production in coagulase-negative staphylococci associated with carriage and disease in neonatal intensive care unit. J Clin Microbiol 40:382–388
Fitzpatrick F, Humphreys H, O'Gara JP (2005) Evidence for icaADBC-independent biofilm development mechanism in methicillin-resistant Staphylococcus aureus clinical isolates. J Clin Microbiol 43:973–1976
Frank KL, del Pozo JL, Pater R (2008) From clinical microbiology to infection pathogenesis: how daring to be different works for Staphylococcus lugdunensis. Clin Microbiol Rev 21:111–133
Fredheim EGA, Klingenberg C, Rodhe H, Frankenberger S, Gaustad P, Flaegstad T, Sollid JE (2009) Biofilm formation by Staphylococcus haemolyticus. J Clin Microbiol 47:1172–1180
Götz F (2006) The genera Staphylococcus and Macrococcus. In: Dworkin M, Falkow S, Rosenberig E, Schleiferr K-H, Stackebrands E (eds) The Prokaryotes, vol 4, 3rd edn, A Handbook on the biology of bacteria: Firmicutes, Cyanobacteria., pp 5–75
Iorio NLP, Lopez APCN, Schuenck RP, Barcellos AG, Olendzki AN, Lopez GL, dos Santos KRN (2011) A combination of methods to evaluate biofilm production may help to determine the clinical relevance of Staphylococcus in blood cultures. Microbiol Immunol 55:28–33
Izano EA, Amarante MA, Kher WB, Kaplan JB (2008) Differential roles of poly-N acetylglucosamine surface polysaccharide and extracellular DNA in Staphylococcus aureus and Staphylococcus epidermidis biofilms. Appl Environ Microbiol 74:470–476
Kaufman D, Fairchild D (2004) Clinical microbiology of bacterial and fungal sepsis in very-low-birth-weight infants. Clin Microbiol Rev 17:638–680
Kim J-H, Kim C-H, Hacker J, Ziebuhr W, Lee BK, Cho S-H (2008) Molecular characterization of regulatory genes associated with biofilm variation in Staphylococcus aureus strains. J Microbiol Biotechnol 18:28–34
Kloos WE, Bannerman TL (1999) Staphylococcus and Micrococcus. In: Murray PR, Baron E, Pfallen MA, Tenover FC, Yolken R (eds) Manual of clinical microbiology. ASM Press, Washington, pp 264–282
Lim Y, Jana M, Luong TT, Lee CY (2004) Control of glucose- and NaCl-induced biofilm formation by rbf in Staphylococcus aureus. J Bacteriol 186:722–729
Mack D, Davies AP, Harris LG, Rodhe H, Horstkotte MA, Knobloch JK-M (2007) Microbial interaction in Staphylococcus epidermidis biofilms. Anal Bioanal Chem 387:399–408
Nuryastuti T, van der Mei HC, Busscher HJ, Kuijer R, Aman AT, Krom BP (2008) recA mediated spontaneous deletion of the icaADBC operon of clinical Staphylococcus epidermidis isolates: a new mechanism of phenotypic variations. Antonie Van Leeuwenhoek 94:317–328
Qin Z, Yang X, Yang L, Jiang J, Qu Y, Molin S, Qu D (2007a) Formation and properties of in vitro biofilms of ica-negative Staphylococcus epidermidis clinical isolates. J Med Microbiol 56:83–93
Qin Z, Ou Y, Yang L, Zhu Y, Tolker-Nielsen T, Molin S, Qu D (2007b) Role of autolysin-mediated DNA release in biofilm formation of Staphylococcus epidermidis. Microbiology 153:2083–2092
Rodhe H, Mack D, Christner M, Burdelski C, Franke G, Knobloch JK-M (2006) Pathogenesis of staphylococcal device-related infections: from basic science to new diagnostic, therapeutic and prophylactic approaches. Rev Med Microbiol 17:45–54
Rodhe H, Burandt EC, Siemssen N, Frommelt L, Burdelski C, Wurster S, Scherpe S, Davies AP, Harris LG, Horstkotte MA, Knobloch JK-M, Ragunath C, Kaplan JB, Mack D (2007) Polysaccharide intercellular adhesion or protein factors in biofilm accumulation of Staphylococcus epidermidis and Staphylococcus aureus isolated from prosthetic hip and joint infections. Biomaterials 28:1711–1720
Rodhe H, Frankenberger S, Zähringer U, Mack D (2010) Structure, function and contribution of polysaccharide intercellular adhesin (PIA) to Staphylococcus epidermidis biofilm formation and pathogenesis of biofilm-associated infections. Eur J Cell Biol 89:103–111
Schommer NN, Christner M, Hentschke M, Ruckdeschel K, Aepfelbacher M, Rohde H (2011) Staphylococcus epidermidis uses distinct mechanisms of biofilm formation to interfere with phagocytosis and activation of mouse macrophage-like cells 774A.1. Infect Immun 79:2267–2276
Sorlozano A, Gutierrez J, Martinez T, Yuste ME, Perez-Lopez JA, Vindel A, Guillen J, Boquete T (2010) Detection of new mutations conferring resistance to linezolid in glycopeptide-intermediate susceptibility Staphylococcus hominis subspecies hominis circulating in an intensive care unit. Eur J Clin Microbiol Infect Dis 29:73–80
Sousa C, Teixeira P, Oliveira R (2009) The role of extracellular polymers on Staphylococcus epidermidis biofilm biomass and metabolic activity. J Basic Microbiol 49:363–370
Zhang L, Thomas JC, Miragaia M, Bouchami O, Chaves F, Pedro A, d’Azevedo PA, David Aanensen DM, de Lencastre H, Gray BM, Ashley D, Robinson DA (2013) Multilocus sequence typing and further genetic characterization of the enigmatic pathogen, Staphylococcus hominis. PLoS ONE 8:e66496
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Szczuka, E., Telega, K. & Kaznowski, A. Biofilm formation by Staphylococcus hominis strains isolated from human clinical specimens. Folia Microbiol 60, 1–5 (2015). https://doi.org/10.1007/s12223-014-0332-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-014-0332-4