Abstract
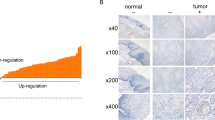
Matrix metalloproteinases-2 (MMP-2) and the tissue inhibitor of metalloproteinase-2 (TIMP-2), may presumably have an important role on the invasion and metastatic spread of malignancies attributed to an uncontrolled degradation of the extracellular matrix (ECM). A retrospective chart analysis was carried out to study the expression of MMP-2 and TIMP-2 on the archival samples of oral squamous cell carcinoma (OSCC) (n = 30) and normal mucosa (n = 10) by immunohistochemistry and compared with the clinicopathologic parameters of cases. Both MMP-2 and TIMP-2 expressions showed a positive correlation with the grades, stages and metastatic capacities of tumors (Spearman’s correlation, p < 0.05). Concomitant increase in the expression of TIMP-2 and MMP-2 suggested that the rate of MMP-2/TIMP-2 expression is a better marker for characterization of MMP-2 concentration. High expression and/or activity of MMP-2 were linked with poorer survival in OSCC cases, while TIMPs have been shown to apparently act as either growth-stimulating or suppressor factors for tumors. It was also revealed that MMP-2 and TIMP-2 were secreted by both tumor cells and stromal cells. A new concept, supposing the dynamic, anticancer partnership between the residual genome stabilizer machinery of tumor cells and defensive cells adjacent to tumors, may illuminate the controversial results. In conclusion, the stronger the infiltrative and metastatic capacity of cancers, the higher is the rate of MMP-2/TIMP-2 expression helping the arrival of humoral and cellular anticancer forces.



Similar content being viewed by others
References
Nagase H, Woessner JF Jr (1999) Matrix metalloproteinases. J Biol Chem 274:21491–21494
de Vicente JC, Fresno MF, Villalain L et al (2005) Expression and clinical significance of matrix metalloproteinase-2 and matrix metalloproteinase-9 in oral squamous cell carcinoma. Oral Oncol 41:283–293
Ruokolainen H, Paakko P, Turpeenniemi-Hujanen T (2006) Tissue and circulating immunoreactive protein for MMP-2 and TIMP-2 in head and neck squamous cell carcinoma--tissue immunoreactivity predicts aggressive clinical course. Mod Pathol 19(208):217
Lambert E, Dasse E, Haye B et al (2004) TIMPs as multifacial proteins. Crit Rev Oncol Hematol 49:187–198
Gaiotto MA, Focchi J, Ribalta JL et al (2004) Comparative study of MMP-2 (matrix metalloproteinase 2) immune expression in normal uterine cervix, intraepithelial neoplasias, and squamous cells cervical carcinoma. Am J Obstet Gynecol 190:1278–1282
Nagase H, Visse R, Murphy G (2006) Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res 69:562–573
Ra HJ, Parks WC (2007) Control of matrix metalloproteinase catalytic activity. Matrix Biol 26:587–596
Gorogh T, Beier UH, Baumken J et al (2006) Metalloproteinases and their inhibitors: influence on tumor invasiveness and metastasis formation in head and neck squamous cell carcinomas. Head Neck 28:31–39
Bhuvarahamurthy V, Kristiansen GO, Johannsen M et al (2006) In situ gene expression and localization of metalloproteinases MMP1, MMP2, MMP3, MMP9, and their inhibitors TIMP1 and TIMP2 in human renal cell carcinoma. Oncol Rep 15:1379–1384
Shima I, Sasaguri Y, Kusukawa J et al (1992) Production of matrix metalloproteinase-2 and metalloproteinase-3 related to malignant behavior of esophageal carcinoma. A clinicopathologic study. Cancer 70:2747–2753
Bindhu OS, Ramadas K, Sebastian P et al (2006) High expression levels of nuclear factor kappa B and gelatinases in the tumorigenesis of oral squamous cell carcinoma. Head Neck 28:916–925
Kusukawa J, Sasaguri Y, Shima I et al (1993) Expression of matrix metalloproteinase-2 related to lymph node metastasis of oral squamous cell carcinoma. A clinicopathologic study. Am J Clin Pathol 99:18–23
Imanishi Y, Fujii M, Tokumaru Y et al (2000) Clinical significance of expression of membrane type 1 matrix metalloproteinase and matrix metalloproteinase-2 in human head and neck squamous cell carcinoma. Hum Pathol 31:895–904
Shimada T, Nakamura H, Yamashita K et al (2000) Enhanced production and activation of progelatinase a mediated by membrane-type 1 matrix metalloproteinase in human oral squamous cell carcinomas: implications for lymph node metastasis. Clin Exp Metastasis 18:179–188
O-Charoenrat P, Rhys-Evans PH, Eccles SA (2001) Expression of matrix metalloproteinases and their inhibitors correlates with invasion and metastasis in squamous cell carcinoma of the head and neck. Arch Otolaryngol Head Neck Surg 127:813–820
Hu X, Li D, Zhang W et al (2004) Matrix metalloproteinase-9 expression correlates with prognosis and involved in ovarian cancer cell invasion. Arch Gynecol Obstet 286:1537–1543
Gao ZB, Duan YQ, Zhang L, Chen DW, Ding PT (2005) Expression of matrix metalloproteinase 2 and its tissue inhibitor in oral squamous cell carcinoma. Int J Mol Med 16:599–603
Basset P, Bellocq JP, Wolf C et al (1990) A novel metalloproteinase gene specifically expressed in stromal cells of breast carcinomas. Nature 348:699–704
Bisson C, Blacher S, Polette M et al (2003) Restricted expression of membrane type 1-matrix metalloproteinase by myofibroblasts adjacent to human breast cancer cells. Int J Cancer 105:7–13
Pyke C, Ralfkiaer E, Huhtala P et al (1992) Localization of messenger RNA for Mr 72,000 and 92,000 type IV collagenases in human skin cancers by in situ hybridization. Cancer Res 52:1336–1341
Charous SJ, Stricklin GP, Nanney LB et al (1997) Expression of matrix metalloproteinases and tissue inhibitor of metalloproteinases in head and neck squamous cell carcinoma. Ann Otol Rhinol Laryngol 106:271–278
Ondruschka C, Buhtz P, Motsch C et al (2002) Prognostic value of MMP-2, −9 and TIMP-1,-2 immunoreactive protein at the invasive front in advanced head and neck squamous cell carcinomas. Pathol Res Pract 198:509–515
Franchi A, Santucci M, Masini E et al (2002) Expression of matrix metalloproteinase 1, matrix metalloproteinase 2, and matrix metalloproteinase 9 in carcinoma of the head and neck. Cancer 95:1902–1910
Monteagudo C, Merino MJ, San-Juan J et al (1990) Immunohistochemical distribution of type IV collagenase in normal, benign, and malignant breast tissue. Am J Pathol 136:585–592
Hong SD, Hong SP, Lee JI, Lim CY (2000) Expression of matrix metalloproteinase-2 and -9 in oral squamous cell carcinomas with regard to the metastatic potential. Oral Oncol 36:207–213
Bramhall SR, Neoptolemos JP, Stamp GW et al (1997) Imbalance of expression of matrix metalloproteinases (MMPs) and tissue inhibitors of the matrix metalloproteinases (TIMPs) in human pancreatic carcinoma. J Pathol 182:347–355
Foda HD, Zucker S (2001) Matrix metalloproteinases in cancer invasion, metastasis and angiogenesis. Drug Discov Today 6:478–482
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174
Kawamata H, Nakashiro K, Uchida D et al (1997) Possible contribution of active MMP2 to lymph-node metastasis and secreted cathepsin L to bone invasion of newly established human oral-squamous-cancer cell lines. Int J Cancer 70:120–127
Polette M, Nawrocki-Raby B, Gilles C et al (2004) Tumour invasion and matrix metalloproteinases. Crit Rev Oncol Hematol 49:179–186
Overall CM, Wrana JL, Sodek J (1989) Independent regulation of collagenase, 72-kDa progelatinase, and metalloendoproteinase inhibitor expression in human fibroblasts by transforming growth factor-beta. J Biol Chem 264:1860–1869
Munshi HG, Wu YI, Mukhopadhyay S et al (2004) Differential regulation of membrane type 1-matrix metalloproteinase activity by ERK 1/2- and p38 MAPK-modulated tissue inhibitor of metalloproteinases 2 expression controls transforming growth factor-beta1-induced pericellularcollagenolysis. J Biol Chem 279:39042–39050
Kato K, Hara A, Kuno T et al (2005) Matrix metalloproteinases 2 and 9 in oral squamous cell carcinomas: manifestation and localization of their activity. J Cancer Res Clin Oncol 131:340–346
Visscher DW, Hoyhtya M, Ottosen SK et al (1994) Enhanced expression of tissue inhibitor of metalloproteinase-2 (TIMP-2) in the stroma of breast carcinomas correlates with tumor recurrence. Int J Cancer 59:339–344
Pickett KL, Harber GJ, DeCarlo AA et al (1999) 92K-GL (MMP-9) and 72 K-GL (MMP-2) are produced in vivo by human oral squamous cell carcinomas and can enhance FIB-CL (MMP-1) activity in vitro. J Dent Res 78:1354–1361
Bjorklund M, Koivunen E (2005) Gelatinase-mediated migration and invasion of cancer cells. Biochim Biophys Acta 1755:37–69
Danilewicz M, Sikorska B, Wagrowska-Danilewicz M (2003) Prognostic significance of the immunoexpression of matrix metalloproteinase MMP2 and its inhibitor TIMP2 in laryngeal cancer. Med Sci Monit 9:MT427
Liu WW, Zeng ZY, Wu QL et al (2005) Overexpression of MMP-2 in laryngeal squamous cell carcinoma: a potential indicator for poor prognosis. Otolaryngol Head Neck Surg 132:395–400
Yoshizaki T, Maruyama Y, Sato H et al (2001) Expression of tissue inhibitor of matrix metalloproteinase-2 correlates with activation of matrix metalloproteinase-2 and predicts poor prognosis in tongue squamous cell carcinoma. Int J Cancer 95:44–50
Verstappen J, Von den Hoff JW (2006) Tissue inhibitors of metalloproteinases (TIMPs): their biological functions and involvement in oral disease. J Dent Res 85:1074–1084
Visscher DW, Hoyhtya M, Ottosen SK et al (1994) Enhanced expression of tissue inhibitor of metalloproteinase-2 (TIMP-2) in the stroma of breast carcinomas correlates with tumor recurrence. Int J Cancer 59:339–344
Katayama A, Bandoh N, Kishibe K et al (2004) Expressions of matrix metalloproteinases in early-stage oral squamous cell carcinoma as predictive indicators for tumor metastases and prognosis. Clin Cancer Res 10:634–640
Kikuchi R, Noguchi T, Takeno S et al (2000) Immunohistochemical detection of membrane-type-1-matrix metalloproteinase in colorectal carcinoma. Br J Cancer 83:215–218
Hayakawa T, Yamashita K, Tanzawa K et al (1992) Growth-promoting activity of tissue inhibitor of metalloproteinases-1 (TIMP-1) for a wide range of cells. A possible new growth factor in serum. FEBS Lett 298:29–32
Ikenaka Y, Yoshiji H, Kuriyama S et al (2003) Tissue inhibitor of metalloproteinases-1 (TIMP-1) inhibits tumor growth and angiogenesis in the TIMP-1 transgenic mouse model. Int J Cancer 105:340–346
Visse R, Nagase H (2003) Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res 92:827–839
Hernandez-Barrantes S, Bernardo M, Toth M et al (2002) Regulation of membrane type-matrix metalloproteinases. Semin Cancer Biol 12:131–138
Brew K, Dinakarpandian D, Nagase H (2000) Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochim Biophys Acta 1477:267–283
Kugler A, Hemmerlein B, Thelen P et al (1998) Expression of metalloproteinase 2 and 9 and their inhibitors in renal cell carcinoma. J Urol 160:1914–1918
Kallakury BV, Karikehalli S, Haholu A et al (2001) Increased expression of matrix metalloproteinases 2 and 9 and tissue inhibitors of metalloproteinases 1 and 2 correlate with poor prognostic variables in renal cell carcinoma. Clin Cancer Res 7:3113–3119
Di Nezza LA, Misajon A, Zhang J et al (2002) Presence of active gelatinases in endometrial carcinoma and correlation of matrix metalloproteinase expression with increasing tumor grade and invasion. Cancer 94:1466–1475
Baker EA, Leaper DJ, Hayter JP et al (2006) The matrix metalloproteinase system in oral squamous cell carcinoma. Br J Oral Maxillofac Surg 44:482–486
Suba Z (2015) DNA-stabilization by the upregulation of estrogen signaling in BRCA gene mutation carriers. Drug Des Devel Ther 9:2663–2675
Schiewer MJ, Knudsen KE (2016) Linking DNA damage and hormone signaling pathways in cancer. Trends Endocrinol Metab 27:216–225
Suba Z (2016) Causal therapy of breast cancer irrelevant of age, tumor stage and ER-status. Recent Pat Anticancer Drug Discov 11:254–266
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shrestha, B., Bajracharya, D., Byatnal, A.A. et al. May High MMP-2 and TIMP-2 Expressions Increase or Decrease the Aggressivity of Oral Cancer?. Pathol. Oncol. Res. 23, 197–206 (2017). https://doi.org/10.1007/s12253-016-0149-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-016-0149-3