Abstract
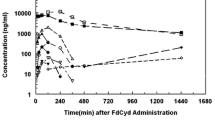
A 1:1 mixture of acriflavine (ACF; CAS 8063-24-9) and guanosine is under evaluation in preclinical studies as a possible antitumor agent. Guanosine is known to potentiate the anti-cancer activity of ACF. We therefore investigated the pharmacokinetics of guanosine following administration of the ACF/guanosine mixture in rats. Rats were given guanosine (1 or 5 mg/kg) or ACF/guanosine (2 or 10 mg/kg) by i.v. bolus; or guanosine (3 or 15 mg/kg) or ACF/guanosine (6 or 30 mg/kg) by i.m. injection. We found that guanosine was rapidly cleared from the blood and transferred to tissues after i.m. administration of ACF/guanosine. The mean plasma half-lives (t1/2) at the α and β phases were 0.091 and 6.86 h, or 0.09 and 7.51 h at a dose of 1 or 5 mg/kg guanosine, respectively. ACF had no effect on the plasma disappearance of guanosine following either i.v. bolus or i.m. administration of the combination mixture. Moreover, the ACF combination with guanosine did not significantly alter the values of MRT, Vdss, and CLt of guanosine. Guanosine exhibited linear pharmacokinetics over the dose range from 1 to 5 mg/kg for i.v. doses and 3 to 15 mg/kg for i.m. doses. The bioavailability of guanosine after i.m. administration was 84% for 3 mg/kg dose and 88% for 15 mg/kg dose. ACF had no effects on biliary and urinary excretion of guanosine after i.m. administration. The cumulative amount of guanosine in urine after i.m. administration was about 5-fold larger than that in bile, indicating that guanosine is mostly excreted into the urine. Guanosine was widely distributed in all tissues examined in this study, but was most highly concentrated in the kidney after i.m. administration, followed by slow excretion to bile or urine. ACF had no effect on the tissue distribution of guanosine following i.m. administration. These characterizations of the pharmacokinetics of guanosine after administration of the ACF/guanosine combination will be useful in providing preclinical and clinical bases for the potential application of this combination to the treatment of cancer.
Similar content being viewed by others
References
Bose. S., Gothoskar. B. P., and Ranadive. K. J., Studies of biological macromolecules: II. Effect of acrifiavine exposure on the synthesis of macromolecules in liver cells in vivo. Experi. Cell Res., 42, 89–98 (1966).
Canellakis, E. S. and Chen, T.-K., Relationship of biochemical drug effects to their antitumor activity. Biochem. Pharmacol., 28, 1971–1976 (1979).
Chakraborty, N. G., Bose, S. R., and Chowdhury, J., Enhancement of immunogenicity of tumor cells by modification of cell surface with acridine dyes. Ind. J. Experiment. Biol., 18, 927–930 (1980).
Chantin, C., Bonin, B., Boulieu, R., and Bory, C., Liquid-chromatographic study of purine metabolism abnormalities in purine nucleoside phosphorylase deficiency. Clin. Chem., 42, 326–328 (1996).
Ferey, L., Herlin, F., Marnay, J., Mandard, A.-M., Catania, R., Lubet, P., Lande, R., and Bloyet, D., Pararosaniline or acriflavine-Schiff staining of epoxy-embedded tissue periodic acid oxidation in ethanol: a method suitable for morphometric and fluorometric analysis of glycogen. Stain Tech., 61, 107–110 (1986).
Hannun, Y. A. and Bell, R. M., Aminoacridines, potent inhibitors of protein kinase C. J. Biol. Chem., 263, 5124–5131 (1988).
Iigo, M., Miwa, M., Ishitsuka, H., and Nitta, K., Potentiation of the chemotherapeutic action of 5′-deoxyfluorouridine in combination with guanosine and related compounds. Cancer Chemother. Pharmacol., 19, 61–64 (1987).
Kim, S. G., Kim, C. W., Ahn, E. T., Lee, K. Y., Hong, E. K., Yoo, B. I., and Han, Y. B., Enhanced anti-tumor effects of acriflavine in combination with guanosine in mice. J. Pharm. Pharmacol., 49, 216–222 (1997).
Macadam, R. F. and Williamson, J., Drug effects on the fine structure of Trypanosomema rhodisience: acriflavine, ethidinn, and antrycide. Ann. Tropi. Med. Parasitol., 68, 291–299 (1974).
Mathe, G., Pontiggia, P., Bourut, C., Clenu, E., and Orbach-Arbouys, S., In vivo eradication of Friend virus as an experimental HIV-model, by combination of zidovudine, acriflavine and an ellipticine analogue: possible application to the treatment of human pre-AIDS? Biomed. Pharmacother., 48, 51–53 (1994).
Plakas, S. M., EL Said, K. R., Bencsath, F. A., Musser, S. M., and Hayton, W. L., Pharmacokinetics, tissue distribution and metabolism of acriflavine and proflavine in the channel catfish (Ictalurus punctatus). Xenobiot., 28, 605–616 (1998).
Roth, D., London, M., and Manjon, M., Binding specificity and affinity of acriflavine for nucleic acids. Stain Tech., 42, 125–132 (1967).
Wiegand, H., Wirz, B., Schweitzer, A., Gross, G., Perez, M. I. R., Andres, H., Kimmerlin, T., Rueping, M., and Seebach, D., Pharmacokinetic investigation of a 14C-Labelled β/α tetrapeptide in rats. Chem. Biodivers., 1, 1812–1828 (2004).
Yamaoka, K., Tanigawara, Y., Nakagawa, Y., and Uno, T., A pharmacokinetic analysis program (MULTI) for microcomputer. J. Pharmcobio. Dynam., 4, 879–885 (1981).
Yu, Z., Hayton, W. L., and Chan, K. K., Characterization of proflavine metabolites in rainbow trout. Drug Metab. Dispos., 25, 431–436 (1977).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, D.H., Choi, K.S., Cho, B.S. et al. Pharmacokinetics of guanosine in rats following intravenous or intramuscular administration of a 1:1 mixture of guanosine and acriflavine, a potential antitumor agent. Arch. Pharm. Res. 31, 1347–1353 (2008). https://doi.org/10.1007/s12272-001-2116-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-2116-z