Abstract
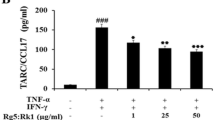
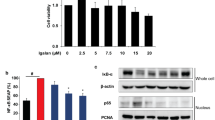
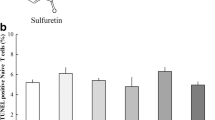
Sulforaphane (4-methylsulfinylbutyl isothiocyanate, SFN) from broccoli has been used a chemopreventive photochemical as detoxification of xenobiotics and anti-inflammatory, however, there is no studies for Th2 chemokine expression through heme oxygenase-1 and NF-κB in keratinocytes. Atopic dermatitis is a chronically relapsing pruritic inflammatory skin disease. SFN is demonstrated to have anti-inflammatory and anti-oxidant effects. This study aimed to define whether and how SFN regulates Th2-related chemokine production in human HaCaT keratinocytes. The level of chemokine expression was measured by reverse transcription polymerase chain reaction (RT-PCR) and signaling study was performed by Western blot analysis. Chemokine production was determined by enzyme-linked immunosorbent assay. Pretreatment with SFN suppressed interferon-γ (IFN-γ) and tumor necrosis factor (TNF)-α- induced thymus- and activation-regulated chemokine (TARC/CCL17) and macrophage-derived chemokine (MDC/CCL22) production in HaCaT keratinocytes. SFN inhibited IFN-γ and TNF-α-induced NF-κB activation as well as STAT1 activation. Interestingly, pretreatment with SFN result in significantly suppressed IFN-γ and TNF-α-induced TARC/CCL17 and MDC/CCL22 production through the induction of HO-1. This suppression was completely abolished by HO-1 siRNA. Furthermore, Carbon monoxide, but not other end products of HO-1 activity, also suppressed IFN-γ and TNF-α-induced TARC/CCL17 and MDC/CCL22 production. These results demonstrate that SFN has an inhibitory role in IFN-γ and TNF-α-induced production of TARC/CCL17 and MDC/CCL22 in human HaCaT cells by inhibition of NF-κB activation and induction of HO-1.
Similar content being viewed by others
References
Aggarwal, B. B., Signalling pathways of the TNF superfamily: a double-edged sword. Nat. Rev. Immunol., 3, 745–756 (2003).
Beltrani, V. S., The clinical spectrum of atopic dermatitis. J. Allergy Clin. Immunol., 104, S87–S98 (1999).
Bowler, R. P. and Crapo, J. D., Oxidative stress in airways: is there a role for extracellular superoxide dismutase? Am. J. Respir. Crit. Care Med., 166, S38–S43 (2002).
Bussolati, B., Ahmed, A., Pemberton, H., Landis, R. C., Di Carlo, F., Haskard, D. O., and Mason, J. C., Bifunctional role for VEGF-induced heme oxygenase-1 in vivo: induction of angiogenesis and inhibition of leukocytic infiltration. Blood, 103, 761–766 (2004).
Chang, M., McNinch, J., Elias, C. 3rd, Manthey, C. L., Grosshans, D., Meng, T., Boone, T., and Andrew, D. P., Molecular chloning and functional characterization of a novel CC chemokine, stimulated T cell chemotactic protein (STCP-1) that specifically acts on activated T lymphocytes. J. Biol. Chem., 272, 25229–25237 (1997).
Chen, S., Kapturczak, M. H., Wasserfall, C., Glushakova, O. Y., Campbell-Thompson, M., Deshane, J. S., Joseph, R., Cruz, P. E., Hauswirth, W. W., Madsen, K. M., Croker, B. P., Berns, K. I., Atkinson, M. A., Flotte, T. R., Tisher, C. C., and Agarwal, A., Interleukin 10 attenuates neointimal proliferation and inflammation in aortic allografts by a heme oxygenase-dependent pathway. Proc. Natl. Acad. Sci. U.S.A., 102,7251–7256 (2005).
Choi, B. M., Pae, H. O., Jeong, Y. R., Oh, G. S., Jun, C. D., Kim, B. R., and Chung, H. T., Overexpression of heme oxygenase (HO)-1 renders Jurkat T cells resistant to fasmediated apoptosis: involvement of iron released by HO-1. Free Radic. Biol. Med., 36, 858–871 (2004).
Choi, S. and Singh, S. V., Bax and Bak are required for apoptosis induction by sulforaphane, a cruciferous vegetable-derived cancer chemopreventive agent. Cancer Res., 65, 2035–2043 (2005).
Clark, J. E., Foresti, R., Green, C. J., and Motterlini, R., Dynamics of haem oxygenase-1 expression and bilirubin production in cellular protection against oxidative stress. Biochem. J., 348, 615–619 (2000).
Clarke, J. D., Dashwood, R. H., and Ho, E., Multi-targeted prevention of cancer by sulforaphane. Cancer Lett., 269, 291–304 (2008).
Darnell, J. E., Jr., Kerr, I. M., and Stark, G. R., Jak-STAT1 pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science, 264, 1415–1421 (1994).
Godiska, R., Chantry, D., Raport, C. J., Sozzani, S., Allavena, P., Leviten, D., Mantovani, A., and Gray, P. W., Human macrophage-derived chemokine (MDC), a novel chemoattractant for monocytes, monocyte-derived dendritic cells, and natural killer cells. J. Exp. Med., 185, 1595–1604 (1997).
Haagerup, A., Bjerke, T., Schiotz, P. O., Dahl, R., Binderup, H. G., Tan, Q., and Kruse, T. A., Atopic dermatitis: a total genome-scan for susceptibility genes. Acta Derm. Venereol., 84, 346–352 (2004).
Heiss, E., Herhaus, C., Klimo, K., Bartsch, H., and Gerhäuser, C., Nuclear factor kappa B is a molecular target for sulforaphane-mediated anti-inflammatory mechanisms. J. Biol. Chem., 276, 32008–32015 (2001).
Hino, R., Kobayashi, M., Mori, T., Orimo, H., Shimauchi, T., Kabashima, K., and Tokura, Y., Inhibition of T helper 2 chemokine production by narrowband ultraviolet B in cultured keratinocytes. Br. J. Dermatol., 156, 830–837 (2007).
Hu, R., Khor, T. O., Shen, G., Jeong, W. S., Hebbar, V., Chen, C., Xu, C., Reddy, B., Chada, K., and Kong, A. N., Cancer chemoprevention of intestinal polyposis in ApcMin/+ mice by sulforaphane, a natural product derived from cruciferous vegetable. Carcinogenesis, 27, 2038–2046 (2006).
Imai, T., Chantry, D., Raport, C. J., Wood, C. L., Nishimura, M., Godiska, R., Yoshie, O., and Gray, P. W., Macrophagederived chemokines is a functional ligand for the CC chemokine receptor 4. J. Biol. Chem., 273, 1764–1768 (1998).
Ju, S. M., Song, H. Y., Lee, S. J., Seo, W. Y., Sin, D. H., Goh, A. R., Kang, Y. H., Kang, I. J., Won, M. H., Yi, J. S., Kwon, D. J., Bae, Y. S., Choi, S. Y., and Park, J., Suppression of thymus- and activation-regulated chemokine (TARC/CCL17) production by 1,2,3,4,6-penta-O-galloyl-β-D-glucose via blockade of NF-κB and STAT1 activation in the HaCaT cells. Biochem. Biophys. Res. Commun., 387, 115–120 (2009).
Kapturczak, M. H., Wasserfall, C., Brusko, T., Campbell-Thompson, M., Ellis, T. M., and Atkinson, M. A., Heme oxygenase-1 modulates early inflammatory responses: evidence from the heme oxygenase-1-deficient mouse. Am. J. Pathol., 165, 1045–1053 (2004).
Kirino, M., Kirino, Y., Takeno, M., Nagashima, Y., Takahashi, K., Kobayashi, M., Murakami, S., Hirasawa, T., Ueda, A., Aihara, M., Ikezawa, Z., and Ishigatsubo, Y., Heme oxygenase 1 attenuates the development of atopic dermatitislike lesions in mice: implications for human disease. J. Allergy Clin. Immunol., 122, 290–297 (2008).
Kong, A. N., Yu, R., Hebbar, V., Chen, C., Owuor, E., Hu, R., and Ee, R., Mandlekar, S., Signal transduction events elicited by cancer prevention compounds. Mutat. Res., 480–481, 231–241 (2001).
Li, H., van Berlo, D., Shi, T., Speit, G., Knaapen, A. M., Borm, P. J., Albrecht, C., and Schins, R. P., Curcumin protects against cytotoxic and inflammatory effects of quartz particles but causes oxidative DNA damage in a rat lung epithelial cell line. Toxicol. Appl. Pharmacol., 227, 115–124 (2008).
Lin, W., Wu, R. T., Wu, T., Khor, T. O., Wang, H., and Kong, A. N., Sulforaphane suppressed LPS-induced inflammation in mouse peritoneal macrophages through Nrf2 dependent pathway. Biochem Pharmacol., 76, 967–973 (2008).
Mi, L., Xiao, Z., Hood, B. L., Dakshanamurthy, S., Wang, X., Govind, S., Conrads, T. P., Veenstra, T. D., and Chung, F. L., Covalent binding to tubulin by isothiocyanates. A mechanism of cell growth arrest and apoptosis. J. Biol. Chem., 283, 22136–22146 (2008).
Minamino, T., Christou, H., Hsieh, C. M., Liu, Y., Dhawan, V., Abraham, N. G., Perrella, M. A., Mitsialis, S. A., and Kourembanas, S., Targeted expression of heme oxygenase-1 prevents the pulmonary inflammatory and vascular responses to hypoxia. Proc. Natl. Acad. Sci. U.S.A., 98, 8798–8803 (2001).
Nakazato, J., Kishida, M., Kuroiwa, R., Fujiwara, J., Shimoda, M., and Shinomiya, N., Serum levels of Th2 chemokines, CCL17, CCL22, and CCL27, were the important markers of severity in infantile atopic dermatitis. Pediatr. Allergy Immunol., 19, 605–613 (2008).
Nakao, A., Moore, B. A., Murase, N., Liu, F., Zuckerbraun, B. S., Bach, F. H., Choi, A. M., Nalesnik, M. A., Otterbein, L. E., and Bauer, A. J., Immunomodulatory effects of inhaled carbon monoxide on rat syngeneic small bowel graft motility. Gut, 52, 1278–1285 (2003).
Nickel, R., Beck, L. A., Stellato, C., and Schleimer, R. P., Chemokines and allergic disease. J. Allergy Clin. Immunol., 104, 723–742 (1999).
Perocco, P., Bronzetti, G., Canistro, D., Valgimigli, L., Sapone, A., Affatato, A., Pedulli, G. F., Pozzetti, L., Broccoli, M., Iori, R., Barillari, J., Sblendorio, V., Legator, M. S., Paolini, M., and Abdel-Rahman, S. Z., Glucoraphanin, the bioprecursor of the widely extolled chemopreventive agent sulforaphane found in broccoli, induces phase-I xenobiotic metabolizing enzymes and increases free radical generation in rat liver. Mutat. Res., 595, 125–136 (2006).
Qi, X. F., Kim, D. H., Yoon, Y. S., Li, J. H., Jin, D., Teng, Y. C., Kim, S. K., and Lee, K. J., Fluvastatin inhibits expression of the chemokine MDC/CCL22 induced by interferong in HaCaT cells, a human keratinocyte cell line. Br. J. Pharmacol., 157, 1441–1450 (2009).
Rossi, D. and Zlotnik, A., The biology of chemokines and their receptors. Annu. Rev. Immunol., 18, 217–242 (2000).
Ryter, S. W. and Otterbein, L. E., Carbon monoxide in biology and medicine. Bioessays, 26, 270–280 (2004).
Schreiber, E., Matthias, P., Muller, M. M., and Schaffner, W., Rapid detection of octamer binding proteins with ‘miniextracts’, prepared from a small number of cells. Nucleic. Acids Res., 17, 6419 (1989).
Shibata, A., Nakagawa, K., Yamanoi, H., Tsuduki, T., Sookwong, P., Higuchi, O., Kimura, F., and Miyazawa, T., Sulforaphane suppresses ultraviolet B-induced inflammation in HaCaT keratinocytes and HR-1 hairless mice. J. Nutr. Biochem., 21, 702–709 (2010).
Shimada, Y., Takehara, K., and Sato, S., Both Th2 and Th1 chemokines (TARC/CCL17, MDC/CCL22, Mig/CCL9) are elevated in sera form patients with atopic dermatitis. J. Dermatol. Sci., 34, 201–218 (2004).
Takamiya, R., Murakami, M., Kajimura, M., Goda, N., Makino, N., Takamiya, Y., Yamaguchi, T., Ishimura, Y., Hozumi, N., and Suematsu, M., Stabilization of mast cells by heme oxygenase-1: an anti-inflammatory role. Am. J. Physiol. Heart Circ. Physiol., 283, H861–H870 (2002).
Tsukahara, H., Shibata, R., Ohshima, Y., Todoroki, Y., Sato, S., Ohta, N., Hiraoka, M., Yoshida, A., Nishima, S., and Mayumi, M., Oxidative stress and altered antioxidant defenses in children with acute exacerbation of atopic dermatitis. Life. Sci., 72, 2509–2516 (2003).
Vestergaard, C., Yoneyama, H., Murai, M., Nakamura, K., Tamaki, K., Terashima, Y., Imai, T., Yoshie, O., Irimura, T., Mizutani, H., and Matsushima, K., Overproduction of Th2-specific chemokines in NC/Nga mic exhibiting atopic dermatitis-like lesions. J. Clin. Invest., 104, 1097–1105 (1999).
Vestergaard, C., Bang, K., Gesser, B., Yoneyama, H., Matsushima, K., and Larsen, C. G., A Th2 chemokine, TARC, produced by keratinocytes may recruit CLA+CCR4+ lymphocytes into lesional atopic dermatitis skin. J. Invest. Dermatol., 115, 640–646 (2000).
Xu, C., Shen, G., Chen, C., Gélinas, C., and Kong, A. N., Suppression of NF-kappaB and NF-kappaB-regulated gene expression by sulforaphane and PEITC through IkappaBalpha, IKK pathway in human prostate cancer PC-3 cells. Oncogene, 24, 4486–4495 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeong, SI., Choi, BM. & Jang, S.I. Sulforaphane suppresses TARC/CCL17 and MDC/CCL22 expression through heme oxygenase-1 and NF-κB in human keratinocytes. Arch. Pharm. Res. 33, 1867–1876 (2010). https://doi.org/10.1007/s12272-010-1120-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-1120-6