Abstract
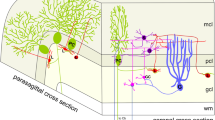
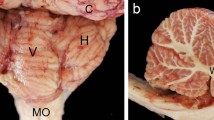
The turtle's cerebellum (Cb) is an unfoliated sheet, so the topography of its entire cortex can be easily studied physiologically by optical recordings. However, unlike the mammalian Cb, little is known about the topography of turtle Purkinje cells (PCs). Here, topography was examined using calbindin-D28K immunohistochemistry of adult and hatchling turtles (Trachemys scripta elegans, 2.5–15 cm carapace length). Each Cb was flattened between two Sylgard sheets and fixed in paraformaldehyde. Sections (52 µm thick) were cut parallel to the flattened cortex (tangential), resulting in calbindin-immunolabeled PCs being localized to three to six sections for each turtle. PC position and size were quantified using Neurolucida Image Analysis system. Although hatchling Cb were medial-laterally narrower (3.0 vs. 6.5 mm) and rostral-caudally shorter (2.5 vs. 5.5 mm) than adult Cb, both averaged near 15,000 PCs distributed uniformly. Hatchling PCs were smaller than adult PCs (178 vs. 551 µm2) and more densely packed (2,180 vs. 625 cells/mm2). Calbindin immunoreactivity also labeled non-PCs along the Cb's marginal rim and its caudal pole. Many of these were very small (22.9 µm2) ovoid-shaped cells clustered together, possibly proliferating external granule layer cells. Other labeled cells were larger and fusiform-shaped (12.6 × 33.4 µm) adjacent to inner granule cells along the marginal rim, suggestive of migrating cells. It is not known whether these are new neurons being generated within the adult and hatchling Cb and if they connect to efferent and afferent paths. Based on these anatomical findings, we suggest that unique physiological features may exist along the rim of the turtle Cb.








Similar content being viewed by others
References
Anderson CW, Keifer J (1997) The cerebellum and red nucleus are not required for in vitro classical conditioning of the turtle abducens nerve response. J Neurosci 17:9736–9745
Keifer J, Armstrong KE, Houk JC (1995) In vitro classical conditioning of abducens nerve discharge in turtles. J Neurosci 15:5036–5048
Reali C, Russo RE (2005) An integrated spinal cord-hindlimbs preparation for studying the role of intrinsic properties in somatosensory information processing. J Neurosci Methods 142:317–326
Senseman DM (1996) Correspondence between visually evoked voltage-sensitive dye signals and synaptic activity recorded in cortical pyramidal cells with intracellular microelectrodes. Vis Neurosci 13:963–977
Tolbert DL, Conoyer B, Ariel M (2004) Quantitative analysis of granule cell axons and climbing fiber afferents in the turtle cerebellar cortex. Anat Embryol 209:49–58
Chesler M, Chan CY (1988) Stimulus-induced extracellular pH transients in the in vitro turtle cerebellum. Neuroscience 27:941–948
Krizaj D, Rice ME, Wardle RA, Nicholson C (1996) Water compartmentalization and extracellular tortuosity after osmotic changes in cerebellum of Trachemys scripta. J Physiol 492:887–896
Okada YC, Huang JC, Rice ME, Tranchina D, Nicholson C (1994) Origin of the apparent tissue conductivity in the molecular and granular layers of the in vitro turtle cerebellum and the interpretation of current source-density analysis. J Neurophysiol 72:742–753
Perez-Pinzon MA, Rosenthal M, Lutz PL, Sick TJ (1992) Anoxic survival of the isolated cerebellum of the turtle Pseudemis scripta elegans. J. Comp Physiol, B. Biochem Syst Environ Physiol 162:68–73
Brown ME, Ariel M (2009) Topography and response timing of intact cerebellum stained with absorbance voltage-sensitive dye. J Neurophysiol 101:474–490
Martin J, Ariel M (2005) Localization of GABA ([gamma]-aminobutyric acid) markers in the turtle's basal optic nucleus. Brain Res 1066:109–119
Rosenberg AF, Ariel M (1990) Visual-response properties of neurons in turtle basal optic nucleus in vitro. J Neurophysiol 63:1033–1045
Graham RC Jr, Karnovsky MJ (1966) The early stages of absorption of injected horseradish peroxidase in the proximal tubules of mouse kidney: ultrastructural cytochemistry by a new technique. J Histochem Cytochem 14:291–302
Abercrombie M (1946) Estimation of nuclear population from microtome sections. Anat Rec 94:239–247
Larsell O (1932) The cerebellum of reptiles: chelonians and alligator. J Comp Neurol 41:59–94
Yan XX, Garey LJ (1998) Complementary distributions of calbindin, parvalbumin and calretinin in the cerebellar vermis of the adult cat. J Hirnforsch 39:9–14
Fortin M, Marchand R, Parent A (1998) Calcium-binding proteins in primate cerebellum. Neurosci Res 30:155–168
Scotti AL, Nitsch C (1992) Differential Ca2+ binding properties in the human cerebellar cortex: distribution of parvalbumin and calbindin D-28 k immunoreactivity. Anat Embryol 185:163–167
Sequier JM, Hunziker W, Richards G (1988) Localization of calbindin D28 mRNA in rat tissues by in situ hybridization. Neurosci Lett 86:155–160
Mutema GK, Rhoten WB (1994) Occurrence and localization of calbindin-D28K in kidney and cerebellum of the slider turtle, Trachemys scripta. Anat Rec 239:185–190
Bangma GC, ten Donkelaar HJ, Pellegrino A (1983) Cerebellar corticonuclear projections in the red-eared turtle Pseudemys scripta elegans. J Comp Neurol 215:258–274
Gerrits NM, Voogd J (1973) The distribution of the Purkinje cells in the cerebellum of testudo hermanni (turtle). Acta Morphol Neerl Scand 11:357–358
Whitney ER, Kempera TL, Rosenea DL, Baumana ML, Blatta GJ (2007) Calbindin-D28k is a more reliable marker of human Purkinje cells than standard Nissl stains: a stereological experiment. J Neurosci Methods 168:42–47
Bell CC, Meek J, Yang JY (2005) Immunocytochemical identification of cell types in the mormyrid electrosensory lobe. J Comp Neurol 483:124–142
Uray NJ, Gona AG (1999) Calbindin immunoreactivity in the auricular lobe and interauricular granular band of the cerebellum in bullfrogs. Brain Behav Evol 53:10–19
Kevetter GA, Leonard RB (1997) Use of calcium-binding proteins to map inputs in vestibular nuclei of the gerbil. J Comp Neurol 386:317–327
Rodriguez-Moldes I, Timmermans JP, Adriaensen D, De Groodt-Lasseel MH, Scheuermann DW, Anadon R (1990) Immunohistochemical localization of calbindin-D28K in the brain of a cartilaginous fish, the dogfish (Scyliorhinus canicula L.). Acta Anat 137:293–302
Fujii JT, Lucaj Z (1993) Calcium-binding proteins in the chick Edinger Westphal nucleus. Brain Res 605:200–206
Mugnaini E, Atluri RL, Houk JC (1974) Fine structure of granular layer in turtle cerebellum with emphasis on large glomeruli. J Neurophysiol 37:1–29
Kunzle H (1983) Spinocerebellar projections in the turtle. Observations on their origin and terminal organization. Exp Brain Res 53:129–141
Armstrong CL, Hawkes R (2000) Pattern formation in the cerebellar cortex. Biochem Cell Biol 78:551–562
Hawkes R (1997) An anatomical model of cerebellar modules. Prog Brain Res 114:39–52
Brand S, Mugnaini E (1980) Pattern of distribution of acetylcholinesterase in the cerebellar cortex of the pond turtle, with emphasis on parallel fibers. A histochemical and biochemical study. Anat Embryol 158:271–287
Cruce WL, Nieuwenhuys R (1974) The cell masses in the brain stem of the turtle Testudo hermanni; a topographical and topological analysis. J Comp Neurol 156:277–306
Larsell O (1967) The comparative anatomy and histology of the cerebellum from myxinoids through birds. University of Minnesota Press, Minneapolis
Voogd J (2003) The human cerebellum. J Chem Neuroanat 26:243–252
Chambers WW, Sprague JM (1955) Functional localization in the cerebellum. I. Organization in longitudinal cortico-nuclear zones and their contribution to posture, both extrapyramidal and pyramidal. J Comp Neurol 103:105–130
Stamatakis A, Barbas H, Dermon CR (2004) Late granule cell genesis in quail cerebellum. J Comp Neurol 474:173–189
Uray NJ (1985) Early stages in the formation of the cerebellum in the frog. J Comp Neurol 232:129–142
Martin J, Chanasue L, Ariel M (2008) Neurogenesis in the turtle cerebellum. Soc Neurosci Abstr 32
Zupanc GK, Horschke I (1995) Proliferation zones in the brain of adult gymnotiform fish: a quantitative mapping study. J Comp Neurol 353:213–233
Eriksson PS, Perfilieva E, Bjork-Eriksson T, Alborn AM, Nordborg C, Peterson DA et al (1998) Neurogenesis in the adult human hippocampus. Nat Med 4:1313–1317
Bedard A, Parent A (2004) Evidence of newly generated neurons in the human olfactory bulb. Brain Res Dev Brain Res 151:159–168
Goldman SA, Nottebohm F (1983) Neuronal production, migration, and differentiation in a vocal control nucleus of the adult female canary brain. Proc Natl Acad Sci U S A 80:2390–2394
Chetverukhin VK, Polenov AL (1993) Ultrastructural radioautographic analysis of neurogenesis in the hypothalamus of the adult frog, Rana temporaria, with special reference to physiological regeneration of the preoptic nucleus. I. Ventricular zone cell proliferation. Cell Tissue Res 271:341–350
Polenov AL, Chetverukhin VK (1993) Ultrastructural radioautographic analysis of neurogenesis in the hypothalamus of the adult frog, Rana temporaria, with special reference to physiological regeneration of the preoptic nucleus. II. Types of neuronal cells produced. Cell Tissue Res 271:351–362
Perez-Canellas MM, Garcia-Verdugo JM (1996) Adult neurogenesis in the telencephalon of a lizard: a [3H] thymidine autoradiographic and bromodeoxyuridine immunocytochemical study. Brain Res Dev Brain Res 93:49–61
Zupanc GK, Horschke I, Ott R, Rascher GB (1996) Postembryonic development of the cerebellum in gymnotiform fish. J Comp Neurol 370:443–464
Maeyama K, Nakayasu H (2000) Postembryonic neurogenesis in zebrafish (Danio rerio) brain: presence of two different systems. Zoolog Sci 17:959–966
Perez-Canellas MM, Font E, Garcia-Verdugo JM (1997) Postnatal neurogenesis in the telencephalon of turtles: evidence for nonradial migration of new neurons from distant proliferative ventricular zones to the olfactory bulbs. Brain Res Dev Brain Res 101:125–137
Fernandez A, Radmilovich M, Trujillo-Cenoz O (2002) Neurogenesis and gliogenesis in the spinal cord of turtles. J Comp Neurol 453:131–144
Radmilovich M, Fernandez A, Trujillo-Cenoz O (2003) Environment temperature affects cell proliferation in the spinal cord and brain of juvenile turtles. J Exp Biol 206:3085–3093
ten Donkelaar HJ, Lammens M, Wesseling P, Thijssen HO, Renier WO (2003) Development and developmental disorders of the human cerebellum. J Neurol 250:1025–1036
Hatten ME (1999) Central nervous system neuronal migration. Annu Rev Neurosci 22:511–539
Brown ME, Ariel M (2008) Stimulation of an eighth cranial nerve activates discrete bilateral regions of the lateral edge of the turtle cerebellum. Soc Neurosci Abstr 32
Acknowledgments
We thank Dr. John Martin for his help with the Neurolucida system and for comments on the manuscript. This work was supported by grant NS-46499 from the National Institute of Neurological Disorders and Stroke.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ariel, M., Ward, K.C. & Tolbert, D.L. Topography of Purkinje Cells and Other Calbindin-Immunoreactive Cells Within Adult and Hatchling Turtle Cerebellum. Cerebellum 8, 463–476 (2009). https://doi.org/10.1007/s12311-009-0123-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-009-0123-7