Abstract
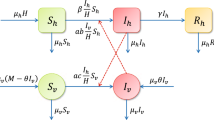
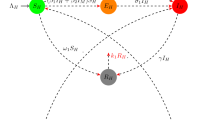
In this paper, a deterministic model is proposed to perform a thorough investigation of the transmission dynamics of Zika fever. Our model, in particular, takes into account the effects of horizontal as well as vertical disease transmission of both humans and vectors. The expression for basic reproductive number \(R_0\) is determined in terms of horizontal and vertical disease transmission rates. An in-depth stability analysis of the model is performed, and it is shown, that model is locally asymptotically stable when \(R_0 < 1\). In this case, there is a possibility of backward bifurcation in the model. With the assumption that total population is constant, we prove that the disease free state is globally asymptotically stable when \(R_0 < 1\). It is also shown that disease strongly uniformly persists when \(R_0> 1\) and there exists an endemic equilibrium which is unique if the total population is constant. The endemic state is locally asymptotically stable when \(R_0> 1\).








Similar content being viewed by others
References
Agusto, F.B., Bewick, S., Fagan, W.F.: Mathematical model for Zika virus dynamics with sexual transmission route. Ecol. Complex. 29, 1–92 (2017)
Boorman, J.P., Porterfield, J.S.: A simple technique for infection of mosquitoes with viruses; transmission of Zika virus. Trans. R. Soc. Trop. Med. Hyg. 50(3), 238–242 (1956)
Besnard, M., Lastre, S., Teissier, A., Cao-Lormeau, V.M., Musso, D.: Evidence of perinatal transmission of Zika virus. Eurosurveillance 19(13), 1–4 (2014)
Compartmental Models in Epidemiology. Mathematical Epidemiology, Lecture Notes in Mathematics, vol. 1945. Springer, Berlin (2008)
Castillo-Chavez, C., Song, B.: Dynamical model of tuberclosis and their applications. Math. Biosci. Eng. 1(2), 361–404 (2004)
Castillo-Chavez, C., Thieme, H.R.: Asymptotically autonomous epidemic models. In: Arino, O., Axelrod, D., Kimmel, M., Langlais, M. (eds.) Mathematical Population Dynamics: Analysis of Heterogeneity, 1: Theory of Epidemics, pp. 33–50. Winnipeg, Wuerz (1993)
Centers for disease control and prevention (CDC). Symptoms, diagnosis, and treatment. http://www.cdc.gov/zika/symptoms/
Calisher, C.H., Gould, E.A.: Taxonomy of the virus family Flaviviridae. Adv. Virus Res. 59, 1–19 (2003)
Gao, D., Lou, Y., He, D., Porco, T.C., Kuang, Y., Shigui, R., Gerardo, C.: Prevention and control of Zika as a mosquito-borne and sexually transmitted disease: a mathematical modeling analysis. Nat. Sci. Rep. 6, 28070 (2016). doi:10.1038/srep28070
Derouich, M., Boutayeb, A.: Dengue fever: mathematical modelling and computer simulation. Appl. Math. Comput. 177(2), 528–544 (2006)
Donnelly, C.A., Ghani, A.C., Leung, G.M., Hedley, A.J., Fraser, C., Riley, S., Anderson, R.M.: Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 361(9371), 1832 (2003)
Esteva, L., Vargas, C.: Analysis of a dengue disease transmission model. Math. Biosci. 150(2), 131–151 (1998)
Esteva, L., Vargas, C.: A model for Dengue disease with variable human population. J. Math. Biol. 38(3), 220–240 (1999)
Esteva, L., Vargas, C.: Influence of vertical and mechanical transmission on the dynamics of dengue disease. Math. Biosci. 167(1), 51–64 (2000)
Esteva, L., Vargas, C.: Coexistence of different serotypes of dengue virus. J. Math. Biol. 46(1), 31–47 (2003)
European Centre for Disease prevention and Control (ECDC).: Microcephaly in Brazil potentially linked to the Zika virus epidemic. http://ecdc.europa.eu/en/publications/Publications/zika-virus-americas-association-with-microcephaly-rapid-risk-assessment.pdf
Kelser, E.A.: Meet dengue’s cousin, Zika. Microbes Infect. 18, 163–166 (2016). doi:10.1016/j.micinf.2015.12.003
Ferguson, N., Anderson, R., Gupta, S.: The effect of antibody-dependent enhancement on the transmission dynamics and persistence of multiple-strain pathogens. Proc. Natl. Acad. Sci. 96(2), 790–794 (1999)
Garba, S.M., Gumel, A.B., Abu Bakar, M.R.: Backward bifurcations in dengue transmission dynamics. Math. Biosci. 215(1), 11–25 (2008)
Garba, S.M., Gumel, A.B., Abu Bakar, M.R.: Effect of cross-immunity on the transmission dynamics of two strains of dengue. Int. J. Comput. Math. 87(10), 2361–2384 (2010)
Gumel, A.B., Ruan, S., Day, T., Watmough, J., Brauer, F., van den Driessche, P., Sahai, B.M.: Modelling strategies for controlling SARS outbreaks. Proc. R. Soc. Ser. B 271, 2223–2232 (2003)
Hayes, E.B.: Zika Virus Outside Africa. Emerg. Infect. Dis. 15(9), 1347–1350 (2009)
Hethcote, H.W.: The mathematics of infectious diseases. SIAM Rev. 42, 599–653 (2000)
Imran, M., Hassan, M., Khan, A.: A comparison of a deterministic and stochastic model for hepatitis C with an isolation stage. J. Biol. Dyn. 7, 276–304 (2013)
Hethcote, H.W., Thieme, H.R.: Stability of the endemic equilibrium in epidemic models with subpopulations. Math. Biosci. 75, 205–227 (1985)
Hethcote, H.W., van Ark, J.W.: Epidemiology models for heterogeneous populations: proportionate mixing, parameter estimation, and immunization programs. Math. Biosci. 84, 85–118 (1987)
Kautner, I., Robinson, M.J., Kuhnle, U.: Dengue virus infection: epidemiology, pathogenesis, clinical presentation, diagnosis, and prevention. J. Pediatr. 131(4), 516–524 (1997)
Kindhauser, M., Allen, T., Frank, V., Santhana, R., Dye, C.: Zika: the origin and spread of a mosquito-borne virus. WHO, Geneva (2016)
Khan, A., Hassan, M., Imran, M.: Estimating the basic reproduction number for single-strain dengue fever epidemics. Infect. Dis. Poverty 3, 12 (2014)
Kucharski, A.J., Funk, S., Eggo, R.M., Mallet, H., Edmunds, W., Nilles, E.: Transmission dynamics of Zika virus in island populations: a modelling analysis of the 2013–14 French Polynesia outbreak. PLoS Negl. Trop. Dis. 10, e0004726 (2016)
Lanciotti, R.S., Kosoy, O.L., Laven, J.J.: Genetic and serologic properties of Zika virus associated with an epidemic, Yap State, Micronesia 2007. Emerg. Infect. Dis. 14(8), 1232–1239 (2008)
Li, M.Y., Smith, H.L., Wang, L.: Global dynamics of an SEIR epidemic model with vertical transmission. SIAM J. Appl. Math. 62(1), 58–69 (2001)
Lipsitch, M., Cohen, T., Cooper, B., Robins, J.M., Ma, S., James, L., Murray, M.: Transmission dynamics and control of severe acute respiratory syndrome. Science 300, 1966–1970 (2003)
Lloyd-Smith, J.O., Galvani, A.P., Getz, W.M.: Curtailing transmission of severe acute respiratory syndrome within a community and its hospital. Proc. R. Soc. Lond. Ser. B Biol. Sci. 170, 1979–1989 (2003)
Attar, N.: ZIKA virus circulates in new regions. Nat. Rev. Microbiol. 14, 62 (2016)
PAHO issues Zika virus alert, Centre for Infectious Disease Control Policy (2015). http://www.cidrap.umn.edu/news-perspective/2015/12/paho-issues-zika-virus-alert
Pan American Health Organization: Neurological syndrome, congenital malformations and Zika virus infection. Implications for public health in the America (2015). http://reliefweb.int/report/world/epidemiological-alert-neurological-syndrome-congenital-malformations-and-zika-virus
Salceanu, P.L.: Robust uniform persistence in discrete and continuous dynamical systems using Lyapunov Exponents. Math. Biosci. Eng. 8(3), 807–825 (2011)
Semenza, J.C., Zeller, H.: Integrated surveillance for prevention and control of emerging vector-borne diseases in Europe. Eur. Surveill. 19, 3 (2014)
Smith, H.L., Thieme, H.: Dynamical systems and population persistence, Graduate Studies in Mathematics, vol. 118. American Mathematical Society, Providence (2011)
Summers, D.J., Acosta, R.W.: Zika virus in an American recreational traveler. J. Travel Med. 22(5), 338–340 (2015)
Thangamani, S., Huang, J., Hart, C., Guzman, H., Tesh, R.: Vertical transmission of zika virus in Aedes aegypti mosquitoes. Am. J. Trop. Med. Hyg. 95, 1169–1173 (2016)
Thieme, H.R.: Convergence results and a Poincare Bendixson trichotomy for asymptotically autonomous differential equations. J. Math. Biol. 30, 755–763 (1992)
Thieme, H.R.: Asymptotically autonomous differential equations in the plane. Rocky Mt. J. Math. 24, 351–380 (1994)
van den Driessche, P., Watmough, J.: Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 180, 29–48 (2002)
Ventura, C.V., et al.: Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet 387(10015), 228 (2016)
Vivas-Barber, A., Castillo-Chavez, C., Barany, E.: Dynamics of an SAIQR influenza model. Biomath 3, 1–3 (2015)
Wearing, H.J., Rohani, P.: Ecological and immunological determinants of Dengue epidemics. Proc. Natl. Acad. Sci. 103(31), 11802–11807 (2006)
WHO sees Zika outbreak spreading through the Americas. Reuters. http://www.straitstimes.com/world/americas/who-sees-zikaoutbreak-spreading-through-the-americas. Accessed 25 Jan 2016 (2016)
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this article.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Backward Bifurcation in Model (1)
The model given in the paper has the variables: \(S_h, E_h, I_h, R_h, S_v, E_v, I_v\).
Now we redefine these equations by assigning them the following values:
Let \(S_h = x_1, E_h= x_2, I_h= x_3, R_h= x_4, S_v= x_5, E_v= x_6, I_v= x_7\). Also let \(\hat{f} = [f_1,\ldots ,f_7]\) denote the vector field of the original model in terms of \(x_i's\).
Then we have the following model in terms of \(x_i's \)
Now the disease free equilibrium (DFE) of the system is given by \({\varepsilon _0}\) as follows:
Consider the case when \(R_0=1\). Take \(C_{hv}=C_{hv}^*\) as a bifurcation parameter.
The Jacobian of the matrix at the DFE is given by
where:
The above Jacobian matrix of the linearized system has a simple zero eigenvalue (with all other eigenvalues having negative real part). Hence, the Centre Manifold Theory can be used to analyze the dynamics of the system (1). We will use the theorem given by Castillo-Chavez and Song [5].
The Right Eigenvector
The right eigenvector of the Jacobian matrix correspond to zero eigenvalue at \(C_{hv}^*\) is given by: w \(={w_1,\ldots ,w_7}\), where
The Left Eigenvector
Similarly, the left eigenvector of the Jacobian matrix correspond to zero eigenvalue at \(C_{hv}^*\) is given by: v \(={v_1,\ldots ,v_7}\), where
The Non-zero Derivatives

The value of
So, that
The value of
is given by:
Since b is always positive, backward bifurcation occurs whenever \(a>0\).
Appendix 2: Stability of Endemic Steady State
Proof
The proof of Theorem 8 is based on using a Krasnoselskii sub-linearity trick [25, 26].
Rewrite (14) as:
Linearizing the system (21) around the endemic equilibrium \(\displaystyle N_1=(S_h^{\varnothing }, E_h^{\varnothing }, I_h^{\varnothing }, R_h^{\varnothing }, S_v^{\varnothing }, E_v^{\varnothing }, I_v^{\varnothing }) \), gives:
It follows that the Jacobian of the system evaluated at \(N_1\) is:
where \(\displaystyle j_1=\frac{C I_h^{\varnothing }}{N_h}\), \(\displaystyle j_2=\frac{C I_v^{\varnothing }}{N_h}\), \(\displaystyle j_3=\frac{C S_h^{\varnothing }}{N_h}\) and \(\displaystyle j_4=\frac{C S_v^{\varnothing }}{N_h}.\)
Assume that the model (22) has solution of the form
where \(Z=(Z_1, Z_2, Z_3, Z_4, Z_5)\). Substituting Z into (22) gives:
We rearranged the above system of equations as follows. First we move the negative terms in the last four equations of (23) to the respective left-hand sides. Secondly, the last four equations are then re-written in terms of \(Z_1\). We have
Substituting the last five equations in the first equation of (23) leads to:
where \(\displaystyle F_i(\omega )\) for \(i=1,\ldots ,5\) are positive functions of parameters and
such that the matrix M has non-negative entries. Define \(F(\omega )=min_i|1 + F_i|\). It is easy to verify that the equilibrium point \(N_1\) satisfies, \(N_1=MN_1.\) The notation \((MZ)_i\) denotes the ith coordinate of the vector MZ. If Z is a solution of (25), then it is possible to find a minimal positive real number r such that \(||Z||\le r N_1\) [25, 26]. We want to show that \(Re (\omega ) < 0\). Assume, \(Re (\omega ) \ge 0\), and consider the following two cases.
Case 1 \(\omega = 0.\) In this case (23) is a homogeneous linear system. It is easy to show that the determinant of this system is negative, and it follows that the system (23) has a unique solution, given by \(Z = 0\), which correspond to disease free steady state of the model (14).
Case 2 \(\displaystyle \omega \ne 0.\) By our assumption in this case \(|1+F_i(\omega )|> 1.\) Since r is a minimal positive real number, it follows that
Where \(F(\omega )\) is minimal of \(|1+F_i(\omega )|.\) From the second equation of (25), we have
which contradicts \(||Z||>\frac{r}{F(w)}N_1.\) Hence, \(Re (\omega ) < 0\). Thus, all eigenvalues of the characteristic equation associated with the linearized system (14) will have negative real parts. This implies local asymptotical stability of endemic state. \(\square \)
Appendix 3: Model Parameters
The estimated parameters are presented in Table 3.
Rights and permissions
About this article
Cite this article
Imran, M., Usman, M., Dur-e-Ahmad, M. et al. Transmission Dynamics of Zika Fever: A SEIR Based Model. Differ Equ Dyn Syst 29, 463–486 (2021). https://doi.org/10.1007/s12591-017-0374-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12591-017-0374-6